Are you over 18 and want to see adult content?
More Annotations
A complete backup of confessionpost.com
Are you over 18 and want to see adult content?
A complete backup of alsolis.blogspot.com
Are you over 18 and want to see adult content?
A complete backup of bolivar-s.livejournal.com
Are you over 18 and want to see adult content?

A complete backup of lifeguardtrainer.com
Are you over 18 and want to see adult content?
A complete backup of dulivre.blogspot.com
Are you over 18 and want to see adult content?
A complete backup of kalabhairavaguruji.blogspot.com
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of www.biobiochile.cl/noticias/futbol-internacional/chilenos-en-el-exterior/2020/02/27/a-revertir-la-llave-aud
Are you over 18 and want to see adult content?

A complete backup of www.radio.kielce.pl/pl/post-100674
Are you over 18 and want to see adult content?
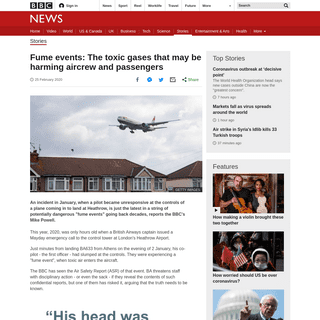
A complete backup of www.bbc.co.uk/news/stories-51633897
Are you over 18 and want to see adult content?

A complete backup of holofote.sapo.pt/celebridades/2020-02-27-afinal-joao-felix-traiu-margarida-corceiro-eis-o-que-as-mensagens-
Are you over 18 and want to see adult content?

A complete backup of www.bbc.co.uk/news/business-51626529
Are you over 18 and want to see adult content?

A complete backup of www.forbes.com/sites/scottmendelson/2020/02/25/invisible-man-review-best-universal-monster-movie-since-mumm
Are you over 18 and want to see adult content?
Text
ACIDS VS. BASES
Acids vs. Bases - Home. Hi! My name is Chloe, I'm a 10th grade chemistry, student and I've created this website so that you can learn about acids, bases, and alkalis! You'll learn their definitions, properties, how to identify them, some calculations, and so much more. CONJUGATE ACIDS & BASES The definitions stated that acids are proton (H+) donors, and bases are proton (H+) acceptors. After a base receives a proton, the remainder becomes a conjugate acid, and after an acid donates a proton, the remainder becomes a conjugate base. Lets make things simple. Think of a proton as a ball being passed between two people like in the gif below.CALCULATIONS
Note: If a page name is blue, click it to visit that page. Now that you understand Strength and Concentration , it's time to learn how to calculate them The PROPERTIES, REACTIONS, AND DEFINITONS Properties of Bases & Alkalis:. pH over 7 (cold colors on pH strip); Bitter; Corrosive; Electrolyte; Turns red litmus paper blue; Produces OH- ions when they dissolve in water (if it does not dissolve, it's a base, if it does, its an alkali; Neutralizes acids to form salt and water; Slippery; Neutralization Reactions: INDICATORS - ACIDS VS. BASES When combined with a base, it turns pink. If it's neutral (pH of 7), it's clear (useful for neutralization reactions). Another common indicator is red litmus paper and blue litmus paper. In a base, litmus paper will turn to or stay blue. In an acid, it will turn to or stay red. (see picture below). Sometimes you'll need to identify ifsomething
TITRATION LAB
To the right is a slideshow displaying the materials. Procedure: Follow safety precautions (put on safety gear, tie hair back if necessary) Clean all materials if necessary and dry them thoroughly. Set up the buret on a buret stand. Label 2 beakers (A for acid and B for base). Pour Hydrochloric Acid into beaker A and Sodium Hydroxideinto
ACIDS VS. BASES
Acids vs. Bases - Home. Hi! My name is Chloe, I'm a 10th grade chemistry, student and I've created this website so that you can learn about acids, bases, and alkalis! You'll learn their definitions, properties, how to identify them, some calculations, and so much more.CALCULATIONS
Note: If a page name is blue, click it to visit that page. Now that you understand Strength and Concentration , it's time to learn how to calculate them The CONJUGATE ACIDS & BASES The definitions stated that acids are proton (H+) donors, and bases are proton (H+) acceptors. After a base receives a proton, the remainder becomes a conjugate acid, and after an acid donates a proton, the remainder becomes a conjugate base. Lets make things simple. Think of a proton as a ball being passed between two people like in the gif below. PROPERTIES, REACTIONS, AND DEFINITONS Properties of Bases & Alkalis:. pH over 7 (cold colors on pH strip); Bitter; Corrosive; Electrolyte; Turns red litmus paper blue; Produces OH- ions when they dissolve in water (if it does not dissolve, it's a base, if it does, its an alkali; Neutralizes acids to form salt and water; Slippery; Neutralization Reactions:TITRATION LAB
