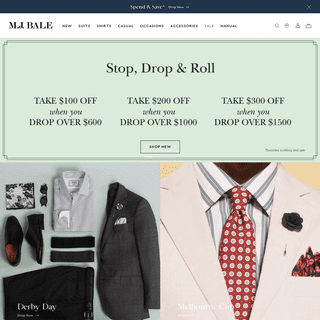
Are you over 18 and want to see adult content?
More Annotations
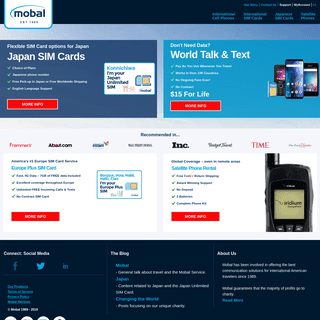
A complete backup of theyachtweek.com
Are you over 18 and want to see adult content?
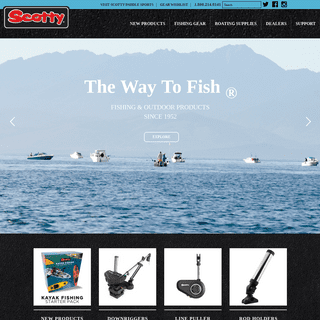
scotty | Leaders in manufacturing innovative fishing, marine and outdoor products.
Are you over 18 and want to see adult content?

Cuoieria Shop on-line Valigie e Trolley, Borse, Scarpe, Abbigliamento e Accessori
Are you over 18 and want to see adult content?
PureSanté - Des idées claires sur la santé naturelle
Are you over 18 and want to see adult content?
Мой Харьков инфо | Популярный портал Харькова - MyKharkov.info
Are you over 18 and want to see adult content?

MyGiftCardsPlus: Get cash back on gift cards purchases. Top-rated by customers
Are you over 18 and want to see adult content?

Japan Private Tours & Local Tour Guides | TripleLights
Are you over 18 and want to see adult content?
Favourite Annotations
A complete backup of packagingdigest.com
Are you over 18 and want to see adult content?

A complete backup of cryptosomniac.com
Are you over 18 and want to see adult content?
A complete backup of miriacrochesepinturas.blogspot.com
Are you over 18 and want to see adult content?
A complete backup of feltboardmagic.com
Are you over 18 and want to see adult content?

A complete backup of demimarathonlachine.com
Are you over 18 and want to see adult content?
Text
THERMODYNAMIC WORK
Thermodynamic Work. A common type of work associated by a chemical process is through gas expansion or compression. An example that you have experienced is the energy produced from the combustion ofgasoline.
ELECTRON AFFINITY TRENDS Notice the sign on the energy is negative. This is because energy is usually released in this process, as opposed to ionization energy, which requires energy. A more negative electron affinity corresponds to a greater attraction for an electron. (An unbound electron has an energy of zero.) Trends: As with ionization energy, there are two rules that govern the periodic trends of electron CHAPTER 17 - QUANTUM MECHANICS OF ROTATIONAL MOTION Rotational Angular Momentum of a System of Particles System of particles: L⃗ total divided into orbital and spin contributions ⃗L total = L⃗orbital + ⃗Lspin Imagine earth orbiting sun at origin with L⃗ orbital and spinning about its center of mass with ⃗Lspin. For molecules, relabel CHAPTER 14 - RADIATING DIPOLES IN QUANTUM MECHANICS Electric dipole moment vector operator Electric dipole moment vector operator for collection of charges is ⃗ ̂= ∑N k=1 qk⃗̂r Single charged quantum particle bound in some potential well, e.g., anegatively charged
CHAPTER 03 - KINETIC THEORY OF GASES - GRANDINETTI Force on wall is momentum change when a molecule hits it. Force along y is given by ratio of change in momentum to time between collisions. Fy = Δpy Δt vy-vy Linear momentum is conserved in collision with wall Δpy = py,final −py,initial = (−mvy)−(mvy) = −2mvy Time to travel length of box, hit wall, and travel back is Δt = 2𝓁∕vy Average force of 1 molecule hitting 1 wall ofCANNELLONI AL FORNO
Step 1: To make the besciamello: In a medium saucepan, heat butter until melted. Add flour and stir until smooth. Over medium heat, cook until light golden brown, about 6 to 7 minutes. WELCOME | LA CUCINA GRANDINETTI Maria's Sausage, Chicken and Shrimp Jambalaya. manager. Date OatmealCookies
OXTAIL RAGU
