Are you over 18 and want to see adult content?
More Annotations

A complete backup of cyfrowypolsat.pl
Are you over 18 and want to see adult content?

A complete backup of pacificweekofagriculture.com
Are you over 18 and want to see adult content?
A complete backup of standardaero.com
Are you over 18 and want to see adult content?

A complete backup of singaporepools.com.sg
Are you over 18 and want to see adult content?
A complete backup of bizzarroceramica.com
Are you over 18 and want to see adult content?

A complete backup of girlsheaven-job.net
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of https://justbusiness.com
Are you over 18 and want to see adult content?

A complete backup of https://specialarad.ro
Are you over 18 and want to see adult content?

A complete backup of https://teranicouture.com
Are you over 18 and want to see adult content?
A complete backup of https://cuonda.com
Are you over 18 and want to see adult content?
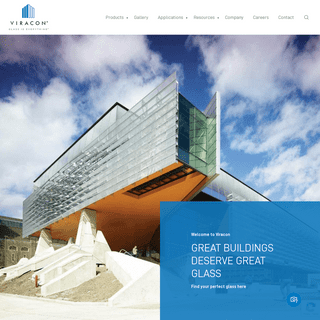
A complete backup of https://viracon.com
Are you over 18 and want to see adult content?
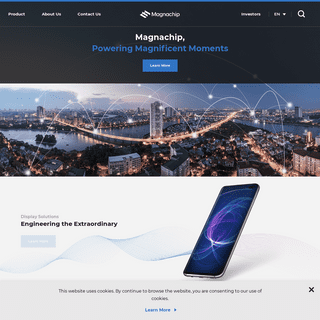
A complete backup of https://magnachip.cn
Are you over 18 and want to see adult content?
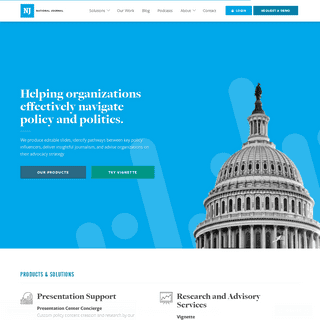
A complete backup of https://nationaljournal.com
Are you over 18 and want to see adult content?

A complete backup of https://leancoffee.org
Are you over 18 and want to see adult content?

A complete backup of https://eldoradio.de
Are you over 18 and want to see adult content?

A complete backup of https://kiwirail.co.nz
Are you over 18 and want to see adult content?

