Are you over 18 and want to see adult content?
More Annotations
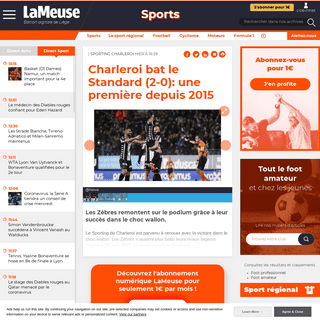
A complete backup of www.thebirdwrites.com/2020/2/25/21152432/new-orleans-pelicans-los-angeles-lakers-gamethread-zion-williamson
Are you over 18 and want to see adult content?

A complete backup of news.mt.co.kr/mtview.php?no=2020030201040022413
Are you over 18 and want to see adult content?
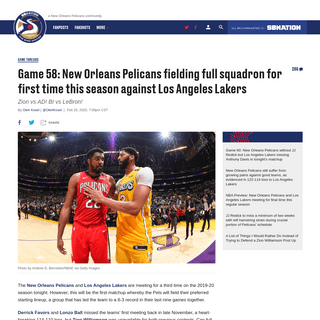
A complete backup of www.bbc.co.uk/news/stories-51633897
Are you over 18 and want to see adult content?

A complete backup of www.hln.be/sport/voetbal/buitenlands-voetbal/premier-league/tottenham-verslikt-zich-op-eigen-veld-en-lijdt-
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of https://baldi.am
Are you over 18 and want to see adult content?

A complete backup of https://f4map.com
Are you over 18 and want to see adult content?
A complete backup of https://antibiopl.com
Are you over 18 and want to see adult content?
A complete backup of https://icaenel.com
Are you over 18 and want to see adult content?

A complete backup of https://thriftymommastips.com
Are you over 18 and want to see adult content?

A complete backup of https://abelgratis.co.uk
Are you over 18 and want to see adult content?

A complete backup of https://kamagrajellynow.com
Are you over 18 and want to see adult content?

A complete backup of https://dreams.co.uk
Are you over 18 and want to see adult content?

A complete backup of https://bikexchange.com
Are you over 18 and want to see adult content?

A complete backup of https://manhattanconstructiongroup.com
Are you over 18 and want to see adult content?
A complete backup of https://biotrack.com
Are you over 18 and want to see adult content?
Text
MANUFACTURING
Red blood cells and platelets are filtered out leaving only the white blood cells behind. This is then put into freezer bags which we call leukopaks. Step 2: Thawed PBMCS. The manufacturing process begins by thawing the frozen cells and selectively activating the T-cells. Step 3: TALEN-Mediated Gene Editing. MAIN PRODUCT CANDIDATES UCART22 is one of Cellectis’ wholly owned, allogeneic, off-the-shelf gene-edited T-cell product candidates designed for the treatment of relapsed and refractory B-cell acute lymphoblastic leukemia (R/R B-ALL). Like CD19, CD22 is a cell surface antigen expressed from thepre-B
PRESS | CELLECTIS
Cellectis is a clinical-stage biopharmaceutical company developing the Next-Generation CAR T-Cells to Cure Cancer. CELLECTIS AND SERVIER EXPAND COLLABORATION ON UCART19 Cellectis and Servier Expand Collaboration on UCART19 Products. February 18, 2020 – New York (N.Y.) – Cellectis (Euronext Growth: ALCLS; Nasdaq: CLLS), a biopharmaceutical company focused on developing immunotherapies based on gene-edited allogeneic CAR T-cells (UCART), and Servier, an international pharmaceutical company, todayannounced
CELLECTIS PROVIDES BUSINESS UPDATE AND REPORTS FINANCIAL First Quarter Financial Results. Cash: As of March 31, 2021, Cellectis, including Calyxt, had $231 million in consolidated cash, cash equivalents, current financial assets and restricted cash of which $211 million are attributable to Cellectis on a stand-alone basis. This compares to $274 million in consolidated cash, cash equivalents, currentTALEN® | CELLECTIS
