Are you over 18 and want to see adult content?
More Annotations
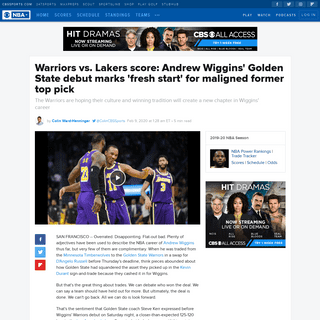
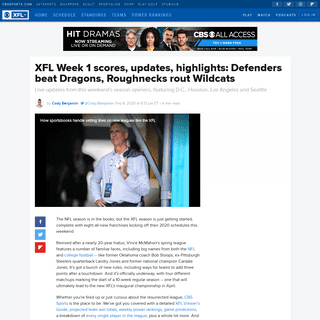
A complete backup of www.cbssports.com/nba/news/warriors-vs-lakers-score-andrew-wiggins-golden-state-debut-marks-fresh-start-for
Are you over 18 and want to see adult content?
Favourite Annotations

MACHINES FOR SMALL BUSINESS
Are you over 18 and want to see adult content?

Palo Alto Counseling, Psychotherapist in Palo Alto and Menlo Park, CA, California - Carol Campbell, MFT
Are you over 18 and want to see adult content?

Think AboutIt – REAL – UFO & Alien Sightings by Date & Location
Are you over 18 and want to see adult content?

LOCKSMITH SEATTLE WASHINGTON - Special Offer - (Second Key - 50% OFF )
Are you over 18 and want to see adult content?
Text
CHEMIDAY.COM
ChemiDay.com - it is a big chemical and biological encyclopedia. It is a large base of chemical reactions, food additives. Moreover, it is very simply in using. You could find correctly what you want. CUCL2 + NA2SO3 + CUCL + H2O = CU2 • H2O + NACL CuCl 2 + 2Na 2 SO 3 + 2CuCl + 2H 2 O → Cu 2 •2H 2 O + 4NaCl. Copper chloride react with sodium sulfite, copper (I) chloride and water to produce dihydrate copper (II) sulfitocuprate (I) and sodium chloride. NAALO2 + CO2 + H2O = AL(OH)3 + NA2CO3 2NaAlO 2 + CO 2 + 3H 2 O → 2Al (OH) 3 + Na 2 CO 3. Sodium aluminate react with carbon dioxide and water to produce aluminium hydroxide and sodium carbonate. The reaction slowly takes place in a boiling solution. CL2 + KOH = KCLO3 + KCL + H2O 3Cl 2 + 6KOH KClO 3 + 5KCl + 3H 2 O. Chlorine react with potassium hydroxide to produce potassium chlorate, potassium chloride and water. Potassium hydroxide - concentrated, heated solution.AS + O2 = AS2O3
4As + 3O 2 2As 2 O 3. Arsenic react with oxygen to produce arsenic (III) oxide. Arsenic combustion on air. This reaction takes place at a temperature of over 350°C. AL + KOH + H2O = K + H2 2Al + 2KOH + 6H 2 O → 2K + 3H 2. Aluminum react with potassium hydroxide and water to produce potassium tetrahydroxoaluminate (III) and hydrogen. Potassium hydroxide - concentrated, heated solution. If playback doesn't begin shortly, try restarting your device. Videos you watch may be added to the TV's watch history and influence NH4VO3 + SO2 + H2SO4 = VOSO4 + (NH4)2SO4 + H2O 2NH 4 VO 3 + SO 2 + 2H 2 SO 4 → 2VOSO 4 + (NH 4) 2 SO 4 + 2H 2 O. Ammonium metavanadate react with sulfur dioxide and sulfuric acid to produce vanadyl sulfate, ammonium sulfate andwater.
