Are you over 18 and want to see adult content?
More Annotations
A complete backup of autohaus-inoglu.de
Are you over 18 and want to see adult content?

A complete backup of downblouseloving.com
Are you over 18 and want to see adult content?
A complete backup of drivevauxhall.co.uk
Are you over 18 and want to see adult content?

A complete backup of healinghousefamily.com
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of travestismexico.com
Are you over 18 and want to see adult content?

A complete backup of www.artofzoo.com
Are you over 18 and want to see adult content?
A complete backup of www.topboard.org
Are you over 18 and want to see adult content?
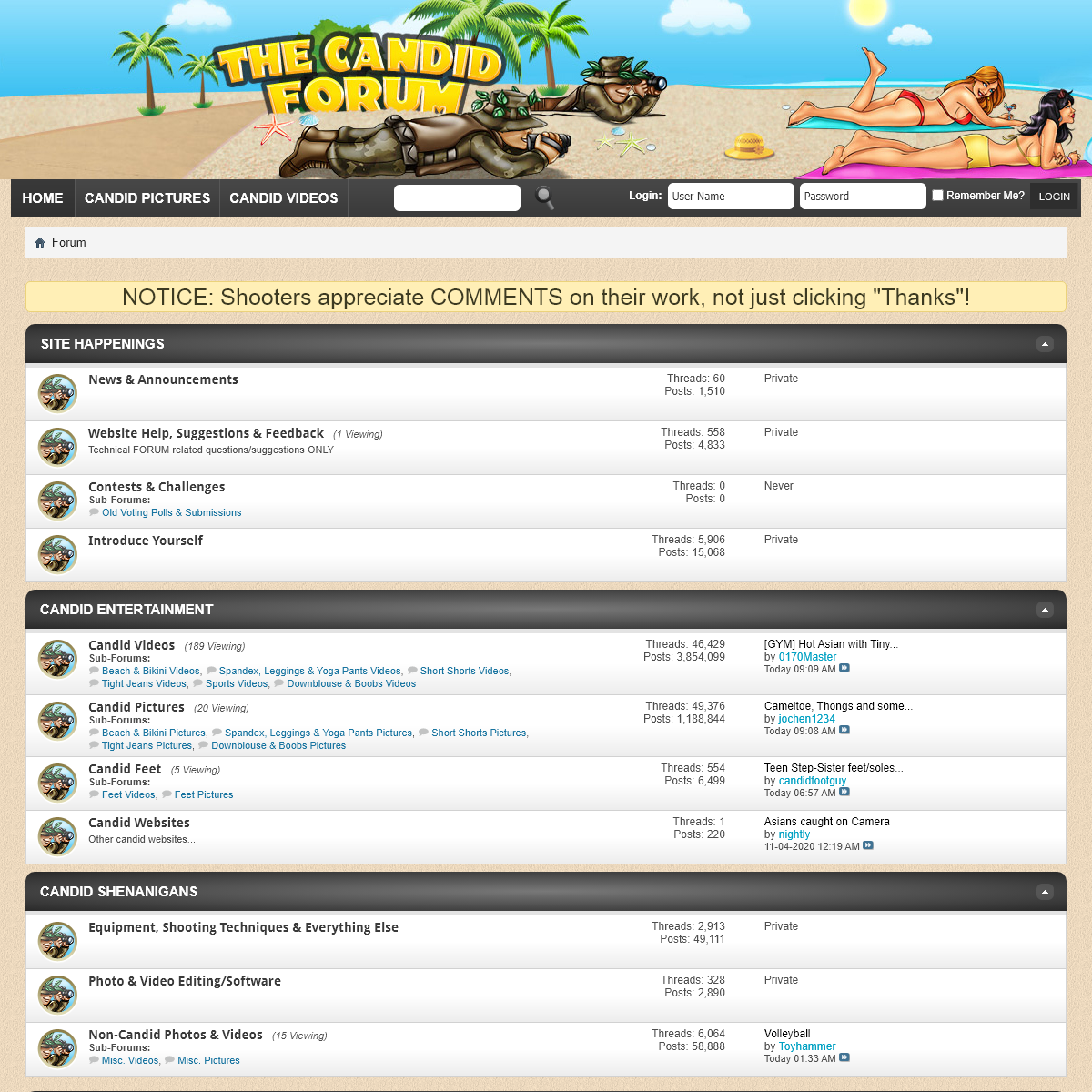
A complete backup of www.thecandidforum.com
Are you over 18 and want to see adult content?
A complete backup of www.www.streamray.com
Are you over 18 and want to see adult content?

A complete backup of www.www.zenra.net
Are you over 18 and want to see adult content?

A complete backup of www.www.nanabook.com
Are you over 18 and want to see adult content?
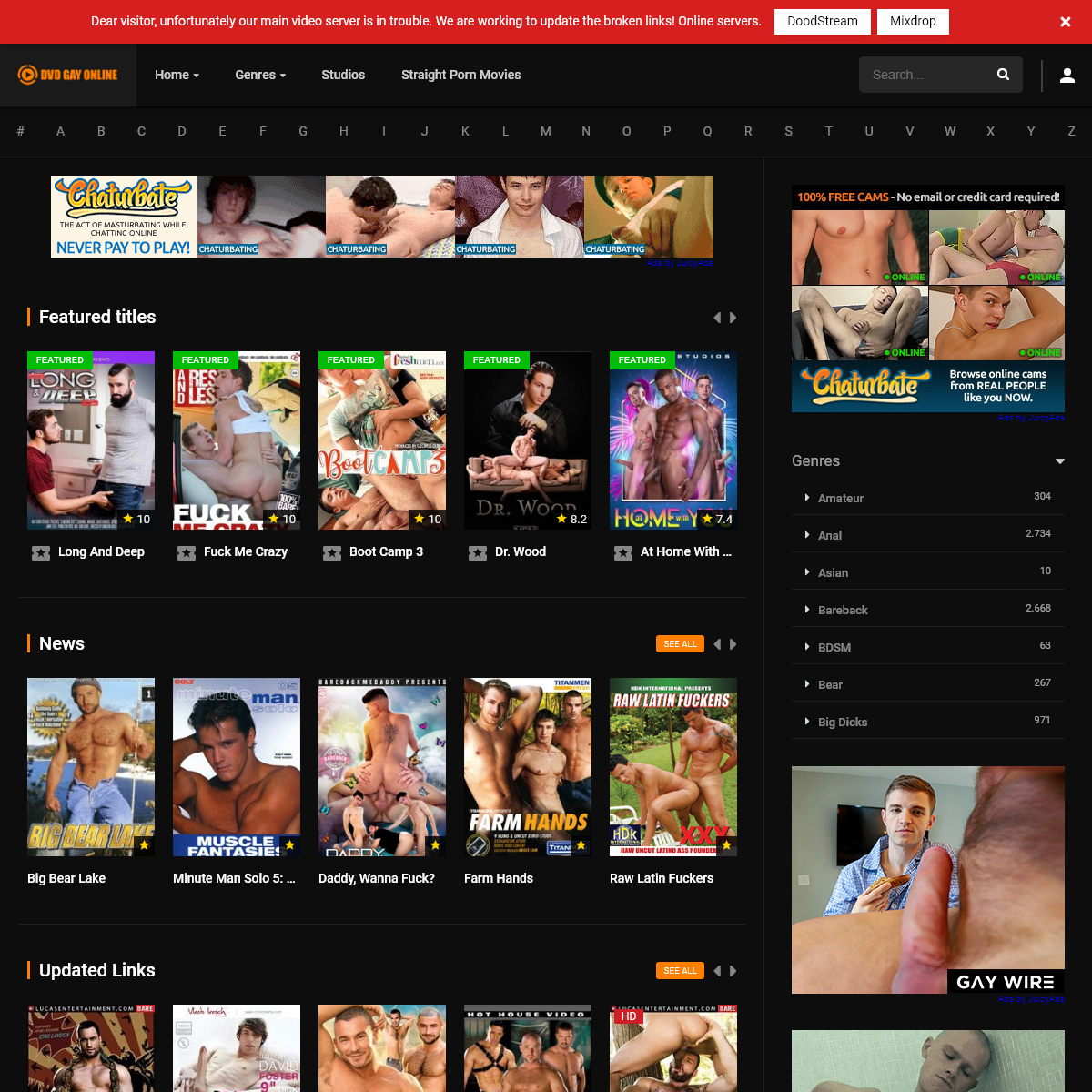
A complete backup of www.dvdgayonline.com
Are you over 18 and want to see adult content?