TALEN® is a registered trademark owned by the Cellectis Group. Talen. Our approach aims to deliver an off-the-shelf product with the following benefits: Broad availability. Enable products to be shipped globally, thereby reducing deployment obstacles and providing accessibility to a broad patients population; Cost-effectiveness. CYTOVIA THERAPEUTICS AND CELLECTIS PARTNER TO DEVELOP CAMBRIDGE, MA and NEW YORK, NY, February 16th, 2021 – Cytovia Therapeutics, Inc., a biopharmaceutical company developing allogeneic “off-the-shelf” gene-edited Natural Killer (NK) and Chimeric Antigen Receptor (CAR)-NK cells derived from induced pluripotent stem cells (iPSCs), and Cellectis (Euronext Growth: ALCLS - Nasdaq: CLLS) a clinical-stage biopharmaceutical company HOME | CELLECTISABOUTRESEARCHPRODUCTSINVESTORSCAREERSAGENDA April 26, 2021 – New York (N.Y.) – Cellectis (Euronext Growth: ALCLS; Nasdaq: CLLS) Dear Shareholders, In the context of the Covid-19 pandemic and in accordance with Ordinance no. 2020-321 of March 25, 2020, the provisions of which were extended until July 31, 2021 by Decree no. 2021-255 of March 9, 2021, this general meeting will be held in closed session, i.e. without the physical PRODUCTS | CELLECTIS Product candidates. Cellectis is pioneering robust first-in-class allogeneic CAR T-cell programs that address multiple unmet cancer needs. These programs were designed with simplicity, availability and cost-effectiveness for patients. Read more. product-candidates. ucart. INVESTORS | CELLECTIS Cellectis is a clinical-stage biopharmaceutical company developing the Next-Generation CAR T-Cells to Cure Cancer.MANUFACTURING
Red blood cells and platelets are filtered out leaving only the white blood cells behind. This is then put into freezer bags which we call leukopaks. Step 2: Thawed PBMCS. The manufacturing process begins by thawing the frozen cells and selectively activating the T-cells. Step 3: TALEN-Mediated Gene Editing. MAIN PRODUCT CANDIDATES UCART22 is one of Cellectis’ wholly owned, allogeneic, off-the-shelf gene-edited T-cell product candidates designed for the treatment of relapsed and refractory B-cell acute lymphoblastic leukemia (R/R B-ALL). Like CD19, CD22 is a cell surface antigen expressed from thepre-B
PRESS | CELLECTIS
Cellectis is a clinical-stage biopharmaceutical company developing the Next-Generation CAR T-Cells to Cure Cancer. CELLECTIS AND SERVIER EXPAND COLLABORATION ON UCART19 Cellectis and Servier Expand Collaboration on UCART19 Products. February 18, 2020 – New York (N.Y.) – Cellectis (Euronext Growth: ALCLS; Nasdaq: CLLS), a biopharmaceutical company focused on developing immunotherapies based on gene-edited allogeneic CAR T-cells (UCART), and Servier, an international pharmaceutical company, todayannounced
CELLECTIS PROVIDES BUSINESS UPDATE AND REPORTS FINANCIAL First Quarter Financial Results. Cash: As of March 31, 2021, Cellectis, including Calyxt, had $231 million in consolidated cash, cash equivalents, current financial assets and restricted cash of which $211 million are attributable to Cellectis on a stand-alone basis. This compares to $274 million in consolidated cash, cash equivalents, currentTALEN® | CELLECTIS