To the right is a slideshow displaying the materials. Procedure: Follow safety precautions (put on safety gear, tie hair back if necessary) Clean all materials if necessary and dry them thoroughly. Set up the buret on a buret stand. Label 2 beakers (A for acid and B for base). Pour Hydrochloric Acid into beaker A and Sodium Hydroxideinto
INDICATORS - ACIDS VS. BASES When combined with a base, it turns pink. If it's neutral (pH of 7), it's clear (useful for neutralization reactions). Another common indicator is red litmus paper and blue litmus paper. In a base, litmus paper will turn to or stay blue. In an acid, it will turn to or stay red. (see picture below). Sometimes you'll need to identify ifsomething
EXAMPLES IN EVERYDAY LIFE Examples in Everyday Life - Acids vs. Bases. pH review: 1-3 is a Strong Acid, 4-7 is a Weak Acid, 7 is Neutral, 8-10 is a Weak Alkali, 11-14 is a Strong Alkali. Examples of acids in everyday life: Examples of bases and alkalis in everyday life: Vinegar is diluted acetic acid, which is what gives salad dressings and pickled vegetables their tartINFORMATION
"Acids, Bases, and PH." Essential Biochemistry.N.p., n.d. Web. . VOCABULARY - ACIDS VS. BASES Dissociation: The disconnection or separation of something from something else. Strength: The ability to produce hydrogen/hydroxide ions. Buffer: A solution that can resist pH change upon addition of an acid or base. Concentration: Substance per unit volume. pH: A figure expressing the acidity/alkalinity of ACIDS VS. BASES & ALKALIS Powered by Create your own unique website with customizable templates.Get Started
BIBLIOGRAPHY
Powered by Create your own unique website with customizable templates.Get Started
QUIZ - ACIDS VS. BASES Quiz - Acids vs. Bases. Welcome to the quiz! A calculator and a periodic table are on the page for your convenience. Once you're done with the quiz, thats it! If you want to review some vocabulary before taking the quiz, click here . To visit the bibliography and see where some of this information comes from (other than my teacher of courseACIDS VS. BASES
Acids vs. Bases - Home. Hi! My name is Chloe, I'm a 10th grade chemistry, student and I've created this website so that you can learn about acids, bases, and alkalis! You'll learn their definitions, properties, how to identify them, some calculations, and so much more. CONJUGATE ACIDS & BASES The definitions stated that acids are proton (H+) donors, and bases are proton (H+) acceptors. After a base receives a proton, the remainder becomes a conjugate acid, and after an acid donates a proton, the remainder becomes a conjugate base. Lets make things simple. Think of a proton as a ball being passed between two people like in the gif below.CALCULATIONS
Note: If a page name is blue, click it to visit that page. Now that you understand Strength and Concentration , it's time to learn how to calculate them The PROPERTIES, REACTIONS, AND DEFINITONS Properties of Bases & Alkalis:. pH over 7 (cold colors on pH strip); Bitter; Corrosive; Electrolyte; Turns red litmus paper blue; Produces OH- ions when they dissolve in water (if it does not dissolve, it's a base, if it does, its an alkali; Neutralizes acids to form salt and water; Slippery; Neutralization Reactions: INDICATORS - ACIDS VS. BASES When combined with a base, it turns pink. If it's neutral (pH of 7), it's clear (useful for neutralization reactions). Another common indicator is red litmus paper and blue litmus paper. In a base, litmus paper will turn to or stay blue. In an acid, it will turn to or stay red. (see picture below). Sometimes you'll need to identify ifsomething
EXAMPLES IN EVERYDAY LIFE Examples in Everyday Life - Acids vs. Bases. pH review: 1-3 is a Strong Acid, 4-7 is a Weak Acid, 7 is Neutral, 8-10 is a Weak Alkali, 11-14 is a Strong Alkali. Examples of acids in everyday life: Examples of bases and alkalis in everyday life: Vinegar is diluted acetic acid, which is what gives salad dressings and pickled vegetables their tartTITRATION LAB
To the right is a slideshow displaying the materials. Procedure: Follow safety precautions (put on safety gear, tie hair back if necessary) Clean all materials if necessary and dry them thoroughly. Set up the buret on a buret stand. Label 2 beakers (A for acid and B for base). Pour Hydrochloric Acid into beaker A and Sodium Hydroxideinto
ACIDS VS. BASES
Acids vs. Bases - Home. Hi! My name is Chloe, I'm a 10th grade chemistry, student and I've created this website so that you can learn about acids, bases, and alkalis! You'll learn their definitions, properties, how to identify them, some calculations, and so much more. CONJUGATE ACIDS & BASES The definitions stated that acids are proton (H+) donors, and bases are proton (H+) acceptors. After a base receives a proton, the remainder becomes a conjugate acid, and after an acid donates a proton, the remainder becomes a conjugate base. Lets make things simple. Think of a proton as a ball being passed between two people like in the gif below.CALCULATIONS