Text
BLUEPRINT ORPHAN
Introducing RareValue - A decision model for preclinical and clinical stage investments to help unleash the full treatment and commercialization potential in rare and orphan diseases. RareValue is a robust and repeatable analytic tool for the 7,000 rare and orphandiseases
SEARCH - BLUEPRINT ORPHAN Find financial forecast reports and epidemiology assessments on orphan diseases, rare diseases, and ultra-rare diseases. Search by therapy indevelopment.
THERAPEUTICS
Find financial forecast reports and epidemiology assessments on orphan diseases, rare diseases, and ultra-rare diseases. Search by therapy indevelopment.
TEAM - BLUEPRINT ORPHAN BluePrint Orphan is an amalgamation of interdisciplinary professionals who enjoy leveraging the best data and thinking from around the world into actionable business development strategy. We are a one-stop solutions provider, delivering end-to-end services for clients from epidemiology to launch strategy to revenue forecasting. THERAPEUTIC DEVELOPMENT OF ALK-001 ALK-001 is a drug candidate being developed by Alkeus Pharmaceuticals for the treatment of Stargardt Disease (STGD1). Vitamin A dimers are the primary cause of vision loss associated with Stargardt Disease due to retinal toxicity. THERAPEUTIC DEVELOPMENT OF MA09-HRPE CELLS MA09-hRPE cell implantation is a regenerative approach to treating retinal disease being developed by Astellas and Ocata. The primary cause of Stargardt disease is the buildup of toxic vitamin A aggregates due to a genetic defect resulting in damage to retinaltissue.
BLUEPRINT ORPHAN
Introducing RareValue - A decision model for preclinical and clinical stage investments to help unleash the full treatment and commercialization potential in rare and orphan diseases. RareValue is a robust and repeatable analytic tool for the 7,000 rare and orphandiseases
SEARCH - BLUEPRINT ORPHAN Find financial forecast reports and epidemiology assessments on orphan diseases, rare diseases, and ultra-rare diseases. Search by therapy indevelopment.
THERAPEUTICS
Find financial forecast reports and epidemiology assessments on orphan diseases, rare diseases, and ultra-rare diseases. Search by therapy indevelopment.
TEAM - BLUEPRINT ORPHAN BluePrint Orphan is an amalgamation of interdisciplinary professionals who enjoy leveraging the best data and thinking from around the world into actionable business development strategy. We are a one-stop solutions provider, delivering end-to-end services for clients from epidemiology to launch strategy to revenue forecasting. THERAPEUTIC DEVELOPMENT OF ALK-001 ALK-001 is a drug candidate being developed by Alkeus Pharmaceuticals for the treatment of Stargardt Disease (STGD1). Vitamin A dimers are the primary cause of vision loss associated with Stargardt Disease due to retinal toxicity. THERAPEUTIC DEVELOPMENT OF MA09-HRPE CELLS MA09-hRPE cell implantation is a regenerative approach to treating retinal disease being developed by Astellas and Ocata. The primary cause of Stargardt disease is the buildup of toxic vitamin A aggregates due to a genetic defect resulting in damage to retinaltissue.
TEAM - BLUEPRINT ORPHAN BluePrint Orphan is an amalgamation of interdisciplinary professionals who enjoy leveraging the best data and thinking from around the world into actionable business development strategy. We are a one-stop solutions provider, delivering end-to-end services for clients from epidemiology to launch strategy to revenue forecasting. RAREVALUE - BLUEPRINT ORPHAN BluePrint Orphan created a novel decision model for preclinical and clinical stage investments to help unleash the full treatment and commercialization potential in rare and orphan diseases. The objective was to create a robust and repeatable analytic foINDICATIONS
Find epidemiology assessments on orphan diseases, rare diseases, and ultra-rare diseases. Search by indicationWHITE PAPERS
Asset Identification Series - Early Stage Asset Identification (pdf) The cost of drug development averages >$1.3B from laboratory to FDA approval and is increasing.To decrease barriers and reduce pressure on R&D teams, it is becoming increasingly common to outsource technologies through acquisition of smaller companies and licensing agreements of early stage therapeutics.RAREVALUE EXAMPLE
The overall score (11.9) is based on the individual scores from price to treatment. Although we assign discrete scoring, we include statistical ranges to account for (1) where assumptions were made in the absence of exact data and (b) to allow our clients to execute sensitivity analysis as they apply their insights or as more information becomes available. ASSET IDENTIFICATION SERIES success, and 𝑅𝑖 = probability of success at this stage. The result is an estimate that adjusts the expected value down to reflect the probability of success and adjusts the expected costs down toreflect costs
ASSET IDENTIFICATION SERIES evaluate the existing competition and current pricing space. These pieces will be the framework of a more complete evaluation, discussed in the second part of this series. Capabilities reflect the quality of the potential partner.BLUEPRINT ORPHAN
Find financial forecast reports, market size assessments, and global patient information for orphan diseases, rare diseases, and ultra-rarediseases.
PHARMACEUTICAL ACQUISITION AND LICENSING AGREEMENTS Why License? Development of new pharmaceutical and biotech projects can be an extremely costly and time-consuming process. Many new drug candidates and platforms are the result of innovative work in academic institutions or small startups, however these institutions rarely have the funding available to carry a candidate through clinical trials andcommercialization.
CEREBROTENDINOUS XANTHOMATOSIS (CTX): PREVALENCE, INCIDENCE 10-year Global patient forecast for Cerebrotendinous Xanthomatosis (CTX). Estimates are provided by country with sources and methodologyexplanation.