TALEN® is a registered trademark owned by the Cellectis Group. Talen. Our approach aims to deliver an off-the-shelf product with the following benefits: Broad availability. Enable products to be shipped globally, thereby reducing deployment obstacles and providing accessibility to a broad patients population; Cost-effectiveness. CYTOVIA THERAPEUTICS AND CELLECTIS PARTNER TO DEVELOP CAMBRIDGE, MA and NEW YORK, NY, February 16th, 2021 – Cytovia Therapeutics, Inc., a biopharmaceutical company developing allogeneic “off-the-shelf” gene-edited Natural Killer (NK) and Chimeric Antigen Receptor (CAR)-NK cells derived from induced pluripotent stem cells (iPSCs), and Cellectis (Euronext Growth: ALCLS - Nasdaq: CLLS) a clinical-stage biopharmaceutical company PRODUCTS | CELLECTIS Product candidates. Cellectis is pioneering robust first-in-class allogeneic CAR T-cell programs that address multiple unmet cancer needs. These programs were designed with simplicity, availability and cost-effectiveness for patients. Read more. product-candidates. ucart.ABOUT | CELLECTIS
Cellectis is focused on addressing the medical and food needs of an ever-growing global population through the use of proprietary gene-editing technologies. By leveraging the expertise of our management, research and clinical teams, we believe we can make strong advancements in the immuno-oncology and plant sciences fields. CELLECTIS S.A. REPORTS RESULTS FROM ANNUAL SHAREHOLDERS June 2, 2021 – New York (N.Y.) - Cellectis S.A. held its Annual Shareholders’ General Meeting (Euronext Growth: ALCLS – Nasdaq: CLLS) on June 1, 2021 at its head office in Paris, France. At the meeting, during which more than 66% of voting rights were exercised,Resolutions 1
CAREERS | CELLECTIS
Cellectis continues to be an equal opportunity employer, which values a diverse workforce. Cellectis’ hiring process is based solely upon job-related qualifications without regard to race, color, religion, age, sex, gender identity or expression, sexual orientation and disability. Our employees are part of our genes and we’ve edited inAGENDA | CELLECTIS
Dr. André Choulika, Chief Executive Officer, and Simon Harnest, Chief Investment Officer, will present on Thursday June 3rd from 9:00- 9:25AM ET. Replay for this webcast will be available 15 minutes after the conclusion of the live event, and will expire on July 3rd, 2021. The Next Generation CAR & T Cell Therapies will be held from June22-24
GENERAL MEETINGS
Cellectis is a clinical-stage biopharmaceutical company developing the Next-Generation CAR T-Cells to Cure Cancer. CELLECTIS AND SANOFI PARTNER ON ALEMTUZUMAB AS May 11, 2021 – Paris - Cellectis S.A. (NASDAQ: CLLS – Euronext Growth: ALCLS), a clinical-stage biotechnological company employing its core proprietary technologies to develop best-in-class products based on gene-edited allogeneic CAR T-cells (“UCART”) in the field of immuno-oncology, and Sanofi (Euronext: SAN – NYSE: SNY), today entered into a partnership agreement and a supply CORPORATE GOVERNANCE Governance. André Choulika, Ph.D., is one of the founders of Cellectis and served as Chief Executive Officer since the company's inception in 1999. He served as Chairman of the Board of Directors from 2011 to November 2020 and President of Calyxt from August 2010 toJuly 2020.