A complete backup of https://sparklecat.com
Are you over 18 and want to see adult content?
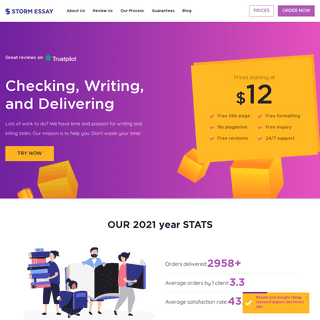
A complete backup of https://stormessay.org
Are you over 18 and want to see adult content?
Text
LOGIN - MYBMS
Loading Skip to page content. Skip to page content INDEPENDENT RESEARCH t Bristol Myers Squibb, we are committed to providing innovative, high-quality medicines that strive to address the unmet medical needs of patients with serious diseases.Additionally, Bristol Myers Squibb wishes to advance science through supporting novel, independent research that addresses unmet need surrounding our products andtherapeutic areas.
BMS LOGIN
This resource is restricted to authorized users. BMS User ID. BMSPassword
BRISTOL MYERS SQUIBB Opdivo is the first and only PD-1/L1 inhibitor to demonstrate superior first-line efficacy in upper GI cancers across histologies and tumor locations. Statistically significant overall survival benefit demonstrated with Opdivo plus chemotherapy and Opdivo plus Yervoy in PD-L1 positive and all-randomized populations in CheckMate -648 (PRINCETON, N.J., April 8, 2021) – Bristol Myers SquibbBMS 360 - HOME
BMS 360 - Home
PRODUCT MONOGRAPH
Page 1 of 73. PRODUCT MONOGRAPH . Pr ORENCIA®. abatacept for injection . Intravenous Infusion, 250 mg / vial . Pr ®ORENCIA. abatacept injection . Solution for Subcutaneous Injection, 125 mg / mLMY TRIALS AT BMS
This website is best viewed with the following browsers: Chrome, Internet Explorer (6.0 and above) or Firefox (1.5 and above).BMS 360
This resource is restricted to authorized users. BMS User ID. BMSPassword
GLOBAL BIOPHARMACEUTICAL COMPANY Bristol Myers Squibb is a global biopharmaceutical company committed to discovering, developing and delivering innovative medicines to patients with serious diseases. BRISTOL MYERS SQUIBB PRODUCTS & MEDICINES For Medical Information, or to Report Side Effects/Adverse Reactions or Product Quality Complaints (Pharmacovigilance) 1-866-463-6267. (toll free) Expand All. ABRAXANE ® for Injectable Suspension (paclitaxel powder for injectable suspension nanoparticle, albumin-bound (nab®) paclitaxel) ABRAXANE Product Monograph.LOGIN - MYBMS
Loading Skip to page content. Skip to page content INDEPENDENT RESEARCH t Bristol Myers Squibb, we are committed to providing innovative, high-quality medicines that strive to address the unmet medical needs of patients with serious diseases.Additionally, Bristol Myers Squibb wishes to advance science through supporting novel, independent research that addresses unmet need surrounding our products andtherapeutic areas.
BMS LOGIN
This resource is restricted to authorized users. BMS User ID. BMSPassword
BRISTOL MYERS SQUIBB Opdivo is the first and only PD-1/L1 inhibitor to demonstrate superior first-line efficacy in upper GI cancers across histologies and tumor locations. Statistically significant overall survival benefit demonstrated with Opdivo plus chemotherapy and Opdivo plus Yervoy in PD-L1 positive and all-randomized populations in CheckMate -648 (PRINCETON, N.J., April 8, 2021) – Bristol Myers SquibbBMS 360 - HOME
BMS 360 - Home
PRODUCT MONOGRAPH
Page 1 of 73. PRODUCT MONOGRAPH . Pr ORENCIA®. abatacept for injection . Intravenous Infusion, 250 mg / vial . Pr ®ORENCIA. abatacept injection . Solution for Subcutaneous Injection, 125 mg / mLMY TRIALS AT BMS
This website is best viewed with the following browsers: Chrome, Internet Explorer (6.0 and above) or Firefox (1.5 and above).BMS 360
This resource is restricted to authorized users. BMS User ID. BMSPassword
BRISTOL MYERS SQUIBB Discover why the new Bristol Myers Squibb has what it takes to help more patients than ever before. Learn More A Letter From Our CEO. The I-O Quest. Exploring the research behind why some people respond to immunotherapy and others do not. Read More. INDEPENDENT RESEARCH t Bristol Myers Squibb, we are committed to providing innovative, high-quality medicines that strive to address the unmet medical needs of patients with serious diseases.Additionally, Bristol Myers Squibb wishes to advance science through supporting novel, independent research that addresses unmet need surrounding our products andtherapeutic areas.
CONTACT US - BRISTOL MYERS SQUIBB - BMS Contact Bristol Myers Squibb through our contact form, customer relations line or live chat. For product side-effect inquiries call(800) 721-5072.
BRISTOL MYERS SQUIBB (BMS) Pour en savoir plus sur Bristol Myers Squibb et sa mission visant � découvrir, développer et fournir des médicaments innovants aux patients atteints de maladies graves.ANNUAL REPORTS