Note: If a page name is blue, click it to visit that page. Now that you understand Strength and Concentration , it's time to learn how to calculate them The PROPERTIES, REACTIONS, AND DEFINITONS Properties of Bases & Alkalis:. pH over 7 (cold colors on pH strip); Bitter; Corrosive; Electrolyte; Turns red litmus paper blue; Produces OH- ions when they dissolve in water (if it does not dissolve, it's a base, if it does, its an alkali; Neutralizes acids to form salt and water; Slippery; Neutralization Reactions: INDICATORS - ACIDS VS. BASES When combined with a base, it turns pink. If it's neutral (pH of 7), it's clear (useful for neutralization reactions). Another common indicator is red litmus paper and blue litmus paper. In a base, litmus paper will turn to or stay blue. In an acid, it will turn to or stay red. (see picture below). Sometimes you'll need to identify ifsomething
EXAMPLES IN EVERYDAY LIFE Examples in Everyday Life - Acids vs. Bases. pH review: 1-3 is a Strong Acid, 4-7 is a Weak Acid, 7 is Neutral, 8-10 is a Weak Alkali, 11-14 is a Strong Alkali. Examples of acids in everyday life: Examples of bases and alkalis in everyday life: Vinegar is diluted acetic acid, which is what gives salad dressings and pickled vegetables their tartTITRATION LAB
To the right is a slideshow displaying the materials. Procedure: Follow safety precautions (put on safety gear, tie hair back if necessary) Clean all materials if necessary and dry them thoroughly. Set up the buret on a buret stand. Label 2 beakers (A for acid and B for base). Pour Hydrochloric Acid into beaker A and Sodium Hydroxideinto
CALCULATIONS
Note: If a page name is blue, click it to visit that page. Now that you understand Strength and Concentration , it's time to learn how to calculate them TheINFORMATION
"Acids, Bases, and PH." Essential Biochemistry.N.p., n.d. Web. . VOCABULARY - ACIDS VS. BASES Dissociation: The disconnection or separation of something from something else. Strength: The ability to produce hydrogen/hydroxide ions. Buffer: A solution that can resist pH change upon addition of an acid or base. Concentration: Substance per unit volume. pH: A figure expressing the acidity/alkalinity of ACIDS VS. BASES & ALKALIS Powered by Create your own unique website with customizable templates.Get Started
QUIZ - ACIDS VS. BASES Quiz - Acids vs. Bases. Welcome to the quiz! A calculator and a periodic table are on the page for your convenience. Once you're done with the quiz, thats it! If you want to review some vocabulary before taking the quiz, click here . To visit the bibliography and see where some of this information comes from (other than my teacher of courseACIDS VS. BASES
Hi! My name is Chloe, I'm a 10th grade chemistry, student and I've created this website so that you can learn about acids, bases, and alkalis! You'll learn their definitions, properties, how to CONJUGATE ACIDS & BASES Lets make things simple. Think of a proton as a ball being passed between two people like in the gif below. The person wearing red is an acid, and the person in blue is a base (just like on a pH scale).CALCULATIONS
Note: If a page name is blue, click it to visit that page. Now that you understand Strength and Concentration , it's time to learn how to calculate them The PROPERTIES, REACTIONS, AND DEFINITONS Properties of Bases & Alkalis:. pH over 7 (cold colors on pH strip); Bitter; Corrosive; Electrolyte; Turns red litmus paper blue; Produces OH- ions when they dissolve in water (if it does not dissolve, it's a base, if it does, its an alkali; Neutralizes acids to form salt and water; Slippery; Neutralization Reactions: INDICATORS - ACIDS VS. BASES Other indicators include the Universal Indicator, which is a chemical that changes the color of an acid or base (see picture above). You match this color with one on the chart above, and then you will knowif
TITRATION LAB
So what is titration? Titration is a technique used to find the concentration of a known solution (If you've forgotten what concentration is, visit the Vocabulary Page If the concentration of an acid (like HCl) is unknown, you can use the known concentration of a hydroxide base to complete the experiment by using this neutralization reaction: Acid + Base --> Salt + Water EXAMPLES IN EVERYDAY LIFE pH review: 1-3 is a Strong Acid, 4-7 is a Weak Acid, 7 is Neutral, 8-10 is a Weak Alkali, 11-14 is a Strong Alkali STRENGTH, BUFFERS, AND CONCENTRATION A buffer is a solution that can resist pH change upon addition of an acid It can neutralize small amounts of acids or bases, which keeps the pH level of the solution relatively stable. This is necessary for processes that require a stable pH level, such as blood (which maintains a pH of 7: Neutral).PICTURES/GIFS