PARTNERS | CELLECTIS Iovance is a late-stage biotechnology company developing novel T-cell-based cancer immunotherapies. Cellectis and Iovance have a research collaboration and exclusive worldwide license agreement whereby Iovance licenses certain TALEN ® technology from Cellectis in order to develop tumor infiltrating lymphocytes (TIL) that have been genetically edited to create more potent cancer therapeutics. 9TH ANTIBODY INDUSTRIAL SYMPOSIUM (AIS) 9th Antibody Industrial Symposium (AIS) June 22, 2021 ‐ June 25, 2021 9th Antibody Industrial Symposium (AIS) will be held on June 22-25, 2021, virtually HOME | CELLECTISABOUTRESEARCHPRODUCTSINVESTORSCAREERSAGENDA April 26, 2021 – New York (N.Y.) – Cellectis (Euronext Growth: ALCLS; Nasdaq: CLLS) Dear Shareholders, In the context of the Covid-19 pandemic and in accordance with Ordinance no. 2020-321 of March 25, 2020, the provisions of which were extended until July 31, 2021 by Decree no. 2021-255 of March 9, 2021, this general meeting will be held in closed session, i.e. without the physical PRODUCTS | CELLECTIS Cellectis is a clinical-stage biopharmaceutical company developing the Next-Generation CAR T-Cells to Cure Cancer. INVESTORS | CELLECTIS Cellectis is a clinical-stage biopharmaceutical company developing the Next-Generation CAR T-Cells to Cure Cancer. CELLECTIS AND SERVIER EXPAND COLLABORATION ON UCART19 Cellectis is a clinical-stage biopharmaceutical company developing the Next-Generation CAR T-Cells to Cure Cancer.MANUFACTURING
With more than 8 years of experience in allogeneic CAR-T manufacturing, we have utilized our validated TALEN® gene-editing technology and process development capabilities for the scalable manufacture of 5 Universal CAR-T product candidates (UCART). MAIN PRODUCT CANDIDATES UCART19. What is UCART19? UCART19 is an allogeneic CAR T-cell product candidate developed for the treatment of CD19-expressing hematological malignancies, which has been engineered using TALEN ® gene editing technology. This product candidate was developed by Cellectis before entering into a license agreement with Servier, and is initially being developed in relapsed/refractory acutePRESS | CELLECTIS
Cellectis is a clinical-stage biopharmaceutical company developing the Next-Generation CAR T-Cells to Cure Cancer.CAREERS | CELLECTIS
With 20 years of expertise, we are pioneers in the gene-editing field. Utilizing our proprietary gene editing technology, TALEN®, and electroporator innovation, PulseAgile, we have the tools necessary to continue creating first-in-class, off-the-shelf CAR-T therapies for certain types of blood cancers. CORPORATE GOVERNANCE Leopold Bertea, Ph.D., was appointed in May 2020 to the role of Senior Vice President of Technical Operations Europe. He is responsible for ensuring execution across Technical Operations functions, including process development, analytical development, external supply, and the GMP Paris manufacturing facility that support the development and production of Cellectis’ proprietary productTALEN® | CELLECTIS
The real power of gene editing is being able to choose exactly where to edit a gene. Editing genes allows disabling a functional gene, correcting a gene, or replacing or inserting a DNA sequence at a specific chosen location in a genome. HOME | CELLECTISABOUTRESEARCHPRODUCTSINVESTORSCAREERSAGENDA April 26, 2021 – New York (N.Y.) – Cellectis (Euronext Growth: ALCLS; Nasdaq: CLLS) Dear Shareholders, In the context of the Covid-19 pandemic and in accordance with Ordinance no. 2020-321 of March 25, 2020, the provisions of which were extended until July 31, 2021 by Decree no. 2021-255 of March 9, 2021, this general meeting will be held in closed session, i.e. without the physical PRODUCTS | CELLECTIS Cellectis is a clinical-stage biopharmaceutical company developing the Next-Generation CAR T-Cells to Cure Cancer. INVESTORS | CELLECTIS Cellectis is a clinical-stage biopharmaceutical company developing the Next-Generation CAR T-Cells to Cure Cancer. CELLECTIS AND SERVIER EXPAND COLLABORATION ON UCART19 Cellectis is a clinical-stage biopharmaceutical company developing the Next-Generation CAR T-Cells to Cure Cancer.MANUFACTURING