FE3O4 + HCL = FECL2 + FECL3 + H2O Fe 3 O 4 + 8HCl → FeCl 2 + 2FeCl 3 + 4H 2 O. Iron (II,III) oxide react with hydrogen chloride to produce iron (II) chloride iron (III) chloride and water. Hydrogen chloride - KMNO4 + H2S = MNO2 + S + KOH + H2O 2KMnO 4 + 3H 2 S → 2MnO 2 + 3S + 2KOH + 2H 2 O. Potassium permanganate react with hydrogen sulfide to produce manganese (IV) oxide, sulfur, potassium hydroxide and water. Hydrogen sulfide - saturated solution. NAH + CL2 = NACL + HCL Sodium hydride react with chlorine to produce sodium chloride and hydrogen chloride. Chemical reaction. Balancing chemical equations.CHEMIDAY.COM
ChemiDay.com - it is a big chemical and biological encyclopedia. It is a large base of chemical reactions, food additives. Moreover, it is very simply in using. You could find correctly what you want. CUCL2 + NA2SO3 + CUCL + H2O = CU2 • H2O + NACL CuCl 2 + 2Na 2 SO 3 + 2CuCl + 2H 2 O → Cu 2 •2H 2 O + 4NaCl. Copper chloride react with sodium sulfite, copper (I) chloride and water to produce dihydrate copper (II) sulfitocuprate (I) and sodium chloride. NAALO2 + CO2 + H2O = AL(OH)3 + NA2CO3 2NaAlO 2 + CO 2 + 3H 2 O → 2Al (OH) 3 + Na 2 CO 3. Sodium aluminate react with carbon dioxide and water to produce aluminium hydroxide and sodium carbonate. The reaction slowly takes place in a boiling solution. CL2 + KOH = KCLO3 + KCL + H2O 3Cl 2 + 6KOH KClO 3 + 5KCl + 3H 2 O. Chlorine react with potassium hydroxide to produce potassium chlorate, potassium chloride and water. Potassium hydroxide - concentrated, heated solution.AS + O2 = AS2O3
4As + 3O 2 2As 2 O 3. Arsenic react with oxygen to produce arsenic (III) oxide. Arsenic combustion on air. This reaction takes place at a temperature of over 350°C. AL + KOH + H2O = K + H2 2Al + 2KOH + 6H 2 O → 2K + 3H 2. Aluminum react with potassium hydroxide and water to produce potassium tetrahydroxoaluminate (III) and hydrogen. Potassium hydroxide - concentrated, heated solution. If playback doesn't begin shortly, try restarting your device. Videos you watch may be added to the TV's watch history and influence NH4VO3 + SO2 + H2SO4 = VOSO4 + (NH4)2SO4 + H2O 2NH 4 VO 3 + SO 2 + 2H 2 SO 4 → 2VOSO 4 + (NH 4) 2 SO 4 + 2H 2 O. Ammonium metavanadate react with sulfur dioxide and sulfuric acid to produce vanadyl sulfate, ammonium sulfate andwater.
FE3O4 + HCL = FECL2 + FECL3 + H2O Fe 3 O 4 + 8HCl → FeCl 2 + 2FeCl 3 + 4H 2 O. Iron (II,III) oxide react with hydrogen chloride to produce iron (II) chloride iron (III) chloride and water. Hydrogen chloride - KMNO4 + H2S = MNO2 + S + KOH + H2O 2KMnO 4 + 3H 2 S → 2MnO 2 + 3S + 2KOH + 2H 2 O. Potassium permanganate react with hydrogen sulfide to produce manganese (IV) oxide, sulfur, potassium hydroxide and water. Hydrogen sulfide - saturated solution. NAH + CL2 = NACL + HCL Sodium hydride react with chlorine to produce sodium chloride and hydrogen chloride. Chemical reaction. Balancing chemical equations.CHEMIDAY.COM
ChemiDay.com - it is a big chemical and biological encyclopedia. It is a large base of chemical reactions, food additives. Moreover, it is very simply in using. You could find correctly what you want. SO2 + KMNO4 + H2O = H2SO4 + MNSO4 + K2SO4 5SO 2 + 2KMnO 4 + 2H 2 O → 2H 2 SO 4 + 2MnSO 4 + K 2 SO 4. Sulfur dioxide react with potassium permanganate and water to produce sulfuric acid, manganese (II) sulfate and potassium sulfate. The reaction takes place in a diluted solution sulfuric acid. K2CR2O7 + H2SO4 + KI = CR2(SO4)3 + I2 + K2SO4 + H2O K 2 Cr 2 O 7 + 7H 2 SO 4 + 6KI → Cr 2 (SO 4) 3 + 3I 2 + 4K 2 SO 4 + 7H 2 O. Potassium dichromate react with sulfuric acid and potassium iodide to produce chromium (III) sulfate, iodine, potassium sulfate and water. Sulfuric acid - diluted solution. If playback doesn't begin shortly, try restarting your device. FECL2 + NA2CO3 = FECO3 + NACL Iron(II) chloride react with sodium carbonate to produce iron(II) carbonate and sodium chloride. Chemical reaction. Balancing chemicalequations.