BLUEPRINT ORPHAN
Introducing RareValue - A decision model for preclinical and clinical stage investments to help unleash the full treatment and commercialization potential in rare and orphan diseases. RareValue is a robust and repeatable analytic tool for the 7,000 rare and orphandiseases
TEAM - BLUEPRINT ORPHAN BluePrint Orphan is an amalgamation of interdisciplinary professionals who enjoy leveraging the best data and thinking from around the world into actionable business development strategy. We are a one-stop solutions provider, delivering end-to-end services for clients from epidemiology to launch strategy to revenue forecasting.INDICATIONS
Find epidemiology assessments on orphan diseases, rare diseases, and ultra-rare diseases. Search by indicationLICENSE-ACQUIRE
Partner with BluePrint Orphan: Strategic Advisory Services. As the Orphan industry's preeminent leader in licensing strategy and opportunity identification & assessment, BluePrint Orphan is the optimal corporate partner for streamlining successful deals and meaningfully enhancing ROI and DCF results. Whether exploring genetherapies, vector
RAREVALUE - BLUEPRINT ORPHAN It is an attractive but data- and insights-poor market which may defer or deter investments. BluePrint Orphan’s RareValue model breaks through the fog and uncertainty, giving biopharmaceutical companies the ability to make informed decisions at the individual and portfolio level. RareValue becomes an important accelerant to advancing theWHITE PAPERS
Asset Identification Series - Early Stage Asset Identification (pdf) The cost of drug development averages >$1.3B from laboratory to FDA approval and is increasing.To decrease barriers and reduce pressure on R&D teams, it is becoming increasingly common to outsource technologies through acquisition of smaller companies and licensing agreements of early stage therapeutics. THERAPEUTIC DEVELOPMENT OF MA09-HRPE CELLS Therapeutic Development: MA09-hRPE Cells. MA09-hRPE cell implantation is a regenerative approach to treating retinal disease being developed by Astellas and Ocata. The primary cause of Stargardt disease is the buildup of toxic vitamin A aggregates due to a genetic defect resulting in damage to retinal tissue. MA09-hRPE aims to correct thisvia
THERAPEUTIC DEVELOPMENT OF ALK-001 Therapeutic Development: ALK-001. ALK-001 is a drug candidate being developed by Alkeus Pharmaceuticals for the treatment of Stargardt Disease (STGD1). Vitamin A dimers are the primary cause of vision loss associated with Stargardt Disease due to retinal toxicity. ALK-001 is a modified form of vitamin A that prevents aggregation into dimers. THERAPEUTIC DEVELOPMENT OF SAR 422459 Therapeutic Development: SAR 422459. SAR 422459 is a drug candidate being developed by Sanofi and Oxford BioMedica for the treatment of Stargardt disease (STGD1). The primary cause of Stargardt disease is the buildup of toxic vitamin A aggregates due to a genetic defect. SAR 422459 aims to correct this by using a gene-based approach to insert a PHARMACEUTICAL ACQUISITION AND LICENSING AGREEMENTS Licensing in Action (Deal Structure) In an agreement signed recently between Shire $ SHPG and Parion Sciences, Shire in-licensed rights to a potential treatment for dry eye disease. The treatment, P-321, has been tested in early-stage clinical trials (Phase I/IIa). For the exclusive worldwide rights to develop and commercialize the therapyBLUEPRINT ORPHAN
Introducing RareValue - A decision model for preclinical and clinical stage investments to help unleash the full treatment and commercialization potential in rare and orphan diseases. RareValue is a robust and repeatable analytic tool for the 7,000 rare and orphandiseases
TEAM - BLUEPRINT ORPHAN BluePrint Orphan is an amalgamation of interdisciplinary professionals who enjoy leveraging the best data and thinking from around the world into actionable business development strategy. We are a one-stop solutions provider, delivering end-to-end services for clients from epidemiology to launch strategy to revenue forecasting.INDICATIONS
Find epidemiology assessments on orphan diseases, rare diseases, and ultra-rare diseases. Search by indicationLICENSE-ACQUIRE
Partner with BluePrint Orphan: Strategic Advisory Services. As the Orphan industry's preeminent leader in licensing strategy and opportunity identification & assessment, BluePrint Orphan is the optimal corporate partner for streamlining successful deals and meaningfully enhancing ROI and DCF results. Whether exploring genetherapies, vector
RAREVALUE - BLUEPRINT ORPHAN It is an attractive but data- and insights-poor market which may defer or deter investments. BluePrint Orphan’s RareValue model breaks through the fog and uncertainty, giving biopharmaceutical companies the ability to make informed decisions at the individual and portfolio level. RareValue becomes an important accelerant to advancing theWHITE PAPERS
Asset Identification Series - Early Stage Asset Identification (pdf) The cost of drug development averages >$1.3B from laboratory to FDA approval and is increasing.To decrease barriers and reduce pressure on R&D teams, it is becoming increasingly common to outsource technologies through acquisition of smaller companies and licensing agreements of early stage therapeutics. THERAPEUTIC DEVELOPMENT OF MA09-HRPE CELLS Therapeutic Development: MA09-hRPE Cells. MA09-hRPE cell implantation is a regenerative approach to treating retinal disease being developed by Astellas and Ocata. The primary cause of Stargardt disease is the buildup of toxic vitamin A aggregates due to a genetic defect resulting in damage to retinal tissue. MA09-hRPE aims to correct thisvia
THERAPEUTIC DEVELOPMENT OF ALK-001 Therapeutic Development: ALK-001. ALK-001 is a drug candidate being developed by Alkeus Pharmaceuticals for the treatment of Stargardt Disease (STGD1). Vitamin A dimers are the primary cause of vision loss associated with Stargardt Disease due to retinal toxicity. ALK-001 is a modified form of vitamin A that prevents aggregation into dimers. THERAPEUTIC DEVELOPMENT OF SAR 422459 Therapeutic Development: SAR 422459. SAR 422459 is a drug candidate being developed by Sanofi and Oxford BioMedica for the treatment of Stargardt disease (STGD1). The primary cause of Stargardt disease is the buildup of toxic vitamin A aggregates due to a genetic defect. SAR 422459 aims to correct this by using a gene-based approach to insert a PHARMACEUTICAL ACQUISITION AND LICENSING AGREEMENTS Licensing in Action (Deal Structure) In an agreement signed recently between Shire $ SHPG and Parion Sciences, Shire in-licensed rights to a potential treatment for dry eye disease. The treatment, P-321, has been tested in early-stage clinical trials (Phase I/IIa). For the exclusive worldwide rights to develop and commercialize the therapy PRODUCTS - BLUEPRINT ORPHAN BluePrint Orphan provides accurate patient and financial forecasts for orphan, rare, and ultra-rare diseases. Market assessments may be searched by INDICATION or THERAPY.THERAPEUTICS
Alphabetical List of Therapeutics. 225Ac-lintuzumab (Actimab-A) BAF312 (siponimod) CA4P (fosbretabulin) dabrafenib and trametinib (Tafinlar + Mekinist) E5501 (avatrombopag) farletuzumab (MORAb-003) G17DT immunogen (polyclonal antiboby stimulator) H.P. Acthar Gel (repository corticortropin injection) SEARCH - BLUEPRINT ORPHAN H.P. Acthar Gel (repository corticortropin injection) halofuginone hydrobromide (HT-100) hepatocyte growth factor-X7 (VM202) HSP70 heat shock protein (Minnelide 001) HT-100 (halofuginone hydrobromide) human plasminogen. human retinal progenitor cells (hRPC) hydrocortisone modified release capsules.OUT-LICENSE
BluePrint Orphan maintains active relationships with hundreds of companies with interest in orphan products (preclinical through phase III). As the Orphan industry's preeminent leader in licensing strategy and opportunity identification & assessment, we will likely succeed in locating a partner interested in acquiring or licensing your IP.RAREVALUE EXAMPLE