Step 3: In a 8 quart, heavy-bottomed casserole or Dutch oven, heat the olive oil over high heat until it is just smoking. Quickly dredge the oxtail in the flour and sear on CHAPTER 16 - QUANTUM PARTICLE IN THREE DIMENSIONS Derive g(E) for particle in 3D infinite well Imagine spherical shell in 3D space of nx, and ny, and nz with radius of n = √ n2 x +n2 y +n2 z = √ 8mLE h and thickness of dn associated with states in interval E +dE. Take 1/8 of surface area of sphere (4𝜋r2) times dn as number of states that lie in n to n+dn g3D(E) = 1 8 (4𝜋n2)dn dE = 𝜋n2 2 dn dE Substituting expressions for n and CHAPTER 18 - ELECTRONIC STRUCTURE OF THE HYDROGEN ATOM Two Particle Problem x y z 2 particles with mass mp and me at ⃗rp and ⃗re. Total mass, center of mass, and inter-particle distance vector is M = mp +me, R⃗= mp⃗rp +me⃗re M, and ⃗r= ⃗re −⃗rp Express ⃗rp and ⃗re in terms of M, R⃗and ⃗ras ⃗rp = R⃗− me M ⃗r and ⃗re = R⃗+ mp M ⃗r Express individual momenta of 2 particles asTHERMODYNAMIC WORK
Thermodynamic Work. A common type of work associated by a chemical process is through gas expansion or compression. An example that you have experienced is the energy produced from the combustion ofgasoline.
ELECTRON AFFINITY TRENDS Notice the sign on the energy is negative. This is because energy is usually released in this process, as opposed to ionization energy, which requires energy. A more negative electron affinity corresponds to a greater attraction for an electron. (An unbound electron has an energy of zero.) Trends: As with ionization energy, there are two rules that govern the periodic trends of electron CHAPTER 17 - QUANTUM MECHANICS OF ROTATIONAL MOTION Rotational Angular Momentum of a System of Particles System of particles: L⃗ total divided into orbital and spin contributions ⃗L total = L⃗orbital + ⃗Lspin Imagine earth orbiting sun at origin with L⃗ orbital and spinning about its center of mass with ⃗Lspin. For molecules, relabel CHAPTER 14 - RADIATING DIPOLES IN QUANTUM MECHANICS Electric dipole moment vector operator Electric dipole moment vector operator for collection of charges is ⃗ ̂= ∑N k=1 qk⃗̂r Single charged quantum particle bound in some potential well, e.g., anegatively charged
CHAPTER 03 - KINETIC THEORY OF GASES - GRANDINETTI Force on wall is momentum change when a molecule hits it. Force along y is given by ratio of change in momentum to time between collisions. Fy = Δpy Δt vy-vy Linear momentum is conserved in collision with wall Δpy = py,final −py,initial = (−mvy)−(mvy) = −2mvy Time to travel length of box, hit wall, and travel back is Δt = 2𝓁∕vy Average force of 1 molecule hitting 1 wall ofCANNELLONI AL FORNO
Step 1: To make the besciamello: In a medium saucepan, heat butter until melted. Add flour and stir until smooth. Over medium heat, cook until light golden brown, about 6 to 7 minutes. WELCOME | LA CUCINA GRANDINETTI Maria's Sausage, Chicken and Shrimp Jambalaya. manager. Date OatmealCookies
OXTAIL RAGU
Step 3: In a 8 quart, heavy-bottomed casserole or Dutch oven, heat the olive oil over high heat until it is just smoking. Quickly dredge the oxtail in the flour and sear on CHAPTER 16 - QUANTUM PARTICLE IN THREE DIMENSIONS Derive g(E) for particle in 3D infinite well Imagine spherical shell in 3D space of nx, and ny, and nz with radius of n = √ n2 x +n2 y +n2 z = √ 8mLE h and thickness of dn associated with states in interval E +dE. Take 1/8 of surface area of sphere (4𝜋r2) times dn as number of states that lie in n to n+dn g3D(E) = 1 8 (4𝜋n2)dn dE = 𝜋n2 2 dn dE Substituting expressions for n and CHAPTER 18 - ELECTRONIC STRUCTURE OF THE HYDROGEN ATOM Two Particle Problem x y z 2 particles with mass mp and me at ⃗rp and ⃗re. Total mass, center of mass, and inter-particle distance vector is M = mp +me, R⃗= mp⃗rp +me⃗re M, and ⃗r= ⃗re −⃗rp Express ⃗rp and ⃗re in terms of M, R⃗and ⃗ras ⃗rp = R⃗− me M ⃗r and ⃗re = R⃗+ mp M ⃗r Express individual momenta of 2 particles asORBITAL ENERGIES