NA2S2O3 { NA2SO3S } + H2SO4 = NA2SO4 + SO2 + S + H2O Na 2 S 2 O 3 + H 2 SO 4 → Na 2 SO 4 + SO 2 + S↓ + H 2 O. Sodium thiosulfate react with sulfuric acid to produce sodium sulfate, carbon dioxide, sulfur and water. KMNO4 + KOH = K2MNO4 + O2 + H2O 4KMnO 4 + 4KOH → 4K 2 MnO 4 + O 2 + 2H 2 O. Potassium permanganate react with potassium hydroxide to produce potassium manganate, oxygen and water. Potassium permanganate - saturated solution. Potassium hydroxide - 15% solution. The reaction takes place in a boiling solution. NI + HNO3 = NI(NO3)2 + NO + H2O 3Ni + 8HNO 3 → 3Ni (NO 3) 2 + 2NO + 4H 2 O. Nickel react with nitric acid to produce nickel (II) nitrate, nitric oxide and water. Nitric acid - diluted solution. NA2CO3 + H3PO4 = NA3PO4 + H2O + CO2 3Na 2 CO 3 + 2H 3 PO 4 → 2Na 3 PO 4 + 3H 2 O + 3CO 2. Sodium carbonate react with phosphoric acid to produce sodium triphosphate, water and carbon dioxide. Sodium carbonate - concentrated solution. Phosphoric acid - diluted solution. The reaction takes place in ALCL3 + KOH = AL(OH)3 + KCL AlCl 3 + 3KOH → Al (OH) 3 + 3KCl. Aluminum chloride react with potassium hydroxide to produce aluminium hydroxide and potassium chloride. Potassium hydroxide - PB(NO3)2 + NH3•H2O = PB(OH)2 + NH4NO3 Pb (NO 3) 2 + 2 (NH 3 •H 2 O) → Pb (OH) 2 + 2NH 4 NO 3. Lead (II) nitrate react with ammonium hydroxide to produce lead (II) hydroxide and ammonium nitrate. Ammonium hydroxide - concentrated solution.CHEMIDAY.COM
ChemiDay.com - it is a big chemical and biological encyclopedia. It is a large base of chemical reactions, food additives. Moreover, it is very simply in using. You could find correctly what you want. CUCL2 + NA2SO3 + CUCL + H2O = CU2 • H2O + NACL Copper chloride react with sodium sulfite, copper(I) chloride and water to produce dihydrate copper(II) sulfitocuprate(I) and sodium chloride. Chemical reaction. Balancing chemical equations. KMNO4 + HCL = MNCL2 + CL2 + KCL + H2O Potassium permanganate react with hydrogen chloride to produce manganese(II) chloride, chlorine, potassium chloride and water. Chemical reaction. Balancing chemical equations. CR2(SO4)3 + NAOH + H2O2 = NA2CRO4 + H2O + NA2SO4 Chromium(III) sulfate react with sodium hydroxide and hydrogen peroxide to produce sodium chromate, water and sodium sulfate. Chemical reaction. Balancing chemical equations. NA2S2O3 + H2O2 = NA2S3O6 + NA2SO4 + H2O Sodium thiosulfate react with hydrogen peroxide to produce sodium trithionate, sodium sulfate and water. Chemical reaction. Balancingchemical equations.
NANO2 + KMNO4 + H2SO4 = NANO3 + MNSO4 + K2SO4 + H2O Sodium nitrite react with potassium permanganate and sulfuric acid to produce sodium nitrate, manganese(II) sulfate, potassium sulfate andwater.
CACO3 + CH3COOH = CA(CH3COO)2 + H2O + CO2 Calcium carbonate react with acetic acid to produce calcium acetate, water and carbon dioxide. Chemical reaction. Balancing chemicalequations.
SO2 + KMNO4 + H2O = H2SO4 + MNSO4 + K2SO4 Sulfur dioxide react with potassium permanganate and water to produce sulfuric acid, manganese(II) sulfate and potassium sulfate. Chemical reaction. Balancing chemical equations. AL + H2O = AL(OH)3 + H2 Aluminum react with water to produce aluminium hydroxide and hydrogen. Chemical reaction. Balancing chemical equations. FE2O3 + H2 = FE + H2O Iron(III) oxide react with hydrogen to produce iron and water. This reaction takes place at a temperature of 1050-1100°C.CHEMIDAY.COM
ChemiDay.com - it is a big chemical and biological encyclopedia. It is a large base of chemical reactions, food additives. Moreover, it is very simply in using. You could find correctly what you want. CUCL2 + NA2SO3 + CUCL + H2O = CU2 • H2O + NACL Copper chloride react with sodium sulfite, copper(I) chloride and water to produce dihydrate copper(II) sulfitocuprate(I) and sodium chloride. Chemical reaction. Balancing chemical equations. KMNO4 + HCL = MNCL2 + CL2 + KCL + H2O Potassium permanganate react with hydrogen chloride to produce manganese(II) chloride, chlorine, potassium chloride and water. Chemical reaction. Balancing chemical equations. CR2(SO4)3 + NAOH + H2O2 = NA2CRO4 + H2O + NA2SO4 Chromium(III) sulfate react with sodium hydroxide and hydrogen peroxide to produce sodium chromate, water and sodium sulfate. Chemical reaction. Balancing chemical equations. NA2S2O3 + H2O2 = NA2S3O6 + NA2SO4 + H2O Sodium thiosulfate react with hydrogen peroxide to produce sodium trithionate, sodium sulfate and water. Chemical reaction. Balancingchemical equations.