RareValue Example: IgA Nephropathy. IgA Nephropathy is the most common form of glomerulonephritis and a principal cause of ESRD worldwide with no current treatment. Typical clinical features include hematuria and proteinuria with 30% of patients developing progressive renal failure. The overall score (11.9) is based on the individual scores CEREBROTENDINOUS XANTHOMATOSIS (CTX): PREVALENCE, INCIDENCE Cerebrotendinous Xanthomatosis (CTX) 10-year Global patient forecast for Cerebrotendinous Xanthomatosis (CTX). Estimates are provided by country with sources and methodology explanation. Additional countries can be line-itemed by request. Literature PDFs and other source files will be provided in addition to the forecast. VOLANESORSEN FORECAST & PRICING 10-year Global therapeutic forecast for volanesorsen indicated for Hypolipoproteinemia. (The treatment of familial chylomicronemia syndrome.) Forecasts are provided by ASSET IDENTIFICATION SERIES success, and 𝑅𝑖 = probability of success at this stage. The result is an estimate that adjusts the expected value down to reflect the probability of success and adjusts the expected costs down toreflect costs
ECOPIPAM HYDROCHLORIDE FORECAST & PRICING ecopipam hydrochloride. SKU: THE0159. $0.00. Unavailable. per item. 10-year Global therapeutic forecast for ecopipam hydrochloride indicated for Tourette Syndrome. (The treatment of Tourette's syndrome in children 0-16 years old.) Forecasts are provided by WTX-101 (BIS-CHOLINE TETRATHIOMOLYBDATE) FORECAST & PRICING 10-year Global forecast & Pricing for WTX-101 (bis-choline tetrathiomolybdate), studied for Wilson Disease.BLUEPRINT ORPHAN
Introducing RareValue - A decision model for preclinical and clinical stage investments to help unleash the full treatment and commercialization potential in rare and orphan diseases. RareValue is a robust and repeatable analytic tool for the 7,000 rare and orphandiseases
TEAM - BLUEPRINT ORPHAN BluePrint Orphan is an amalgamation of interdisciplinary professionals who enjoy leveraging the best data and thinking from around the world into actionable business development strategy. We are a one-stop solutions provider, delivering end-to-end services for clients from epidemiology to launch strategy to revenue forecasting.INDICATIONS
Find epidemiology assessments on orphan diseases, rare diseases, and ultra-rare diseases. Search by indicationLICENSE-ACQUIRE
Partner with BluePrint Orphan: Strategic Advisory Services. As the Orphan industry's preeminent leader in licensing strategy and opportunity identification & assessment, BluePrint Orphan is the optimal corporate partner for streamlining successful deals and meaningfully enhancing ROI and DCF results. Whether exploring genetherapies, vector
RAREVALUE - BLUEPRINT ORPHAN It is an attractive but data- and insights-poor market which may defer or deter investments. BluePrint Orphan’s RareValue model breaks through the fog and uncertainty, giving biopharmaceutical companies the ability to make informed decisions at the individual and portfolio level. RareValue becomes an important accelerant to advancing theWHITE PAPERS
Asset Identification Series - Early Stage Asset Identification (pdf) The cost of drug development averages >$1.3B from laboratory to FDA approval and is increasing.To decrease barriers and reduce pressure on R&D teams, it is becoming increasingly common to outsource technologies through acquisition of smaller companies and licensing agreements of early stage therapeutics. THERAPEUTIC DEVELOPMENT OF MA09-HRPE CELLS Therapeutic Development: MA09-hRPE Cells. MA09-hRPE cell implantation is a regenerative approach to treating retinal disease being developed by Astellas and Ocata. The primary cause of Stargardt disease is the buildup of toxic vitamin A aggregates due to a genetic defect resulting in damage to retinal tissue. MA09-hRPE aims to correct thisvia
THERAPEUTIC DEVELOPMENT OF ALK-001 Therapeutic Development: ALK-001. ALK-001 is a drug candidate being developed by Alkeus Pharmaceuticals for the treatment of Stargardt Disease (STGD1). Vitamin A dimers are the primary cause of vision loss associated with Stargardt Disease due to retinal toxicity. ALK-001 is a modified form of vitamin A that prevents aggregation into dimers. THERAPEUTIC DEVELOPMENT OF SAR 422459 Therapeutic Development: SAR 422459. SAR 422459 is a drug candidate being developed by Sanofi and Oxford BioMedica for the treatment of Stargardt disease (STGD1). The primary cause of Stargardt disease is the buildup of toxic vitamin A aggregates due to a genetic defect. SAR 422459 aims to correct this by using a gene-based approach to insert a PHARMACEUTICAL ACQUISITION AND LICENSING AGREEMENTS Licensing in Action (Deal Structure) In an agreement signed recently between Shire $ SHPG and Parion Sciences, Shire in-licensed rights to a potential treatment for dry eye disease. The treatment, P-321, has been tested in early-stage clinical trials (Phase I/IIa). For the exclusive worldwide rights to develop and commercialize the therapyBLUEPRINT ORPHAN
Introducing RareValue - A decision model for preclinical and clinical stage investments to help unleash the full treatment and commercialization potential in rare and orphan diseases. RareValue is a robust and repeatable analytic tool for the 7,000 rare and orphandiseases
TEAM - BLUEPRINT ORPHAN BluePrint Orphan is an amalgamation of interdisciplinary professionals who enjoy leveraging the best data and thinking from around the world into actionable business development strategy. We are a one-stop solutions provider, delivering end-to-end services for clients from epidemiology to launch strategy to revenue forecasting.INDICATIONS
Find epidemiology assessments on orphan diseases, rare diseases, and ultra-rare diseases. Search by indicationLICENSE-ACQUIRE
Partner with BluePrint Orphan: Strategic Advisory Services. As the Orphan industry's preeminent leader in licensing strategy and opportunity identification & assessment, BluePrint Orphan is the optimal corporate partner for streamlining successful deals and meaningfully enhancing ROI and DCF results. Whether exploring genetherapies, vector
RAREVALUE - BLUEPRINT ORPHAN It is an attractive but data- and insights-poor market which may defer or deter investments. BluePrint Orphan’s RareValue model breaks through the fog and uncertainty, giving biopharmaceutical companies the ability to make informed decisions at the individual and portfolio level. RareValue becomes an important accelerant to advancing theWHITE PAPERS
Asset Identification Series - Early Stage Asset Identification (pdf) The cost of drug development averages >$1.3B from laboratory to FDA approval and is increasing.To decrease barriers and reduce pressure on R&D teams, it is becoming increasingly common to outsource technologies through acquisition of smaller companies and licensing agreements of early stage therapeutics. THERAPEUTIC DEVELOPMENT OF MA09-HRPE CELLS Therapeutic Development: MA09-hRPE Cells. MA09-hRPE cell implantation is a regenerative approach to treating retinal disease being developed by Astellas and Ocata. The primary cause of Stargardt disease is the buildup of toxic vitamin A aggregates due to a genetic defect resulting in damage to retinal tissue. MA09-hRPE aims to correct thisvia
THERAPEUTIC DEVELOPMENT OF ALK-001 Therapeutic Development: ALK-001. ALK-001 is a drug candidate being developed by Alkeus Pharmaceuticals for the treatment of Stargardt Disease (STGD1). Vitamin A dimers are the primary cause of vision loss associated with Stargardt Disease due to retinal toxicity. ALK-001 is a modified form of vitamin A that prevents aggregation into dimers. THERAPEUTIC DEVELOPMENT OF SAR 422459 Therapeutic Development: SAR 422459. SAR 422459 is a drug candidate being developed by Sanofi and Oxford BioMedica for the treatment of Stargardt disease (STGD1). The primary cause of Stargardt disease is the buildup of toxic vitamin A aggregates due to a genetic defect. SAR 422459 aims to correct this by using a gene-based approach to insert a PHARMACEUTICAL ACQUISITION AND LICENSING AGREEMENTS Licensing in Action (Deal Structure) In an agreement signed recently between Shire $ SHPG and Parion Sciences, Shire in-licensed rights to a potential treatment for dry eye disease. The treatment, P-321, has been tested in early-stage clinical trials (Phase I/IIa). For the exclusive worldwide rights to develop and commercialize the therapy PRODUCTS - BLUEPRINT ORPHAN BluePrint Orphan provides accurate patient and financial forecasts for orphan, rare, and ultra-rare diseases. Market assessments may be searched by INDICATION or THERAPY.INDICATIONS