For the hydrogen atom Z=1 so E n = - Ry/n 2. Notice that the energy level spacing decreases as n increases, that the number of orbitals (i.e. l values) increase with n, and all orbitals with the same n have the same energy (degenerate).(H-atom only). ELECTRON AFFINITY TRENDS Notice the sign on the energy is negative. This is because energy is usually released in this process, as opposed to ionization energy, which requires energy. A more negative electron affinity corresponds to a greater attraction for an electron. (An unbound electron has an energy of zero.) Trends: As with ionization energy, there are two rules that govern the periodic trends of electronLIMITING REAGENTS
A Limiting Reagent is the reagent that limits the amount of products that can be formed.. For example, nitrogen gas is prepared by passing ammonia gas over solid copper(II) oxide at high temperatures. The other products are solid copper and water vapor. RECIPES | LA CUCINA GRANDINETTI Hi Donetta, Thank you for the compliment about my family. I love them dearly too. I think you go to camp with my nieces. You don't have to be a member or sign up to view the recipes. WELCOME | LA CUCINA GRANDINETTI Maria's Sausage, Chicken and Shrimp Jambalaya. manager. Date OatmealCookies
OXTAIL RAGU
Step 3: In a 8 quart, heavy-bottomed casserole or Dutch oven, heat the olive oil over high heat until it is just smoking. Quickly dredge the oxtail in the flour and sear onDEEP FRIED SMELTS
Instructions. Step 1: In a deep fryer or deep skillet, heat oil to 375 �F (190 °C). Step 2: Rinse smelts thoroughly and pat dry. Step 3: Place the bread crumbs and flour into a large bowl. LIMONCELLO BUTTERCREAM Instructions. Step 1: Combine the sugar and egg whites in a medium heatproof bowl and place over a pan of simmering water. Step 2: Whisk the sugar mixture constantly over heat until the sugar is dissolved and the mixture looks smooth and shiny, about 3 minutes. CHAPTER 15 - TIME INDEPENDENT PERTURBATION THEORY Chapter 15 Time Independent Perturbation Theory P. J. Grandinetti Chem. 4300 P. J. Grandinetti Chapter 15: Time Independent PerturbationTheory
CHAPTER 18 - ELECTRONIC STRUCTURE OF THE HYDROGEN ATOM Two Particle Problem x y z 2 particles with mass mp and me at ⃗rp and ⃗re. Total mass, center of mass, and inter-particle distance vector is M = mp +me, R⃗= mp⃗rp +me⃗re M, and ⃗r= ⃗re −⃗rp Express ⃗rp and ⃗re in terms of M, R⃗and ⃗ras ⃗rp = R⃗− me M ⃗r and ⃗re = R⃗+ mp M ⃗r Express individual momenta of 2 particles as WELCOME | GRANDINETTI GROUPTEACHINGRESEARCHSOFTWAREPUBLICATIONSVITACONTACT Welcome to our site. Here you will find many resources for the university courses I teach and information about the ongoing program in my research group. Please take some time to explore our site, and feel free to contact me if you have any questions. Looking for my General Chemistry Tutorials?. Material on this site is based, in part,on work
IONIZATION ENERGY TRENDS Ionization Energy Trends in the Periodic Table . The ionization energy of an atom is the amount of energy required to remove an electron from the gaseous form of that atom or ion.. 1 st ionization energy - The energy required to remove the highest energy electron from a neutral gaseous atom.. For Example:THERMODYNAMIC WORK
Thermodynamic Work. A common type of work associated by a chemical process is through gas expansion or compression. An example that you have experienced is the energy produced from the combustion of gasoline. Gasoline combustion is used to create expanding gases in the cylinders of your car's engine that push out the pistons.ORBITAL ENERGIES
Using our equation for the energy of the hydrogen levels we can write an equation for the change in energy of an electron that charges orbitals and emits or absorbs a photon. Δ E = E final − E initial = h ν = h c λ = − R y Z 2 ( 1 n f 2 − 1 n i 2) With this equation we can calculate the frequency of light emitted or absorbed when an ELECTRON AFFINITY TRENDS Electron Affinity Trends. Electron Affinity is the energy associated with the addition of an electon to a gaseous atom. Example: Cl(g) + e- → Cl-(g) E.A. = -349 kJ/mol. Notice the sign on the energy is negative. This is because energy is usually released in this process,as