With more than 8 years of experience in allogeneic CAR-T manufacturing, we have utilized our validated TALEN® gene-editing technology and process development capabilities for the scalable manufacture of 5 Universal CAR-T product candidates (UCART). MAIN PRODUCT CANDIDATES UCART19. What is UCART19? UCART19 is an allogeneic CAR T-cell product candidate developed for the treatment of CD19-expressing hematological malignancies, which has been engineered using TALEN ® gene editing technology. This product candidate was developed by Cellectis before entering into a license agreement with Servier, and is initially being developed in relapsed/refractory acutePRESS | CELLECTIS
Cellectis is a clinical-stage biopharmaceutical company developing the Next-Generation CAR T-Cells to Cure Cancer.CAREERS | CELLECTIS
With 20 years of expertise, we are pioneers in the gene-editing field. Utilizing our proprietary gene editing technology, TALEN®, and electroporator innovation, PulseAgile, we have the tools necessary to continue creating first-in-class, off-the-shelf CAR-T therapies for certain types of blood cancers. CORPORATE GOVERNANCE Leopold Bertea, Ph.D., was appointed in May 2020 to the role of Senior Vice President of Technical Operations Europe. He is responsible for ensuring execution across Technical Operations functions, including process development, analytical development, external supply, and the GMP Paris manufacturing facility that support the development and production of Cellectis’ proprietary productTALEN® | CELLECTIS
The real power of gene editing is being able to choose exactly where to edit a gene. Editing genes allows disabling a functional gene, correcting a gene, or replacing or inserting a DNA sequence at a specific chosen location in a genome. CELLECTIS S.A. REPORTS RESULTS FROM ANNUAL SHAREHOLDERS June 2, 2021 – New York (N.Y.) - Cellectis S.A. held its Annual Shareholders’ General Meeting (Euronext Growth: ALCLS – Nasdaq: CLLS) on June 1, 2021 at its head office in Paris, France. At the meeting, during which more than 66% of voting rights were exercised,Resolutions 1
CELLECTIS PROVIDES BUSINESS UPDATE AND REPORTS FINANCIAL May 6, 2021 – New York – Cellectis S.A. (NASDAQ: CLLS – EURONEXT GROWTH: ALCLS) (the “Company”), a clinical-stage biotechnological company employing its core proprietary technologies to develop best-in-class products based on gene-edited allogeneic chimeric antigen receptor (CAR) T-cells in the field of immuno-oncology, today announced its results for the three-month period endingAGENDA | CELLECTIS
The Next Generation CAR & T Cell Therapies will be held from June 22-24, 2021, virtually. Shipra Das, Team Leader Immuno-Oncology, will present on June 22nd from 12:25PM - 12:50PM PST. CELLECTIS APPOINTS BIOPHARMA VETERAN STEVE DOARES, PH.D July 21, 2020 – New York (N.Y.) – Cellectis (Euronext Growth: ALCLS; Nasdaq: CLLS), a clinical-stage biopharmaceutical company focused on developing immunotherapies based on gene-edited allogeneic CAR T-cells (UCART), today announced that Steve Doares, Ph.D., joins Cellectis from Biogen as Senior Vice President of US Manufacturing and Site Head of Raleigh manufacturing plant in CORPORATE GOVERNANCE Leopold Bertea, Ph.D., was appointed in May 2020 to the role of Senior Vice President of Technical Operations Europe. He is responsible for ensuring execution across Technical Operations functions, including process development, analytical development, external supply, and the GMP Paris manufacturing facility that support the development and production of Cellectis’ proprietary productCONTACT | CELLECTIS