NANO2 + KMNO4 + H2SO4 = NANO3 + MNSO4 + K2SO4 + H2O Sodium nitrite react with potassium permanganate and sulfuric acid to produce sodium nitrate, manganese(II) sulfate, potassium sulfate andwater.
CACO3 + CH3COOH = CA(CH3COO)2 + H2O + CO2 Calcium carbonate react with acetic acid to produce calcium acetate, water and carbon dioxide. Chemical reaction. Balancing chemicalequations.
SO2 + KMNO4 + H2O = H2SO4 + MNSO4 + K2SO4 Sulfur dioxide react with potassium permanganate and water to produce sulfuric acid, manganese(II) sulfate and potassium sulfate. Chemical reaction. Balancing chemical equations. AL + H2O = AL(OH)3 + H2 Aluminum react with water to produce aluminium hydroxide and hydrogen. Chemical reaction. Balancing chemical equations. FE2O3 + H2 = FE + H2O Iron(III) oxide react with hydrogen to produce iron and water. This reaction takes place at a temperature of 1050-1100°C. CHEMICAL REACTIONS ONLINE It is better use the classical form for inorganic compounds Indicate indices like ordinary numbers after the characters. For example H 2 SO4 type as H2SO4.
KMNO4 + C2H5OH = MNO2 + CH3CHO + KOH + H2O Potassium permanganate react with ethanol to produce manganese(IV) oxide, acetaldehyde, potassium hydroxide and water. Chemical reaction. Balancing chemical equations. SO2 + KMNO4 + H2O = H2SO4 + MNSO4 + K2SO4 Sulfur dioxide react with potassium permanganate and water to produce sulfuric acid, manganese(II) sulfate and potassium sulfate. Chemical reaction. Balancing chemical equations. AGNO3 = AG + NO2 + O2 The thermal decomposition of silver nitrate to produce silver, nitrogen dioxide and oxygen. Chemical reaction. Balancing chemicalequations.
AL + NAOH + H2O = NA + H2 Aluminum react with sodium hydroxide and water to produce sodium tetrahydroxoaluminate and hydrogen. Chemical reaction. Balancingchemical equations.
NAAL(SO4)2 + NAHCO3 = CO2 + AL(OH)3 + NA2SO4 Aluminium-sodium sulfate react with sodium bicarbonate to produce carbon dioxide, aluminium hydroxide and sodium sulfate. Chemical reaction. Balancing chemical equations. S + NAOH = NA2S2O3 + NA2S + H2O Sulfur react with sodium hydroxide to produce sodium thiosulfate, sodium sulfide and water. Chemical reaction. Balancing chemicalequations.
CH3COOH + NH3 • H2O = CH3COONH4 + H2O Acetic acid react with ammonium hydroxide to produce ammonium acetate and water. Chemical reaction. Balancing chemical equations. CA(OH)2 + NH4CL = CACL2 + NH3 + H2O Calcium hydroxide react with ammonium chloride to produce calcium chloride, ammonia and water. Chemical reaction. Balancing chemicalequations.
CACO3 + H2CO3 = CA(HCO3)2 Calcium carbonate react with carbonic acid to produce calcium hydrogen carbonate. Chemical reaction. Balancing chemical equations. Главная Энциклопедия Реакции Блоги ВопросыФорум Ещё
Язык: uk ru en
Авторизация:__________
Энциклопедия Химические реакции Таблицы Блоги Вопросы CHEMIDAY.COM - ХИМИЯ НА ШАГ БЛИЖЕНАШ КАНАЛ
------------------------- ПОКАЗАТЬ ВСЕ ОСНОВНЫЕ РАЗДЕЛЫ САЙТА ------------------------- Химические реакции Удобный поиск уравнений химических реакций. Блоги Мысли, опыты, эксперименты и личные наблюдения пользователей сайта. Химические соединения Структурные формулы, химические и физические свойства веществ... Решение задач Задавайте любые вопросы, имеющие отношение к химии. Энциклопедия База статей на различные тематики в энциклопедическом формате. Периодическая система Периодическая система химических элементов Д. И. Менделеева. Пищевые добавки Пищевые добавки в энциклопедическомформате.