MOLECULAR ORBITAL THEORY The Lewis Structure approach provides an extremely simple method for determining the electronic structure of many molecules. It is a bit simplistic, however, and does have trouble predicting structures for a few molecules. Nevertheless, it gives a reasonable structure for many molecules and its simplicity to use makes it a very useful tool forchemists.
CHAPTER 17 - QUANTUM MECHANICS OF ROTATIONAL MOTION Rotational Angular Momentum of a System of Particles System of particles: L⃗ total divided into orbital and spin contributions ⃗L total = L⃗orbital + ⃗Lspin Imagine earth orbiting sun at origin with L⃗ orbital and spinning about its center of mass with ⃗Lspin. For molecules, relabel CHAPTER 14 - RADIATING DIPOLES IN QUANTUM MECHANICS Electric dipole moment vector operator Electric dipole moment vector operator for collection of charges is ⃗ ̂= ∑N k=1 qk⃗̂r Single charged quantum particle bound in some potential well, e.g., anegatively charged
NONNA'S ITALIAN WEDDING COOKIES Add flavorings. Step 2: Sift flour, baking powder and salt. Add flour and orange juice alternately to egg mixture. Add enough flour to make a soft dough. Dough will be sticky. Refrigerate overnight. Step 3: Roll out and shaped into braids or knots. Step 4: Bake on an ungreased cookie sheet 350 degree until lightly brown, about 10 minutes. CHAPTER 06 - EQUIPARTITION OF ENERGY - GRANDINETTI Diatomic gas molecules Rotational Energy For a diatomic molecule rotational energy is Erot = 1 2 Ib𝜔 2 b + 1 2 Ic𝜔 2 c Ib and Ic are principal moments of inertia and 𝜔b and 𝜔c are components of angular velocity vector. Recall: diatomic molecule is linear so Ia = 0. Vibrational Energy WELCOME | GRANDINETTI GROUPTEACHINGRESEARCHSOFTWAREPUBLICATIONSVITACONTACT Welcome to our site. Here you will find many resources for the university courses I teach and information about the ongoing program in my research group. Please take some time to explore our site, and feel free to contact me if you have any questions. Looking for my General Chemistry Tutorials?. Material on this site is based, in part,on work
IONIZATION ENERGY TRENDS Ionization Energy Trends in the Periodic Table . The ionization energy of an atom is the amount of energy required to remove an electron from the gaseous form of that atom or ion.. 1 st ionization energy - The energy required to remove the highest energy electron from a neutral gaseous atom.. For Example:THERMODYNAMIC WORK
Thermodynamic Work. A common type of work associated by a chemical process is through gas expansion or compression. An example that you have experienced is the energy produced from the combustion of gasoline. Gasoline combustion is used to create expanding gases in the cylinders of your car's engine that push out the pistons.ORBITAL ENERGIES
Using our equation for the energy of the hydrogen levels we can write an equation for the change in energy of an electron that charges orbitals and emits or absorbs a photon. Δ E = E final − E initial = h ν = h c λ = − R y Z 2 ( 1 n f 2 − 1 n i 2) With this equation we can calculate the frequency of light emitted or absorbed when an ELECTRON AFFINITY TRENDS Electron Affinity Trends. Electron Affinity is the energy associated with the addition of an electon to a gaseous atom. Example: Cl(g) + e- → Cl-(g) E.A. = -349 kJ/mol. Notice the sign on the energy is negative. This is because energy is usually released in this process,as
MOLECULAR ORBITAL THEORY The Lewis Structure approach provides an extremely simple method for determining the electronic structure of many molecules. It is a bit simplistic, however, and does have trouble predicting structures for a few molecules. Nevertheless, it gives a reasonable structure for many molecules and its simplicity to use makes it a very useful tool forchemists.