Forward-looking statements contained on this website should be evaluated together with the many uncertainties that affect Bristol Myers Squibb's business, particularly those identified in the cautionary factors discussion in the company’s most recent annual report on Form 10-K, quarterly reports on Form 10-Q and current reports on Form 8-K. BRISTOL MYERS SQUIBB CAREERS Equal Employment Opportunity (EEO) / Disability Assistance. Bristol Myers Squibb is an equal opportunity employer. Qualified applicants will receive consideration for employment without regard to race, color, religion, sex, sexual orientation, gender identity, national origin, age, disability, protected veteran status, pregnancy, citizenship, marital status, gender expression, genetic GRANTS, GIVING & CORPORATE SPONSORSHIP SUPPORT Grants, Giving & Corporate Sponsorship Support. Bristol Myers Squibb provides significant financial and in-kind support to make a difference in people’s lives. Listed below are the various type of initiatives which Bristol Myers Squibb supports. Organizations may apply for any of these types of support using our simple, unifiedLetter of Request.
PULSE WELCOME
Pulse est une nouvelle plateforme de motivation des collaborateurs où vous pourrez recevoir d’importantes informations et mises à jour commerciales concernant notre entreprise. Vous pourrez personnaliser les nouvelles que vous recevez, interagir avec des collègues, partager des retours d’expérience et vous plonger dans notreculture
BRISTOL MYERS SQUIBB PRINCETON, N.J.--(BUSINESS WIRE)-- Bristol Myers Squibb (NYSE:BMY) today announced that additional data from multiple studies evaluating Zeposia (ozanimod) in ulcerative colitis (UC) were presented at Digestive Disease Week ® (DDW), taking place virtually May 21-23, 2021. These data deepen the understanding of Zeposia and reinforce Bristol Myers Squibb’s commitment to understanding and BRISTOL MYERS SQUIBB First presentation of Phase 3 data from a trial evaluating a LAG-3-blocking antibody Fixed-dose combination of relatlimab and nivolumab demonstrated statistically significant and clinically meaningful benefit over Opdivo monotherapy, an established standard of care Data demonstrate inhibiting LAG-3 together with PD-1 helps improve outcomes for patients Data to be featured in an oralLOGIN - MYBMS
Loading Skip to page content. Skip to page contentBMS LOGIN
This resource is restricted to authorized users. BMS User ID. BMSPassword
GRANTS, GIVING & CORPORATE SPONSORSHIP SUPPORT Grants, Giving & Corporate Sponsorship Support. Bristol Myers Squibb provides significant financial and in-kind support to make a difference in people’s lives. Listed below are the various type of initiatives which Bristol Myers Squibb supports. Organizations may apply for any of these types of support using our simple, unifiedLetter of Request.
BRISTOL MYERS SQUIBB Bristol Myers Squibb (NYSE: BMY) and The Rockefeller University today announced that they have entered into a definitive agreement under which Bristol Myers Squibb has been granted a global exclusive license to develop, manufacture and commercialize Rockefeller’s novel monoclonal antibody (“mAb”) duo treatment that neutralizes the SARS-CoV-2 virus for therapy or prevention of COVID-19.PULSE WELCOME
Pulse est une nouvelle plateforme de motivation des collaborateurs où vous pourrez recevoir d’importantes informations et mises à jour commerciales concernant notre entreprise. Vous pourrez personnaliser les nouvelles que vous recevez, interagir avec des collègues, partager des retours d’expérience et vous plonger dans notreculture
BRISTOL MYERS SQUIBB Opdivo is the first and only PD-1/L1 inhibitor to demonstrate superior first-line efficacy in upper GI cancers across histologies and tumor locations. Statistically significant overall survival benefit demonstrated with Opdivo plus chemotherapy and Opdivo plus Yervoy in PD-L1 positive and all-randomized populations in CheckMate -648 (PRINCETON, N.J., April 8, 2021) – Bristol Myers SquibbMY TRIALS AT BMS
This website is best viewed with the following browsers: Chrome, Internet Explorer (6.0 and above) or Firefox (1.5 and above). BRISTOL MYERS SQUIBB Data from pivotal KarMMa study to be presented at ASCO21 show 24.8-month median overall survival in triple-class exposed multiple myeloma With more than 24-month median follow-up, results represent longest follow-up to date from a global clinical trial of a CAR T cell therapy in multiple myeloma with 73% overall response rate and responses ongoing Analysis of characteristics ofBMS 360 - HOME
BMS 360 - Home
BMS 360
This resource is restricted to authorized users. BMS User ID. BMSPassword
LOGIN - MYBMS
Loading Skip to page content. Skip to page contentBMS LOGIN
This resource is restricted to authorized users. BMS User ID. BMSPassword