Find epidemiology assessments on orphan diseases, rare diseases, and ultra-rare diseases. Search by indicationLICENSE-ACQUIRE
Partner with BluePrint Orphan: Strategic Advisory Services. As the Orphan industry's preeminent leader in licensing strategy and opportunity identification & assessment, BluePrint Orphan is the optimal corporate partner for streamlining successful deals and meaningfully enhancing ROI and DCF results. Whether exploring genetherapies, vector
SEARCH - BLUEPRINT ORPHAN H.P. Acthar Gel (repository corticortropin injection) halofuginone hydrobromide (HT-100) hepatocyte growth factor-X7 (VM202) HSP70 heat shock protein (Minnelide 001) HT-100 (halofuginone hydrobromide) human plasminogen. human retinal progenitor cells (hRPC) hydrocortisone modified release capsules.OUT-LICENSE
BluePrint Orphan maintains active relationships with hundreds of companies with interest in orphan products (preclinical through phase III). As the Orphan industry's preeminent leader in licensing strategy and opportunity identification & assessment, we will likely succeed in locating a partner interested in acquiring or licensing your IP.RAREVALUE EXAMPLE
RareValue Example: IgA Nephropathy. IgA Nephropathy is the most common form of glomerulonephritis and a principal cause of ESRD worldwide with no current treatment. Typical clinical features include hematuria and proteinuria with 30% of patients developing progressive renal failure. The overall score (11.9) is based on the individual scores PHARMACEUTICAL ACQUISITION AND LICENSING AGREEMENTS Licensing in Action (Deal Structure) In an agreement signed recently between Shire $ SHPG and Parion Sciences, Shire in-licensed rights to a potential treatment for dry eye disease. The treatment, P-321, has been tested in early-stage clinical trials (Phase I/IIa). For the exclusive worldwide rights to develop and commercialize the therapy CEREBROTENDINOUS XANTHOMATOSIS (CTX): PREVALENCE, INCIDENCE Cerebrotendinous Xanthomatosis (CTX) 10-year Global patient forecast for Cerebrotendinous Xanthomatosis (CTX). Estimates are provided by country with sources and methodology explanation. Additional countries can be line-itemed by request. Literature PDFs and other source files will be provided in addition to the forecast. VOLANESORSEN FORECAST & PRICING 10-year Global therapeutic forecast for volanesorsen indicated for Hypolipoproteinemia. (The treatment of familial chylomicronemia syndrome.) Forecasts are provided by ASSET IDENTIFICATION SERIES success, and 𝑅𝑖 = probability of success at this stage. The result is an estimate that adjusts the expected value down to reflect the probability of success and adjusts the expected costs down toreflect costs
BLUEPRINT ORPHAN
Introducing RareValue - A decision model for preclinical and clinical stage investments to help unleash the full treatment and commercialization potential in rare and orphan diseases. RareValue is a robust and repeatable analytic tool for the 7,000 rare and orphandiseases
TEAM - BLUEPRINT ORPHAN BluePrint Orphan is an amalgamation of interdisciplinary professionals who enjoy leveraging the best data and thinking from around the world into actionable business development strategy. We are a one-stop solutions provider, delivering end-to-end services for clients from epidemiology to launch strategy to revenue forecasting.INDICATIONS
Find epidemiology assessments on orphan diseases, rare diseases, and ultra-rare diseases. Search by indicationLICENSE-ACQUIRE
Partner with BluePrint Orphan: Strategic Advisory Services. As the Orphan industry's preeminent leader in licensing strategy and opportunity identification & assessment, BluePrint Orphan is the optimal corporate partner for streamlining successful deals and meaningfully enhancing ROI and DCF results. Whether exploring genetherapies, vector
RAREVALUE - BLUEPRINT ORPHAN It is an attractive but data- and insights-poor market which may defer or deter investments. BluePrint Orphan’s RareValue model breaks through the fog and uncertainty, giving biopharmaceutical companies the ability to make informed decisions at the individual and portfolio level. RareValue becomes an important accelerant to advancing theWHITE PAPERS
Asset Identification Series - Early Stage Asset Identification (pdf) The cost of drug development averages >$1.3B from laboratory to FDA approval and is increasing.To decrease barriers and reduce pressure on R&D teams, it is becoming increasingly common to outsource technologies through acquisition of smaller companies and licensing agreements of early stage therapeutics. THERAPEUTIC DEVELOPMENT OF MA09-HRPE CELLS Therapeutic Development: MA09-hRPE Cells. MA09-hRPE cell implantation is a regenerative approach to treating retinal disease being developed by Astellas and Ocata. The primary cause of Stargardt disease is the buildup of toxic vitamin A aggregates due to a genetic defect resulting in damage to retinal tissue. MA09-hRPE aims to correct thisvia
THERAPEUTIC DEVELOPMENT OF ALK-001 Therapeutic Development: ALK-001. ALK-001 is a drug candidate being developed by Alkeus Pharmaceuticals for the treatment of Stargardt Disease (STGD1). Vitamin A dimers are the primary cause of vision loss associated with Stargardt Disease due to retinal toxicity. ALK-001 is a modified form of vitamin A that prevents aggregation into dimers. THERAPEUTIC DEVELOPMENT OF SAR 422459 Therapeutic Development: SAR 422459. SAR 422459 is a drug candidate being developed by Sanofi and Oxford BioMedica for the treatment of Stargardt disease (STGD1). The primary cause of Stargardt disease is the buildup of toxic vitamin A aggregates due to a genetic defect. SAR 422459 aims to correct this by using a gene-based approach to insert a PHARMACEUTICAL ACQUISITION AND LICENSING AGREEMENTS Licensing in Action (Deal Structure) In an agreement signed recently between Shire $ SHPG and Parion Sciences, Shire in-licensed rights to a potential treatment for dry eye disease. The treatment, P-321, has been tested in early-stage clinical trials (Phase I/IIa). For the exclusive worldwide rights to develop and commercialize the therapyBLUEPRINT ORPHAN
Introducing RareValue - A decision model for preclinical and clinical stage investments to help unleash the full treatment and commercialization potential in rare and orphan diseases. RareValue is a robust and repeatable analytic tool for the 7,000 rare and orphandiseases
TEAM - BLUEPRINT ORPHAN BluePrint Orphan is an amalgamation of interdisciplinary professionals who enjoy leveraging the best data and thinking from around the world into actionable business development strategy. We are a one-stop solutions provider, delivering end-to-end services for clients from epidemiology to launch strategy to revenue forecasting.INDICATIONS
Find epidemiology assessments on orphan diseases, rare diseases, and ultra-rare diseases. Search by indicationLICENSE-ACQUIRE
Partner with BluePrint Orphan: Strategic Advisory Services. As the Orphan industry's preeminent leader in licensing strategy and opportunity identification & assessment, BluePrint Orphan is the optimal corporate partner for streamlining successful deals and meaningfully enhancing ROI and DCF results. Whether exploring genetherapies, vector
RAREVALUE - BLUEPRINT ORPHAN It is an attractive but data- and insights-poor market which may defer or deter investments. BluePrint Orphan’s RareValue model breaks through the fog and uncertainty, giving biopharmaceutical companies the ability to make informed decisions at the individual and portfolio level. RareValue becomes an important accelerant to advancing theWHITE PAPERS
Asset Identification Series - Early Stage Asset Identification (pdf) The cost of drug development averages >$1.3B from laboratory to FDA approval and is increasing.To decrease barriers and reduce pressure on R&D teams, it is becoming increasingly common to outsource technologies through acquisition of smaller companies and licensing agreements of early stage therapeutics. THERAPEUTIC DEVELOPMENT OF MA09-HRPE CELLS Therapeutic Development: MA09-hRPE Cells. MA09-hRPE cell implantation is a regenerative approach to treating retinal disease being developed by Astellas and Ocata. The primary cause of Stargardt disease is the buildup of toxic vitamin A aggregates due to a genetic defect resulting in damage to retinal tissue. MA09-hRPE aims to correct thisvia