CHAPTER 17 - QUANTUM MECHANICS OF ROTATIONAL MOTION Rotational Angular Momentum of a System of Particles System of particles: L⃗ total divided into orbital and spin contributions ⃗L total = L⃗orbital + ⃗Lspin Imagine earth orbiting sun at origin with L⃗ orbital and spinning about its center of mass with ⃗Lspin. For molecules, relabel CHAPTER 14 - RADIATING DIPOLES IN QUANTUM MECHANICS Electric dipole moment vector operator Electric dipole moment vector operator for collection of charges is ⃗ ̂= ∑N k=1 qk⃗̂r Single charged quantum particle bound in some potential well, e.g., anegatively charged
NONNA'S ITALIAN WEDDING COOKIES Add flavorings. Step 2: Sift flour, baking powder and salt. Add flour and orange juice alternately to egg mixture. Add enough flour to make a soft dough. Dough will be sticky. Refrigerate overnight. Step 3: Roll out and shaped into braids or knots. Step 4: Bake on an ungreased cookie sheet 350 degree until lightly brown, about 10 minutes. CHAPTER 06 - EQUIPARTITION OF ENERGY - GRANDINETTI Diatomic gas molecules Rotational Energy For a diatomic molecule rotational energy is Erot = 1 2 Ib𝜔 2 b + 1 2 Ic𝜔 2 c Ib and Ic are principal moments of inertia and 𝜔b and 𝜔c are components of angular velocity vector. Recall: diatomic molecule is linear so Ia = 0. Vibrational Energy VSEPR | GRANDINETTI GROUP VSEPR Rules for Determining Structure. Draw the Lewis Structure. Add together the number of atoms bound to the central atom and the number of lone pair electrons and choose the appropriate arrangement. ( i.e., linear, triangular planar, tetrahedral, trigonal bipyramidal, or octahedral). Draw the structure, placing the appropriate number of QUANTUM THEORY OF LIGHT Quantum theory tells us that both light and matter consists of tiny particles which have wavelike properties associated with them. Light is composed of particles called photons, and matter is composed of particles called electrons, protons, neutrons. It's only when the mass of a particle gets small enough that its wavelike properties show up. NAMING COVALENT COMPOUNDS Rules for Binary Covalent Compounds . The prefix mono is never used for naming the first element of a compound.; The final o or a of a prefix is often dropped when the element begins with a vowel.; For example, for CO the name will be carbon monoxide, and the final o of mono is dropped. Remember, it's only the final o or a.So, the name of ClO 2 will be chlorine dioxide, and no vowels are dropped.LIMITING REAGENTS
Limiting Reagents. A Limiting Reagent is the reagent that limits the amount of products that can be formed. For example, nitrogen gas is prepared by passing ammonia gas over solid copper (II) oxide at high temperatures. The other products are solid copper and water vapor. QUANTUM THEORY OF MATTER then we can relate these two variables by recalling that the Quantum Theory says. E = h ν = hc/λ, and the theory of Relativity says. E = m c2 = p c. Then let's equate these two equations to get the DeBroglie relationship between momentum (a particle property) and wavelength (a wave property) p = h/λ. The first real experimental proof of this GAS FORMING REACTIONS Gas Forming Reactions. Sometimes a gas will be involved as one of the reactants or products in a solution reaction. For example, 2 H+(aq) + 2Cl-(aq) + 2Na+(aq) + S2-(aq) → H2S(g) + 2Na+(aq) + 2Cl-(aq) Removing the spectator ions we obtain the net ionic equation: H2S is just one example of a gaseous substance that can form in a solutionBOND POLARITY