Cellectis is a clinical-stage biopharmaceutical company developing the Next-Generation CAR T-Cells to Cure Cancer.GENERAL MEETINGS
Cellectis is a clinical-stage biopharmaceutical company developing the Next-Generation CAR T-Cells to Cure Cancer. CELLECTIS APPOINTS CARRIE BROWNSTEIN, M.D., AS CHIEF Industry veteran joins Cellectis leadership team as UCART product pipeline advances in clinical trials . April 13, 2020 – New York (N.Y.) – Cellectis (Euronext Growth: ALCLS; Nasdaq: CLLS), a clinical-stage biopharmaceutical company focused on developing immunotherapies based on gene-edited allogeneic CAR T-cells (UCART), today announced the appointment of Carrie Brownstein, M.D., 9TH ANTIBODY INDUSTRIAL SYMPOSIUM (AIS) 9th Antibody Industrial Symposium (AIS) June 22, 2021 ‐ June 25, 2021 9th Antibody Industrial Symposium (AIS) will be held on June 22-25, 2021, virtually ISPE CALIFORNIA CHAPTER WEBINAR EVENT ISPE California Chapter Webinar Event June 07, 2021 The ISPE California Chapter Webinar Event will be held on June 8th, virtually._ _
About Research
Products
Investors
Careers
Agenda
_ _ _ _
FR
_ _ _ _
_ _ _ _
COMMITMENT TO A CURE_ _
Cellectis' gene edited CAR T-cells: how does it work?_ _
Cellectis Allogeneic CAR-T Cells_ _
What's our recipe for gene editing? TALEN® FOR BEST-IN-CLASS GENE EDITING Cellectis leverages its leadership in gene editing through its flagship technology, TALEN®, which allows the company to create CAR T therapies to BRING NEW HOPE TO CANCER PATIENTS THROUGH BROADLY AVAILABLE, OFF-THE-SHELF THERAPIES. TALEN® has been successfully used in the clinic to solve key challenges with allogeneic CAR T including protection from Graft vs. Host Disease, mitigation of rejection and enhanced safety via the integration of a suicide switch.Learn more
_
_
_
_
"Off-the-shelf" immunotherapies that can work for the largest number of patients Cellectis gene-edits “Chimeric Antigen Receptor” (CAR) T-cells from healthy donors into “off-the-shelf” immunotherapy product candidates that are designed to work for the largest number of patients. It’s the difference between being an off-the-shelf product and a service in which each treatment has to be custom-made. This same technology, developed by Cellectis, could help in designing therapies for many other diseases.Learn more
Strong intellectual property, global strategic partnerships. 20 years of innovation. 138 patent families. 107 granted patents and 568 applications. Global strategic partnerships with Allogene, Servier, and leading universities all over the world.Learn more
_ _
EDITING LIFE
PRESS
FDA LIFTS CLINICAL HOLD ON MELANI-01 STUDY EVALUATING CELLECTIS’ PRODUCT CANDIDATE UCARTCS1 IN MULTIPLE MYELOMA Published on: November 17, 2020, 18:58 E.S.T. CELLECTIS APPOINTS PHARMA HR LEADER KYUNG NAM-WORTMAN AS EXECUTIVE VICE PRESIDENT, CHIEF HUMAN RESOURCES OFFICER Published on: November 16, 2020, 16:30 E.S.T. CELLECTIS PROVIDES BUSINESS UPDATE AND REPORTS FINANCIAL RESULTS FOR THIRD QUARTER AND FIRST NINE MONTHS 2020 Published on: November 05, 2020, 16:30 E.S.T. AMERICAN SOCIETY OF HEMATOLOGY (ASH) ABSTRACT SHOWS INITIAL ANTI-LEUKEMIC ACTIVITY OF UCART22 IN BALLI-01 PHASE 1 STUDY IN R/RADULT B-ALL