THERAPEUTIC DEVELOPMENT OF ALK-001 Therapeutic Development: ALK-001. ALK-001 is a drug candidate being developed by Alkeus Pharmaceuticals for the treatment of Stargardt Disease (STGD1). Vitamin A dimers are the primary cause of vision loss associated with Stargardt Disease due to retinal toxicity. ALK-001 is a modified form of vitamin A that prevents aggregation into dimers. THERAPEUTIC DEVELOPMENT OF SAR 422459 Therapeutic Development: SAR 422459. SAR 422459 is a drug candidate being developed by Sanofi and Oxford BioMedica for the treatment of Stargardt disease (STGD1). The primary cause of Stargardt disease is the buildup of toxic vitamin A aggregates due to a genetic defect. SAR 422459 aims to correct this by using a gene-based approach to insert a PHARMACEUTICAL ACQUISITION AND LICENSING AGREEMENTS Licensing in Action (Deal Structure) In an agreement signed recently between Shire $ SHPG and Parion Sciences, Shire in-licensed rights to a potential treatment for dry eye disease. The treatment, P-321, has been tested in early-stage clinical trials (Phase I/IIa). For the exclusive worldwide rights to develop and commercialize the therapy PRODUCTS - BLUEPRINT ORPHAN BluePrint Orphan provides accurate patient and financial forecasts for orphan, rare, and ultra-rare diseases. Market assessments may be searched by INDICATION or THERAPY.INDICATIONS
Find epidemiology assessments on orphan diseases, rare diseases, and ultra-rare diseases. Search by indicationLICENSE-ACQUIRE
Partner with BluePrint Orphan: Strategic Advisory Services. As the Orphan industry's preeminent leader in licensing strategy and opportunity identification & assessment, BluePrint Orphan is the optimal corporate partner for streamlining successful deals and meaningfully enhancing ROI and DCF results. Whether exploring genetherapies, vector
SEARCH - BLUEPRINT ORPHAN H.P. Acthar Gel (repository corticortropin injection) halofuginone hydrobromide (HT-100) hepatocyte growth factor-X7 (VM202) HSP70 heat shock protein (Minnelide 001) HT-100 (halofuginone hydrobromide) human plasminogen. human retinal progenitor cells (hRPC) hydrocortisone modified release capsules.OUT-LICENSE
BluePrint Orphan maintains active relationships with hundreds of companies with interest in orphan products (preclinical through phase III). As the Orphan industry's preeminent leader in licensing strategy and opportunity identification & assessment, we will likely succeed in locating a partner interested in acquiring or licensing your IP.RAREVALUE EXAMPLE
RareValue Example: IgA Nephropathy. IgA Nephropathy is the most common form of glomerulonephritis and a principal cause of ESRD worldwide with no current treatment. Typical clinical features include hematuria and proteinuria with 30% of patients developing progressive renal failure. The overall score (11.9) is based on the individual scores PHARMACEUTICAL ACQUISITION AND LICENSING AGREEMENTS Licensing in Action (Deal Structure) In an agreement signed recently between Shire $ SHPG and Parion Sciences, Shire in-licensed rights to a potential treatment for dry eye disease. The treatment, P-321, has been tested in early-stage clinical trials (Phase I/IIa). For the exclusive worldwide rights to develop and commercialize the therapy CEREBROTENDINOUS XANTHOMATOSIS (CTX): PREVALENCE, INCIDENCE Cerebrotendinous Xanthomatosis (CTX) 10-year Global patient forecast for Cerebrotendinous Xanthomatosis (CTX). Estimates are provided by country with sources and methodology explanation. Additional countries can be line-itemed by request. Literature PDFs and other source files will be provided in addition to the forecast. VOLANESORSEN FORECAST & PRICING 10-year Global therapeutic forecast for volanesorsen indicated for Hypolipoproteinemia. (The treatment of familial chylomicronemia syndrome.) Forecasts are provided by ASSET IDENTIFICATION SERIES success, and 𝑅𝑖 = probability of success at this stage. The result is an estimate that adjusts the expected value down to reflect the probability of success and adjusts the expected costs down toreflect costs
BLUEPRINT ORPHAN
Introducing RareValue - A decision model for preclinical and clinical stage investments to help unleash the full treatment and commercialization potential in rare and orphan diseases. RareValue is a robust and repeatable analytic tool for the 7,000 rare and orphandiseases
TEAM - BLUEPRINT ORPHAN BluePrint Orphan is an amalgamation of interdisciplinary professionals who enjoy leveraging the best data and thinking from around the world into actionable business development strategy. We are a one-stop solutions provider, delivering end-to-end services for clients from epidemiology to launch strategy to revenue forecasting. RAREVALUE - BLUEPRINT ORPHAN It is an attractive but data- and insights-poor market which may defer or deter investments. BluePrint Orphan’s RareValue model breaks through the fog and uncertainty, giving biopharmaceutical companies the ability to make informed decisions at the individual and portfolio level. RareValue becomes an important accelerant to advancing theWHITE PAPERS
Asset Identification Series - Early Stage Asset Identification (pdf) The cost of drug development averages >$1.3B from laboratory to FDA approval and is increasing.To decrease barriers and reduce pressure on R&D teams, it is becoming increasingly common to outsource technologies through acquisition of smaller companies and licensing agreements of early stage therapeutics. THERAPEUTIC DEVELOPMENT OF MA09-HRPE CELLS Therapeutic Development: MA09-hRPE Cells. MA09-hRPE cell implantation is a regenerative approach to treating retinal disease being developed by Astellas and Ocata. The primary cause of Stargardt disease is the buildup of toxic vitamin A aggregates due to a genetic defect resulting in damage to retinal tissue. MA09-hRPE aims to correct thisvia
THERAPEUTIC DEVELOPMENT OF ALK-001 Therapeutic Development: ALK-001. ALK-001 is a drug candidate being developed by Alkeus Pharmaceuticals for the treatment of Stargardt Disease (STGD1). Vitamin A dimers are the primary cause of vision loss associated with Stargardt Disease due to retinal toxicity. ALK-001 is a modified form of vitamin A that prevents aggregation into dimers. THERAPEUTIC DEVELOPMENT OF SAR 422459 Therapeutic Development: SAR 422459. SAR 422459 is a drug candidate being developed by Sanofi and Oxford BioMedica for the treatment of Stargardt disease (STGD1). The primary cause of Stargardt disease is the buildup of toxic vitamin A aggregates due to a genetic defect. SAR 422459 aims to correct this by using a gene-based approach to insert aRAREVALUE EXAMPLE
RareValue Example: IgA Nephropathy. IgA Nephropathy is the most common form of glomerulonephritis and a principal cause of ESRD worldwide with no current treatment. Typical clinical features include hematuria and proteinuria with 30% of patients developing progressive renal failure. The overall score (11.9) is based on the individual scoresDEVELOPMENT BLOG
Calculated after “two quarter lag”: Q1 sales transactions è Q2 AMP, Best Price, 340B Price Calculated è Q3 340B price becomes effective based on Q1 Sales Orphan Exemption For rural referral centers, sole community hospitals, critical access hospitals, and free-standing cancer hospitals participating in the 340B Program, the term "covered outpatient drug" does not include a drug PHARMACEUTICAL ACQUISITION AND LICENSING AGREEMENTS Licensing in Action (Deal Structure) In an agreement signed recently between Shire $ SHPG and Parion Sciences, Shire in-licensed rights to a potential treatment for dry eye disease. The treatment, P-321, has been tested in early-stage clinical trials (Phase I/IIa). For the exclusive worldwide rights to develop and commercialize the therapyBLUEPRINT ORPHAN
Introducing RareValue - A decision model for preclinical and clinical stage investments to help unleash the full treatment and commercialization potential in rare and orphan diseases. RareValue is a robust and repeatable analytic tool for the 7,000 rare and orphandiseases