Order the following bonds according to polarity: H-H, O-H, Cl-H, S-H, and F-H. In practice no bond is totally ionic. There will always be a small amount of electron sharing. where Q the magnitude of the charge on each end of the dipole and r is the distance between the positive and negative charges. If we have a proton and electron separated by CHAPTER 16 - QUANTUM PARTICLE IN THREE DIMENSIONS Derive g(E) for particle in 3D infinite well Imagine spherical shell in 3D space of nx, and ny, and nz with radius of n = √ n2 x +n2 y +n2 z = √ 8mLE h and thickness of dn associated with states in interval E +dE. Take 1/8 of surface area of sphere (4𝜋r2) times dn as number of states that lie in n to n+dn g3D(E) = 1 8 (4𝜋n2)dn dE = 𝜋n2 2 dn dE Substituting expressions for n and CHAPTER 18 - ELECTRONIC STRUCTURE OF THE HYDROGEN ATOM Two Particle Problem x y z 2 particles with mass mp and me at ⃗rp and ⃗re. Total mass, center of mass, and inter-particle distance vector is M = mp +me, R⃗= mp⃗rp +me⃗re M, and ⃗r= ⃗re −⃗rp Express ⃗rp and ⃗re in terms of M, R⃗and ⃗ras ⃗rp = R⃗− me M ⃗r and ⃗re = R⃗+ mp M ⃗r Express individual momenta of 2 particles as WELCOME | LA CUCINA GRANDINETTI Maria's Sausage, Chicken and Shrimp Jambalaya. manager. Date OatmealCookies
THERMODYNAMIC WORK
Thermodynamic Work. A common type of work associated by a chemical process is through gas expansion or compression. An example that you have experienced is the energy produced from the combustion of gasoline. Gasoline combustion is used to create expanding gases in the cylinders of your car's engine that push out the pistons. GENERAL CHEMISTRY TUTORIAL General Chemistry Tutorial. on Mac OS. is the only calculator app that let's you use units directly in calculations. is an intuitive multi-dimensional signal processing app on MacOS. Feel free to contact us if you have questions about the material on this page. CHAPTER 14 - RADIATING DIPOLES IN QUANTUM MECHANICS Electric dipole moment vector operator Electric dipole moment vector operator for collection of charges is ⃗ ̂= ∑N k=1 qk⃗̂r Single charged quantum particle bound in some potential well, e.g., anegatively charged
CHAPTER 03 - KINETIC THEORY OF GASES - GRANDINETTI Force on wall is momentum change when a molecule hits it. Force along y is given by ratio of change in momentum to time between collisions. Fy = Δpy Δt vy-vy Linear momentum is conserved in collision with wall Δpy = py,final −py,initial = (−mvy)−(mvy) = −2mvy Time to travel length of box, hit wall, and travel back is Δt = 2𝓁∕vy Average force of 1 molecule hitting 1 wall ofCANNELLONI AL FORNO
Step 1: To make the besciamello: In a medium saucepan, heat butter until melted. Add flour and stir until smooth. Over medium heat, cook until light golden brown, about 6 to 7 minutes. Step 2: Meanwhile, heat milk in separate pan until just about to boil. Add milk to butter mixture 1 cup at a time, whisking continuously until very smooth and CHAPTER 15 - TIME INDEPENDENT PERTURBATION THEORY Chapter 15 Time Independent Perturbation Theory P. J. Grandinetti Chem. 4300 P. J. Grandinetti Chapter 15: Time Independent PerturbationTheory
OXTAIL RAGU
Step 1: Preheat the oven to 375 degrees. Step 2: Trim the excess fat from the oxtails and season liberally with salt and pepper. Step 3: In a 8 quart, heavy-bottomed casserole or Dutch oven, heat the olive oil over high heat until it is just smoking. Quickly dredge the oxtail in the flour and sear on all sides until browned, turning with long LEMON AND EGGPLANT RISOTTO Heat up 1/3 cup of the olive oil in a frying pan and fry the eggplant dice in batches until golden and crisp. Transfer to a colander or paper-line plate and sprinkle with salt. Leave to cool. Step 4: Put the onion and remaining oil in a heavy pan and fry slowly until theonion is
WELCOME | LA CUCINA GRANDINETTI Maria's Sausage, Chicken and Shrimp Jambalaya. manager. Date OatmealCookies