Published on: November 04, 2020, 00:00 E.S.T. CELLECTIS PUBLISHED STREAMLINED MANUFACTURING METHOD TO GENERATE ULTRAPURE ALLOGENEIC CAR T-CELL THERAPIES Published on: June 25, 2020, 16:30 E.S.T. FDA LIFTS CLINICAL HOLD ON MELANI-01 STUDY EVALUATING CELLECTIS’ PRODUCT CANDIDATE UCARTCS1 IN MULTIPLE MYELOMA NOVEMBER 17, 2020 – NEW YORK (N.Y.) – Cellectis (Euronext Growth: ALCLS - Nasdaq: CLLS), a biopharmaceutical company focused on developing immunotherapies based on gene-edited allogeneic CAR T-cells (UCART), today announced that the U.S. Food and Drug Administration (FDA) has lifted the clinical hold on the Phase 1 MELANI-01 trial evaluating the UCARTCS1 product candidate for the treatment of patients with relapsed or refractory multiple myeloma (MM).Read more
All press releases
CELLECTIS APPOINTS PHARMA HR LEADER KYUNG NAM-WORTMAN AS EXECUTIVE VICE PRESIDENT, CHIEF HUMAN RESOURCES OFFICER NOVEMBER 16, 2020 – NEW YORK (N.Y.) – Cellectis (Euronext Growth: ALCLS - Nasdaq: CLLS), a clinical-stage biopharmaceutical company focused on developing immunotherapies based on gene-edited allogeneic CAR T-cells (UCART), announced today the appointment of Kyung Nam-Wortman as Cellectis’ Executive Vice President, Chief Human Resources Officer. Ms. Nam-Wortman, who will be based in Cellectis’ New York office, is also joining the Company’s executive committee.Read more
All press releases
CELLECTIS PROVIDES BUSINESS UPDATE AND REPORTS FINANCIAL RESULTS FOR THIRD QUARTER AND FIRST NINE MONTHS 2020 November 5, 2020 – New York – Cellectis (Euronext Growth: ALCLS - Nasdaq: CLLS), a clinical-stage biopharmaceutical company focused on developing immunotherapies based on allogeneic gene-edited CAR T-cells (UCART), today announced its results for the three-month and nine-month periods ending September 30, 2020.Read more
All press releases
AMERICAN SOCIETY OF HEMATOLOGY (ASH) ABSTRACT SHOWS INITIAL ANTI-LEUKEMIC ACTIVITY OF UCART22 IN BALLI-01 PHASE 1 STUDY IN R/RADULT B-ALL
NOVEMBER 4TH, 2020 – NEW YORK (N.Y.) – Cellectis (Euronext Growth: ALCLS - Nasdaq: CLLS), a clinical-stage biopharmaceutical company focused on developing immunotherapies based on gene-edited allogeneic CAR T-cells (UCART), announced the release of two abstracts at the American Society of Hematology (ASH) Annual Meeting, one oral presentation of initial data for its BALLI-01 clinical trial and one Trials in Progress poster presentation of its AMELI-01 clinical trial. This will be the first publicly released data from Cellectis’ Phase 1 dose-escalation study of UCART22 product candidate in adult patients with Relapsed/Refractory CD22+ B-ALL.Read more
All press releases
CELLECTIS PUBLISHED STREAMLINED MANUFACTURING METHOD TO GENERATE ULTRAPURE ALLOGENEIC CAR T-CELL THERAPIES Cellectis (Euronext Growth: ALCLS; Nasdaq: CLLS), a biopharmaceutical company focused on developing immunotherapies based on gene-edited allogeneic CAR T-cells (UCART), announced the publication of a new research paper released today in _Frontiers in Bioengineering and Biotechnology_.
This article describes an innovative and easy-to-implement procedure which will streamline the manufacturing of allogeneic ‘off-the-shelf’ CAR T-cell therapies.Read more
All press releases
PRESS