Gifs: http://25.media.tumblr.com/tumblr_lyni6f8dXK1r9qd2co1_500.gif ; http://gifrific.com/wp-content/uploads/2012/04/Tina-Fey-giving-herself-high-five.gif SALT AND THE OTHER PRODUCTS OF A NEUTRALIZATION REACTION Salt is an inorganic compound formed during neutralization (its always neutral). The salt families include: Chlorides, bromides, iodides, sulphates, and nitrates. Sulphuric acid would form sulphates, nitric acid would form nitrates, and so on.ACIDS VS. BASES
Hi! My name is Chloe, I'm a 10th grade chemistry, student and I've created this website so that you can learn about acids, bases, and alkalis! You'll learn their definitions, properties, how to CONJUGATE ACIDS & BASES Lets make things simple. Think of a proton as a ball being passed between two people like in the gif below. The person wearing red is an acid, and the person in blue is a base (just like on a pH scale).CALCULATIONS
Note: If a page name is blue, click it to visit that page. Now that you understand Strength and Concentration , it's time to learn how to calculate them The PROPERTIES, REACTIONS, AND DEFINITONS Properties of Bases & Alkalis:. pH over 7 (cold colors on pH strip); Bitter; Corrosive; Electrolyte; Turns red litmus paper blue; Produces OH- ions when they dissolve in water (if it does not dissolve, it's a base, if it does, its an alkali; Neutralizes acids to form salt and water; Slippery; Neutralization Reactions: INDICATORS - ACIDS VS. BASES Other indicators include the Universal Indicator, which is a chemical that changes the color of an acid or base (see picture above). You match this color with one on the chart above, and then you will knowif
TITRATION LAB
So what is titration? Titration is a technique used to find the concentration of a known solution (If you've forgotten what concentration is, visit the Vocabulary Page If the concentration of an acid (like HCl) is unknown, you can use the known concentration of a hydroxide base to complete the experiment by using this neutralization reaction: Acid + Base --> Salt + Water EXAMPLES IN EVERYDAY LIFE pH review: 1-3 is a Strong Acid, 4-7 is a Weak Acid, 7 is Neutral, 8-10 is a Weak Alkali, 11-14 is a Strong Alkali STRENGTH, BUFFERS, AND CONCENTRATION A buffer is a solution that can resist pH change upon addition of an acid It can neutralize small amounts of acids or bases, which keeps the pH level of the solution relatively stable. This is necessary for processes that require a stable pH level, such as blood (which maintains a pH of 7: Neutral).PICTURES/GIFS
Gifs: http://25.media.tumblr.com/tumblr_lyni6f8dXK1r9qd2co1_500.gif ; http://gifrific.com/wp-content/uploads/2012/04/Tina-Fey-giving-herself-high-five.gif SALT AND THE OTHER PRODUCTS OF A NEUTRALIZATION REACTION Salt is an inorganic compound formed during neutralization (its always neutral). The salt families include: Chlorides, bromides, iodides, sulphates, and nitrates. Sulphuric acid would form sulphates, nitric acid would form nitrates, and so on.CALCULATIONS
Note: If a page name is blue, click it to visit that page. Now that you understand Strength and Concentration , it's time to learn how to calculate them TheINFORMATION
"Acids, Bases, and PH." Essential Biochemistry.N.p., n.d. Web. . VOCABULARY - ACIDS VS. BASES These words are all from the Properties, Reactions, and Definitions page. To go back to that page, click here . Corrosive: Can destroy or damage by chemical action Litmus Paper: Indicator used to ACIDS VS. BASES & ALKALIS Powered by Create your own unique website with customizable templates.Get Started
QUIZ - ACIDS VS. BASES Welcome to the quiz! A calculator and a periodic table are on the page for your convenience. Once you're done with the quiz, thats it! If you want to review some vocabulary before taking the quiz, click here. To visit the bibliography and see where some of this information Acids vs. Bases* Home
* Acids Vs. Bases & Alkalis * Properties, Reactions, and Definitons * Strength, Buffers, and Concentration* Indicators
* Examples in Everyday Life * Salt and the Other Products of a Neutralization Reaction * Conjugate Acids & Bases* Calculations
* Titration Lab
* Vocabulary
* Quiz
* Bibliography
* Information
* Pictures/Gifs
Hi! My name is Chloe, I'm a 10th grade chemistry, student and I've created this website so that you can learn about acids, bases, and alkalis! You'll learn their definitions, properties, how to identify them, some calculations, and so much more. Get your notebook ready, and click the button below to begin! CLICK HERE TO BEGIN! Powered by Create your own unique website with customizable templates. Get StartedDetails
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0