GRANTS, GIVING & CORPORATE SPONSORSHIP SUPPORT Grants, Giving & Corporate Sponsorship Support. Bristol Myers Squibb provides significant financial and in-kind support to make a difference in people’s lives. Listed below are the various type of initiatives which Bristol Myers Squibb supports. Organizations may apply for any of these types of support using our simple, unifiedLetter of Request.
BRISTOL MYERS SQUIBB Bristol Myers Squibb (NYSE: BMY) and The Rockefeller University today announced that they have entered into a definitive agreement under which Bristol Myers Squibb has been granted a global exclusive license to develop, manufacture and commercialize Rockefeller’s novel monoclonal antibody (“mAb”) duo treatment that neutralizes the SARS-CoV-2 virus for therapy or prevention of COVID-19.PULSE WELCOME
Pulse est une nouvelle plateforme de motivation des collaborateurs où vous pourrez recevoir d’importantes informations et mises à jour commerciales concernant notre entreprise. Vous pourrez personnaliser les nouvelles que vous recevez, interagir avec des collègues, partager des retours d’expérience et vous plonger dans notreculture
BRISTOL MYERS SQUIBB Opdivo is the first and only PD-1/L1 inhibitor to demonstrate superior first-line efficacy in upper GI cancers across histologies and tumor locations. Statistically significant overall survival benefit demonstrated with Opdivo plus chemotherapy and Opdivo plus Yervoy in PD-L1 positive and all-randomized populations in CheckMate -648 (PRINCETON, N.J., April 8, 2021) – Bristol Myers SquibbMY TRIALS AT BMS
This website is best viewed with the following browsers: Chrome, Internet Explorer (6.0 and above) or Firefox (1.5 and above). BRISTOL MYERS SQUIBB Data from pivotal KarMMa study to be presented at ASCO21 show 24.8-month median overall survival in triple-class exposed multiple myeloma With more than 24-month median follow-up, results represent longest follow-up to date from a global clinical trial of a CAR T cell therapy in multiple myeloma with 73% overall response rate and responses ongoing Analysis of characteristics ofBMS 360 - HOME
BMS 360 - Home
BMS 360
This resource is restricted to authorized users. BMS User ID. BMSPassword
BRISTOL MYERS SQUIBB Learn more about Bristol Myers Squibb and our mission to discover, develop and deliver innovative medicines to patients with seriousdiseases.
BRISTOL MYERS SQUIBB Bristol Myers Squibb BRISTOL MYERS SQUIBB Bristol Myers Squibb (NYSE: BMY) and The Rockefeller University today announced that they have entered into a definitive agreement under which Bristol Myers Squibb has been granted a global exclusive license to develop, manufacture and commercialize Rockefeller’s novel monoclonal antibody (“mAb”) duo treatment that neutralizes the SARS-CoV-2 virus for therapy or prevention of COVID-19. BRISTOL MYERS SQUIBB CAREERS Equal Employment Opportunity (EEO) / Disability Assistance. Bristol Myers Squibb is an equal opportunity employer. Qualified applicants will receive consideration for employment without regard to race, color, religion, sex, sexual orientation, gender identity, national origin, age, disability, protected veteran status, pregnancy, citizenship, marital status, gender expression, genetic BRISTOL MYERS SQUIBB Opdivo demonstrated significant overall survival benefit over chemotherapy alone in both PD-L1 positive and all-randomized populations in both treatment arms CheckMate -648 marks the third global trial in which Opdivo demonstrated a significant benefit for patients with upper gastrointestinal cancers Data to be featured in an oral presentation during the 2021 American Society of ClinicalPRODUCT MONOGRAPH
Page 1 of 73. PRODUCT MONOGRAPH . Pr ORENCIA®. abatacept for injection . Intravenous Infusion, 250 mg / vial . Pr ®ORENCIA. abatacept injection . Solution for Subcutaneous Injection, 125 mg / mL BRISTOL MYERS SQUIBB Bristol Myers Squibb (NYSE:BMY) today announced that data from 28 company-sponsored and investigator-sponsored studies will be presented at the EULAR 2021 Virtual Congress taking place June 2-5, 2021. The research highlights the depth and strength of the company's growing immunology pipeline and portfolio, commitment to the rheumatology research community and focus on deliveringMEET TONY - BMS.COM
Born and raised in China, Tony Jiang grew up spending much of his childhood with his beloved grandmother. After Tony moved to the United States, his grandmother was diagnosed with cancer. BRISTOL MYERS SQUIBB Patients treated with Opdivo plus Yervoy with two cycles of chemotherapy showed sustained improvements in overall survival and progression-free survival and increased duration of response vs. chemotherapy at two years Dual immunotherapy-based combination demonstrated clinical benefit across key subgroups of patients, regardless of PD-L1 expression level or histology Data to beMEET MATT - BMS.COM