FDA LIFTS CLINICAL HOLD ON MELANI-01 STUDY EVALUATING CELLECTIS’ PRODUCT CANDIDATE UCARTCS1 IN MULTIPLE MYELOMA Published on: November 17, 2020, 18:58 E.S.T. FDA LIFTS CLINICAL HOLD ON MELANI-01 STUDY EVALUATING CELLECTIS’ PRODUCT CANDIDATE UCARTCS1 IN MULTIPLE MYELOMA NOVEMBER 17, 2020 – NEW YORK (N.Y.) – Cellectis (Euronext Growth: ALCLS - Nasdaq: CLLS), a biopharmaceutical company focused on developing immunotherapies based on gene-edited allogeneic CAR T-cells (UCART), today announced that the U.S. Food and Drug Administration (FDA) has lifted the clinical hold on the Phase 1 MELANI-01 trial evaluating the UCARTCS1 product candidate for the treatment of patients with relapsed or refractory multiple myeloma (MM).Read more
All press releases
CELLECTIS APPOINTS PHARMA HR LEADER KYUNG NAM-WORTMAN AS EXECUTIVE VICE PRESIDENT, CHIEF HUMAN RESOURCES OFFICER Published on: November 16, 2020, 16:30 E.S.T. CELLECTIS APPOINTS PHARMA HR LEADER KYUNG NAM-WORTMAN AS EXECUTIVE VICE PRESIDENT, CHIEF HUMAN RESOURCES OFFICER NOVEMBER 16, 2020 – NEW YORK (N.Y.) – Cellectis (Euronext Growth: ALCLS - Nasdaq: CLLS), a clinical-stage biopharmaceutical company focused on developing immunotherapies based on gene-edited allogeneic CAR T-cells (UCART), announced today the appointment of Kyung Nam-Wortman as Cellectis’ Executive Vice President, Chief Human Resources Officer. Ms. Nam-Wortman, who will be based in Cellectis’ New York office, is also joining the Company’s executive committee.Read more
All press releases
CELLECTIS PROVIDES BUSINESS UPDATE AND REPORTS FINANCIAL RESULTS FOR THIRD QUARTER AND FIRST NINE MONTHS 2020 Published on: November 05, 2020, 16:30 E.S.T. CELLECTIS PROVIDES BUSINESS UPDATE AND REPORTS FINANCIAL RESULTS FOR THIRD QUARTER AND FIRST NINE MONTHS 2020 November 5, 2020 – New York – Cellectis (Euronext Growth: ALCLS - Nasdaq: CLLS), a clinical-stage biopharmaceutical company focused on developing immunotherapies based on allogeneic gene-edited CAR T-cells (UCART), today announced its results for the three-month and nine-month periods ending September 30, 2020.Read more
All press releases
AMERICAN SOCIETY OF HEMATOLOGY (ASH) ABSTRACT SHOWS INITIAL ANTI-LEUKEMIC ACTIVITY OF UCART22 IN BALLI-01 PHASE 1 STUDY IN R/RADULT B-ALL
Published on: November 04, 2020, 00:00 E.S.T. AMERICAN SOCIETY OF HEMATOLOGY (ASH) ABSTRACT SHOWS INITIAL ANTI-LEUKEMIC ACTIVITY OF UCART22 IN BALLI-01 PHASE 1 STUDY IN R/RADULT B-ALL
NOVEMBER 4TH, 2020 – NEW YORK (N.Y.) – Cellectis (Euronext Growth: ALCLS - Nasdaq: CLLS), a clinical-stage biopharmaceutical company focused on developing immunotherapies based on gene-edited allogeneic CAR T-cells (UCART), announced the release of two abstracts at the American Society of Hematology (ASH) Annual Meeting, one oral presentation of initial data for its BALLI-01 clinical trial and one Trials in Progress poster presentation of its AMELI-01 clinical trial. This will be the first publicly released data from Cellectis’ Phase 1 dose-escalation study of UCART22 product candidate in adult patients with Relapsed/Refractory CD22+ B-ALL.Read more
All press releases
CELLECTIS PUBLISHED STREAMLINED MANUFACTURING METHOD TO GENERATE ULTRAPURE ALLOGENEIC CAR T-CELL THERAPIES Published on: June 25, 2020, 16:30 E.S.T. CELLECTIS PUBLISHED STREAMLINED MANUFACTURING METHOD TO GENERATE ULTRAPURE ALLOGENEIC CAR T-CELL THERAPIES Cellectis (Euronext Growth: ALCLS; Nasdaq: CLLS), a biopharmaceutical company focused on developing immunotherapies based on gene-edited allogeneic CAR T-cells (UCART), announced the publication of a new research paper released today in _Frontiers in Bioengineering and Biotechnology_.
This article describes an innovative and easy-to-implement procedure which will streamline the manufacturing of allogeneic ‘off-the-shelf’ CAR T-cell therapies.Read more
All press releases
Legal notice GlossaryMedia library
Contact
Community guidelines_ _ _ _
_ _
Subsidiary: Calyxt Inc._ _
Loading…
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0