TEAM - BLUEPRINT ORPHAN BluePrint Orphan is an amalgamation of interdisciplinary professionals who enjoy leveraging the best data and thinking from around the world into actionable business development strategy. We are a one-stop solutions provider, delivering end-to-end services for clients from epidemiology to launch strategy to revenue forecasting. RAREVALUE - BLUEPRINT ORPHAN It is an attractive but data- and insights-poor market which may defer or deter investments. BluePrint Orphan’s RareValue model breaks through the fog and uncertainty, giving biopharmaceutical companies the ability to make informed decisions at the individual and portfolio level. RareValue becomes an important accelerant to advancing theWHITE PAPERS
Asset Identification Series - Early Stage Asset Identification (pdf) The cost of drug development averages >$1.3B from laboratory to FDA approval and is increasing.To decrease barriers and reduce pressure on R&D teams, it is becoming increasingly common to outsource technologies through acquisition of smaller companies and licensing agreements of early stage therapeutics. THERAPEUTIC DEVELOPMENT OF MA09-HRPE CELLS Therapeutic Development: MA09-hRPE Cells. MA09-hRPE cell implantation is a regenerative approach to treating retinal disease being developed by Astellas and Ocata. The primary cause of Stargardt disease is the buildup of toxic vitamin A aggregates due to a genetic defect resulting in damage to retinal tissue. MA09-hRPE aims to correct thisvia
THERAPEUTIC DEVELOPMENT OF ALK-001 Therapeutic Development: ALK-001. ALK-001 is a drug candidate being developed by Alkeus Pharmaceuticals for the treatment of Stargardt Disease (STGD1). Vitamin A dimers are the primary cause of vision loss associated with Stargardt Disease due to retinal toxicity. ALK-001 is a modified form of vitamin A that prevents aggregation into dimers. THERAPEUTIC DEVELOPMENT OF SAR 422459 Therapeutic Development: SAR 422459. SAR 422459 is a drug candidate being developed by Sanofi and Oxford BioMedica for the treatment of Stargardt disease (STGD1). The primary cause of Stargardt disease is the buildup of toxic vitamin A aggregates due to a genetic defect. SAR 422459 aims to correct this by using a gene-based approach to insert aRAREVALUE EXAMPLE
RareValue Example: IgA Nephropathy. IgA Nephropathy is the most common form of glomerulonephritis and a principal cause of ESRD worldwide with no current treatment. Typical clinical features include hematuria and proteinuria with 30% of patients developing progressive renal failure. The overall score (11.9) is based on the individual scoresDEVELOPMENT BLOG
Calculated after “two quarter lag”: Q1 sales transactions è Q2 AMP, Best Price, 340B Price Calculated è Q3 340B price becomes effective based on Q1 Sales Orphan Exemption For rural referral centers, sole community hospitals, critical access hospitals, and free-standing cancer hospitals participating in the 340B Program, the term "covered outpatient drug" does not include a drug PHARMACEUTICAL ACQUISITION AND LICENSING AGREEMENTS Licensing in Action (Deal Structure) In an agreement signed recently between Shire $ SHPG and Parion Sciences, Shire in-licensed rights to a potential treatment for dry eye disease. The treatment, P-321, has been tested in early-stage clinical trials (Phase I/IIa). For the exclusive worldwide rights to develop and commercialize the therapyTHERAPEUTICS
Alphabetical List of Therapeutics. 225Ac-lintuzumab (Actimab-A) BAF312 (siponimod) CA4P (fosbretabulin) dabrafenib and trametinib (Tafinlar + Mekinist) E5501 (avatrombopag) farletuzumab (MORAb-003) G17DT immunogen (polyclonal antiboby stimulator) H.P. Acthar Gel (repository corticortropin injection)LICENSE-ACQUIRE
Partner with BluePrint Orphan: Strategic Advisory Services. As the Orphan industry's preeminent leader in licensing strategy and opportunity identification & assessment, BluePrint Orphan is the optimal corporate partner for streamlining successful deals and meaningfully enhancing ROI and DCF results. Whether exploring genetherapies, vector
BLUEPRINT ORPHAN
Calculated after “two quarter lag”: Q1 sales transactions è Q2 AMP, Best Price, 340B Price Calculated è Q3 340B price becomes effective based on Q1 Sales Orphan Exemption For rural referral centers, sole community hospitals, critical access hospitals, and free-standing cancer hospitals participating in the 340B Program, the term "covered outpatient drug" does not include a drug SEARCH - BLUEPRINT ORPHAN H.P. Acthar Gel (repository corticortropin injection) halofuginone hydrobromide (HT-100) hepatocyte growth factor-X7 (VM202) HSP70 heat shock protein (Minnelide 001) HT-100 (halofuginone hydrobromide) human plasminogen. human retinal progenitor cells (hRPC) hydrocortisone modified release capsules.OUT-LICENSE
BluePrint Orphan maintains active relationships with hundreds of companies with interest in orphan products (preclinical through phase III). As the Orphan industry's preeminent leader in licensing strategy and opportunity identification & assessment, we will likely succeed in locating a partner interested in acquiring or licensing your IP. PHARMACEUTICAL ACQUISITION AND LICENSING AGREEMENTS Licensing in Action (Deal Structure) In an agreement signed recently between Shire $ SHPG and Parion Sciences, Shire in-licensed rights to a potential treatment for dry eye disease. The treatment, P-321, has been tested in early-stage clinical trials (Phase I/IIa). For the exclusive worldwide rights to develop and commercialize the therapy PLITIDEPSIN FORECAST & PRICING 10-year Global therapeutic forecast for plitidepsin indicated for Multiple Myeloma. (The treatment of multiple myeloma.) Forecasts are provided by country with sources/methodology explanation. CEREBROTENDINOUS XANTHOMATOSIS (CTX): PREVALENCE, INCIDENCE Cerebrotendinous Xanthomatosis (CTX) 10-year Global patient forecast for Cerebrotendinous Xanthomatosis (CTX). Estimates are provided by country with sources and methodology explanation. Additional countries can be line-itemed by request. Literature PDFs and other source files will be provided in addition to the forecast. MIGALASTAT (AT1001) FORECAST & PRICING migalastat (AT1001) SKU: THE0278. $0.00. Unavailable. per item. 10-year Global therapeutic forecast for migalastat (AT1001) indicated for Fabry Disease. (The treatment of Fabry disease.) Forecasts are provided by country with sources/methodology explanation. Additionalcountries can be
LIMB GIRDLE MUSCULAR DYSTROPHY: PREVALENCE, INCIDENCE 10-year Global patient forecast for Limb Girdle Muscular Dystrophy. Estimates are provided by country with sources and methodologyexplanation.
BLUEPRINT ORPHAN
Introducing RareValue - A decision model for preclinical and clinical stage investments to help unleash the full treatment and commercialization potential in rare and orphan diseases. RareValue is a robust and repeatable analytic tool for the 7,000 rare and orphandiseases
TEAM - BLUEPRINT ORPHAN BluePrint Orphan is an amalgamation of interdisciplinary professionals who enjoy leveraging the best data and thinking from around the world into actionable business development strategy. We are a one-stop solutions provider, delivering end-to-end services for clients from epidemiology to launch strategy to revenue forecasting.WHITE PAPERS
Asset Identification Series - Early Stage Asset Identification (pdf) The cost of drug development averages >$1.3B from laboratory to FDA approval and is increasing.To decrease barriers and reduce pressure on R&D teams, it is becoming increasingly common to outsource technologies through acquisition of smaller companies and licensing agreements of early stage therapeutics. RAREVALUE - BLUEPRINT ORPHAN BluePrint Orphan created a novel decision model for preclinical and clinical stage investments to help unleash the full treatment and commercialization potential in rare and orphan diseases. The objective was to create a robust and repeatable analytic fo THERAPEUTIC DEVELOPMENT OF ALK-001 ALK-001 is a drug candidate being developed by Alkeus Pharmaceuticals for the treatment of Stargardt Disease (STGD1). Vitamin A dimers are the primary cause of vision loss associated with Stargardt Disease due to retinal toxicity. THERAPEUTIC DEVELOPMENT OF MA09-HRPE CELLS MA09-hRPE cell implantation is a regenerative approach to treating retinal disease being developed by Astellas and Ocata. The primary cause of Stargardt disease is the buildup of toxic vitamin A aggregates due to a genetic defect resulting in damage to retinaltissue.