Фармакология Фармакология - список элементов в энциклопедическом формате. Лекарства База инструкций и описаний лекарств CHEMIDAY.COM - ЭТО БОЛЬШАЯ ХИМИКО-БИОЛОГИЧЕСКАЯ ЭНЦИКЛОПЕДИЯ, БАЗА УРАВНЕНИЙ ХИМИЧЕСКИХ РЕАКЦИЙ, БАЗА ПИЩЕВЫХ ДОБАВОК И УДОБНЫЙ ПОИСК НЕОБХОДИМОЙ ИНФОРМАЦИИ. МЫ ЕЖЕДНЕВНО УЛУЧШАЕМ НАШИ ИНФОРМАЦИОННЫЕ БАЗЫ И ФУНКЦИОНАЛ САЙТА. ПОСЛЕДНИЕ ПУБЛИКАЦИИ НА САЙТЕ -------------------------Подробно
Подробно
Подробно
СОЛКОСЕРИЛ, мазь - инструкция по применению, описание, дешевые аналоги, побочные эффекты и противопоказания Энциклопедия | Просмотров: 238913.08.2017 ЦИКЛОФЕРОН, таблетки - инструкция по применению, описание, дешевые аналоги, побочные эффекты и противопоказания Энциклопедия | Просмотров: 352913.08.2017 Чем химики моют посуду? Блог | Просмотров: 307519.10.2017 Высокомолекулярные соединения.Часть 2.
Блог | Просмотров: 214014.08.2017ФОРУМ
------------------------- * реактивы советской заводской фотолаборатории (0) * Литература по физхимии (2) * Европий и иттербий (0) * Привет! (1) * Огненная ванна (2) ------------------------- Смотреть всеНОВОСТИ
------------------------- Нейлон. Случайные открытия в химии(0
)
Периодическая система химических элементов Менделеева. Случайные открытия в химии(0
)
------------------------- Смотреть все НОВЫЕ ЛЕКАРСТВА: ------------------------- 19.11.2017 | Био-Макс, таблетки 19.11.2017 | АМЛОДИПИН-ТЕВА, таблетки 19.11.2017 | АЛИМТА, лиофилизат 19.11.2017 | ПУСТЫРНИКА ТРАВА, порошок 19.11.2017 | ФЕРРОГЕМАТОГЕН 19.11.2017 | СПИГЕЛОН, таблетки 19.11.2017 | МЕНТОЛОВОЕ МАСЛО, раствор 19.11.2017 | ИНДИГОКАРМИН, раствор 19.11.2017 | ДЕПРЕНОРМ МВ, таблетки 19.11.2017 | ВИТРУМ БЬЮТИ ЭЛИТ, таблетки 19.11.2017 | НЕЙПОГЕН, раствор 19.11.2017 | ЭФОКС ЛОНГ, таблетки 19.11.2017 | ТОНГИНАЛ, капли 19.11.2017 | СПАЗМОЛ, таблетки 19.11.2017 | РУСТОР, таблетки ВОПРОСЫ ПОЛЬЗОВАТЕЛЕЙ ------------------------- • вычислите массу соли и воды необходимые для приготовления 20 г раствора масовой долей карбоната натрия 5%. • с 250 г раствора натрия карбоната с массовой долей соли в растворе 5% выпарили 120 г воды. определите массовую долю соли в растворе после ча• із 250 г
розчину натрій карбонату з масовою часткою солі в розчині 5% випарували 120 г води . визначте масову частку солі в розчині після ча • вычислите массу воды необходимой для растворения 120 г калий гидроксида для получения раствора с массовой долей вещества 15% • вычислите массу воды необходимой для растворения 120 г калий гидроксида для получения раствора с массовой долей вещества 15% • Запишіть формлу міцели золю ------------------------- Смотреть все Задать вопрос по химии пользователямИгра 2048
Химические элементы Периодическая система Таблица растворимости химических соединений Онлайн калькулятор молярных масс CHEMIDAY не навязывает вам свое мнение. Все что вы делаете - делаете на свой страх и риск. О сайте | Отзывы и предложения | Обратная связь | Страницав ВК
ChemiDay.com © 2019<
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0