BAKED HALIBUT WITH TOMATO CAPER SAUCE Step 3: For the Bake Halibut: Preheat the oven to 450 degrees. Oil a baking dish large enough for the fish to lie flat. Season the fish with salt and pepper, and arrange in the baking dish. Drizzle the olive oil over the fillets, and place a round of lemon on each one.Step 4:
THERMODYNAMIC WORK
Thermodynamic Work. A common type of work associated by a chemical process is through gas expansion or compression. An example that you have experienced is the energy produced from the combustion of gasoline. Gasoline combustion is used to create expanding gases in the cylinders of your car's engine that push out the pistons. GENERAL CHEMISTRY TUTORIAL General Chemistry Tutorial. on Mac OS. is the only calculator app that let's you use units directly in calculations. is an intuitive multi-dimensional signal processing app on MacOS. Feel free to contact us if you have questions about the material on this page. CHAPTER 14 - RADIATING DIPOLES IN QUANTUM MECHANICS Electric dipole moment vector operator Electric dipole moment vector operator for collection of charges is ⃗ ̂= ∑N k=1 qk⃗̂r Single charged quantum particle bound in some potential well, e.g., anegatively charged
CHAPTER 03 - KINETIC THEORY OF GASES - GRANDINETTI Force on wall is momentum change when a molecule hits it. Force along y is given by ratio of change in momentum to time between collisions. Fy = Δpy Δt vy-vy Linear momentum is conserved in collision with wall Δpy = py,final −py,initial = (−mvy)−(mvy) = −2mvy Time to travel length of box, hit wall, and travel back is Δt = 2𝓁∕vy Average force of 1 molecule hitting 1 wall ofCANNELLONI AL FORNO
Step 1: To make the besciamello: In a medium saucepan, heat butter until melted. Add flour and stir until smooth. Over medium heat, cook until light golden brown, about 6 to 7 minutes. Step 2: Meanwhile, heat milk in separate pan until just about to boil. Add milk to butter mixture 1 cup at a time, whisking continuously until very smooth and CHAPTER 15 - TIME INDEPENDENT PERTURBATION THEORY Chapter 15 Time Independent Perturbation Theory P. J. Grandinetti Chem. 4300 P. J. Grandinetti Chapter 15: Time Independent PerturbationTheory
OXTAIL RAGU
Step 1: Preheat the oven to 375 degrees. Step 2: Trim the excess fat from the oxtails and season liberally with salt and pepper. Step 3: In a 8 quart, heavy-bottomed casserole or Dutch oven, heat the olive oil over high heat until it is just smoking. Quickly dredge the oxtail in the flour and sear on all sides until browned, turning with long LEMON AND EGGPLANT RISOTTO Heat up 1/3 cup of the olive oil in a frying pan and fry the eggplant dice in batches until golden and crisp. Transfer to a colander or paper-line plate and sprinkle with salt. Leave to cool. Step 4: Put the onion and remaining oil in a heavy pan and fry slowly until theonion is
WELCOME | LA CUCINA GRANDINETTI Maria's Sausage, Chicken and Shrimp Jambalaya. manager. Date OatmealCookies
BAKED HALIBUT WITH TOMATO CAPER SAUCE Step 3: For the Bake Halibut: Preheat the oven to 450 degrees. Oil a baking dish large enough for the fish to lie flat. Season the fish with salt and pepper, and arrange in the baking dish. Drizzle the olive oil over the fillets, and place a round of lemon on each one.Step 4:
WELCOME | GRANDINETTI GROUP Welcome to our site. Here you will find many resources for the university courses I teach and information about the ongoing program in my research group. Please take some time to explore our site, and feel free to contact me if you have any questions. Looking for my General Chemistry Tutorials?. Material on this site is based, in part,on work
IONIZATION ENERGY TRENDS Ionization Energy Trends in the Periodic Table . The ionization energy of an atom is the amount of energy required to remove an electron from the gaseous form of that atom or ion.. 1 st ionization energy - The energy required to remove the highest energy electron from a neutral gaseous atom.. For Example:ORBITAL ENERGIES