THERAPEUTIC DEVELOPMENT OF SAR 422459 SAR 422459 is a drug candidate being developed by Sanofi and Oxford BioMedica for the treatment of Stargardt disease (STGD1). The primary cause of Stargardt disease is the buildup of toxic vitamin A aggregates due to a genetic defect.RAREVALUE EXAMPLE
The overall score (11.9) is based on the individual scores from price to treatment. Although we assign discrete scoring, we include statistical ranges to account for (1) where assumptions were made in the absence of exact data and (b) to allow our clients to execute sensitivity analysis as they apply their insights or as more information becomes available.DEVELOPMENT BLOG
Calculated after “two quarter lag”: Q1 sales transactions è Q2 AMP, Best Price, 340B Price Calculated è Q3 340B price becomes effective based on Q1 Sales Orphan Exemption For rural referral centers, sole community hospitals, critical access hospitals, and free-standing cancer hospitals participating in the 340B Program, the term "covered outpatient drug" does not include a drug PHARMACEUTICAL ACQUISITION AND LICENSING AGREEMENTS Why License? Development of new pharmaceutical and biotech projects can be an extremely costly and time-consuming process. Many new drug candidates and platforms are the result of innovative work in academic institutions or small startups, however these institutions rarely have the funding available to carry a candidate through clinical trials andcommercialization.
BLUEPRINT ORPHAN
Introducing RareValue - A decision model for preclinical and clinical stage investments to help unleash the full treatment and commercialization potential in rare and orphan diseases. RareValue is a robust and repeatable analytic tool for the 7,000 rare and orphandiseases
TEAM - BLUEPRINT ORPHAN BluePrint Orphan is an amalgamation of interdisciplinary professionals who enjoy leveraging the best data and thinking from around the world into actionable business development strategy. We are a one-stop solutions provider, delivering end-to-end services for clients from epidemiology to launch strategy to revenue forecasting.WHITE PAPERS
Asset Identification Series - Early Stage Asset Identification (pdf) The cost of drug development averages >$1.3B from laboratory to FDA approval and is increasing.To decrease barriers and reduce pressure on R&D teams, it is becoming increasingly common to outsource technologies through acquisition of smaller companies and licensing agreements of early stage therapeutics. RAREVALUE - BLUEPRINT ORPHAN BluePrint Orphan created a novel decision model for preclinical and clinical stage investments to help unleash the full treatment and commercialization potential in rare and orphan diseases. The objective was to create a robust and repeatable analytic fo THERAPEUTIC DEVELOPMENT OF ALK-001 ALK-001 is a drug candidate being developed by Alkeus Pharmaceuticals for the treatment of Stargardt Disease (STGD1). Vitamin A dimers are the primary cause of vision loss associated with Stargardt Disease due to retinal toxicity. THERAPEUTIC DEVELOPMENT OF MA09-HRPE CELLS MA09-hRPE cell implantation is a regenerative approach to treating retinal disease being developed by Astellas and Ocata. The primary cause of Stargardt disease is the buildup of toxic vitamin A aggregates due to a genetic defect resulting in damage to retinaltissue.
THERAPEUTIC DEVELOPMENT OF SAR 422459 SAR 422459 is a drug candidate being developed by Sanofi and Oxford BioMedica for the treatment of Stargardt disease (STGD1). The primary cause of Stargardt disease is the buildup of toxic vitamin A aggregates due to a genetic defect.RAREVALUE EXAMPLE
The overall score (11.9) is based on the individual scores from price to treatment. Although we assign discrete scoring, we include statistical ranges to account for (1) where assumptions were made in the absence of exact data and (b) to allow our clients to execute sensitivity analysis as they apply their insights or as more information becomes available.DEVELOPMENT BLOG
Calculated after “two quarter lag”: Q1 sales transactions è Q2 AMP, Best Price, 340B Price Calculated è Q3 340B price becomes effective based on Q1 Sales Orphan Exemption For rural referral centers, sole community hospitals, critical access hospitals, and free-standing cancer hospitals participating in the 340B Program, the term "covered outpatient drug" does not include a drug PHARMACEUTICAL ACQUISITION AND LICENSING AGREEMENTS Why License? Development of new pharmaceutical and biotech projects can be an extremely costly and time-consuming process. Many new drug candidates and platforms are the result of innovative work in academic institutions or small startups, however these institutions rarely have the funding available to carry a candidate through clinical trials andcommercialization.
SERVICES - BLUEPRINT ORPHAN BluePrint Orphan provides financial and patient forecasts utilizing epidemiology, competition tracking, pricing & availability, and treatment & population data.THERAPEUTICS
Find financial forecast reports and epidemiology assessments on orphan diseases, rare diseases, and ultra-rare diseases. Search by therapy indevelopment.
LICENSE-ACQUIRE
Deal sourcing and optimization. As the Orphan industry's preeminent leader in licensing strategy and opportunity identification & assessment, BluePrint Orphan is the optimal corporate partner for streamlining successful deals and meaningfully enhancing ROI and DCFresults.
SEARCH - BLUEPRINT ORPHAN Find financial forecast reports and epidemiology assessments on orphan diseases, rare diseases, and ultra-rare diseases. Search by therapy indevelopment.
OUT-LICENSE
BluePrint Orphan maintains active relationships with hundreds of companies with interest in orphan products (preclinical through phase III). As the Orphan industry's preeminent leader in licensing strategy and opportunity identification & assessment, we will likely succeed in locating a partner interested in acquiring or licensing your IP. PHARMACEUTICAL ACQUISITION AND LICENSING AGREEMENTS Why License? Development of new pharmaceutical and biotech projects can be an extremely costly and time-consuming process. Many new drug candidates and platforms are the result of innovative work in academic institutions or small startups, however these institutions rarely have the funding available to carry a candidate through clinical trials andcommercialization.
PLITIDEPSIN FORECAST & PRICING 10-year Global therapeutic forecast for plitidepsin indicated for Multiple Myeloma. (The treatment of multiple myeloma.) Forecasts are provided by country with sources/methodology explanation. MIGALASTAT (AT1001) FORECAST & PRICING 10-year Global therapeutic forecast for migalastat (AT1001) indicated for Fabry Disease. (The treatment of Fabry disease.) Forecasts are provided by country with sources/methodology explanation. CEREBROTENDINOUS XANTHOMATOSIS (CTX): PREVALENCE, INCIDENCE 10-year Global patient forecast for Cerebrotendinous Xanthomatosis (CTX). Estimates are provided by country with sources and methodologyexplanation.
LIMB GIRDLE MUSCULAR DYSTROPHY: PREVALENCE, INCIDENCE 10-year Global patient forecast for Limb Girdle Muscular Dystrophy. Estimates are provided by country with sources and methodologyexplanation.
* About
* Services
* Products
* BUSINESS DEVELOPMENT* more...
* Team
* Development Blog
* White Papers
* Contact Us
* Market Access and Forecasting * Data Management and Analytics * Global Pricing and Reimbursement* HEOR
* Marketing and Messaging Analytics * STAKEHOLDER ADVISORY BOARDS* Indications
* Therapeutics
Proven excellence in orphan drug data analytics and market access strategyCORE CAPABILITIES
MARKET ACCESS AND FORECASTING DATA MANAGEMENT AND ANALYTICS GLOBAL PRICING AND REIMBURSEMENT HEalthcare economics and Outcomes Research MARKETING AND MESSAGING ANALYTICS stakeholder advisory boards ------------------------- RECENT NEWS AND UPDATES -------------------------HOME
TEAM
PRODUCTS
BUSINESS DEVELOPMENTCONTACT
info@blueprintorphan.com* About
* Team
* Development Blog
* White Papers
* Contact Us
* Services
* Market Access and Forecasting * Data Management and Analytics * Global Pricing and Reimbursement* HEOR
* Marketing and Messaging Analytics * STAKEHOLDER ADVISORY BOARDS* Products
* Indications
* Therapeutics
* BUSINESS DEVELOPMENT* Out-License
* On-Site Workshop
*
Empty Cart
Subtotal: $0.00
Checkout
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0