Using our equation for the energy of the hydrogen levels we can write an equation for the change in energy of an electron that charges orbitals and emits or absorbs a photon. Δ E = E final − E initial = h ν = h c λ = − R y Z 2 ( 1 n f 2 − 1 n i 2) With this equation we can calculate the frequency of light emitted or absorbed when anTHERMODYNAMIC WORK
Thermodynamic Work. A common type of work associated by a chemical process is through gas expansion or compression. An example that you have experienced is the energy produced from the combustion of gasoline. Gasoline combustion is used to create expanding gases in the cylinders of your car's engine that push out the pistons. MOLECULAR ORBITAL THEORY The Lewis Structure approach provides an extremely simple method for determining the electronic structure of many molecules. It is a bit simplistic, however, and does have trouble predicting structures for a few molecules. Nevertheless, it gives a reasonable structure for many molecules and its simplicity to use makes it a very useful tool forchemists.
CHAPTER 17 - QUANTUM MECHANICS OF ROTATIONAL MOTION Rotational Angular Momentum of a System of Particles System of particles: L⃗ total divided into orbital and spin contributions ⃗L total = L⃗orbital + ⃗Lspin Imagine earth orbiting sun at origin with L⃗ orbital and spinning about its center of mass with ⃗Lspin. For molecules, relabelDEEP FRIED SMELTS
Step 3: Place the bread crumbs and flour into a large bowl. Whisk egg, water, and milk into a bowl. Dip the smelts into the bread crumb/flour mixture, then into the egg mixture, then back into the flour mixture until the fish are well coated. Shake off excess. Step 4: Drop smelts into deep fryer or skillet with heated oil and cook until golden. WELCOME | LA CUCINA GRANDINETTI Maria's Sausage, Chicken and Shrimp Jambalaya. manager. Date OatmealCookies
CHAPTER 06 - EQUIPARTITION OF ENERGY - GRANDINETTI Diatomic gas molecules Rotational Energy For a diatomic molecule rotational energy is Erot = 1 2 Ib𝜔 2 b + 1 2 Ic𝜔 2 c Ib and Ic are principal moments of inertia and 𝜔b and 𝜔c are components of angular velocity vector. Recall: diatomic molecule is linear so Ia = 0. Vibrational Energy CHAPTER 20 - IDENTICAL PARTICLES IN QUANTUM MECHANICS When particles are indistinguishable QM treatment of 2 e− system must account for indistinguishability. Writing Hamiltonian for 2 electrons requires us to use labels, ̂ (1,2) = ⃗p̂ 2 1 2me ⃗p̂ 2 2me +V̂(⃗r 1,⃗r2) To preserve indistinguishability Hamiltonian must be invariant to particle exchange, Skip to main contentSECONDARY MENU
* Log in
GRANDINETTI GROUP
MAIN MENU
* Teaching
* Research
* Software
* Publications
* Vita
* Contact
* Donate
Menu
SEARCH FORM
Search
WELCOME
Welcome to our site. Here you will find many resources for the university courses I teach and information about the ongoing program in my research group. Please take some time to explore our site, and feel free to contact me if you have any questions. Looking for my GENERAL CHEMISTRY TUTORIALS?.
Here are the slides and video for my 2016 ENC tutorial on Phase Cycling, and here are the slidesfor my 2020
Winter School lecture Primer of NMR Electronics, and the slides for my 2020 Winter School lecture Solid-State NMR of Quadrupolar Nuclei, Material on this site is based, in part, on work supported by the U.S. National Science Foundation (NSF). Opinions, findings and conclusions or recomendations expressed in this material do not necessarily reflect the views of the U.S. NSF. � Copyright 1995-2019, Philip J. Grandinetti Privacy -- Terms of useDetails
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0