When Matt Labrecque first experienced the tingling sensation in his thigh, it felt almost like his leg was falling asleep. This tingling sensation was different though. BRISTOL MYERS SQUIBB Discover why the new Bristol Myers Squibb has what it takes to help more patients than ever before. Learn More A Letter From Our CEO. The I-O Quest. Exploring the research behind why some people respond to immunotherapy and others do not. Read More. BRISTOL MYERS SQUIBB Learn more about Bristol Myers Squibb and our mission to discover, develop and deliver innovative medicines to patients with seriousdiseases.
BRISTOL MYERS SQUIBB PRODUCTS & MEDICINES For Medical Information, or to Report Side Effects/Adverse Reactions or Product Quality Complaints (Pharmacovigilance) 1-866-463-6267. (toll free) Expand All. ABRAXANE ® for Injectable Suspension (paclitaxel powder for injectable suspension nanoparticle, albumin-bound (nab®) paclitaxel) ABRAXANE Product Monograph. BRISTOL MYERS SQUIBB Bristol Myers SquibbBMS LOGIN
This resource is restricted to authorized users. BMS User ID. BMSPassword
BRISTOL MYERS SQUIBB CAREERS Equal Employment Opportunity (EEO) / Disability Assistance. Bristol Myers Squibb is an equal opportunity employer. Qualified applicants will receive consideration for employment without regard to race, color, religion, sex, sexual orientation, gender identity, national origin, age, disability, protected veteran status, pregnancy, citizenship, marital status, gender expression, genetic BRISTOL MYERS SQUIBB Discover why the new Bristol Myers Squibb has what it takes to help more patients than ever before. Learn More A Letter From Our CEO. The I-O Quest. Exploring the research behind why some people respond to immunotherapy and others do not. Read More. BRISTOL MYERS SQUIBB Learn more about Bristol Myers Squibb and our mission to discover, develop and deliver innovative medicines to patients with seriousdiseases.
BRISTOL MYERS SQUIBB PRODUCTS & MEDICINES For Medical Information, or to Report Side Effects/Adverse Reactions or Product Quality Complaints (Pharmacovigilance) 1-866-463-6267. (toll free) Expand All. ABRAXANE ® for Injectable Suspension (paclitaxel powder for injectable suspension nanoparticle, albumin-bound (nab®) paclitaxel) ABRAXANE Product Monograph. BRISTOL MYERS SQUIBB Bristol Myers SquibbBMS LOGIN
This resource is restricted to authorized users. BMS User ID. BMSPassword
BRISTOL MYERS SQUIBB CAREERS Equal Employment Opportunity (EEO) / Disability Assistance. Bristol Myers Squibb is an equal opportunity employer. Qualified applicants will receive consideration for employment without regard to race, color, religion, sex, sexual orientation, gender identity, national origin, age, disability, protected veteran status, pregnancy, citizenship, marital status, gender expression, genetic BRISTOL MYERS SQUIBB Discover why the new Bristol Myers Squibb has what it takes to help more patients than ever before. Learn More A Letter From Our CEO. The I-O Quest. Exploring the research behind why some people respond to immunotherapy and others do not. Read More. BRISTOL MYERS SQUIBB PRODUCTS & MEDICINES For Medical Information, or to Report Side Effects/Adverse Reactions or Product Quality Complaints (Pharmacovigilance) 1-866-463-6267. (toll free) Expand All. ABRAXANE ® for Injectable Suspension (paclitaxel powder for injectable suspension nanoparticle, albumin-bound (nab®) paclitaxel) ABRAXANE Product Monograph. BRISTOL MYERS SQUIBB Bristol Myers Squibb CONTACT US - BRISTOL MYERS SQUIBB - BMS Contact Bristol Myers Squibb through our contact form, customer relations line or live chat. For product side-effect inquiries call(800) 721-5072.
BRISTOL MYERS SQUIBB: INVESTOR RELATIONS 05/21/2021. Bristol Myers Squibb Receives Positive CHMP Opinion Recommending Opdivo (nivolumab) plus Yervoy (ipilimumab) for Treatment of Mismatch Repair Deficient or Microsatellite Instability–High Metastatic Colorectal Cancer After Prior Chemotherapy.MEET TONY - BMS.COM
Born and raised in China, Tony Jiang grew up spending much of his childhood with his beloved grandmother. After Tony moved to the United States, his grandmother was diagnosed with cancer.MEET DANIEL
As a young adult growing up in Chile, Daniel had many life plans still ahead of him. All of that shifted when he was diagnosed with cancer, which impacted his outlook on life and his future.MEET MATT - BMS.COM
When Matt Labrecque first experienced the tingling sensation in his thigh, it felt almost like his leg was falling asleep. This tingling sensation was different though.BMS LOGIN
This resource is restricted to authorized users. BMS User ID. BMS Password + Security Code 1 - BMS O365 LANDING PAGE Sign in to your account. Sign in. Enter BMS email address orUserID@bms.com.
Our Medicines Contact UsCareers _ _
_ _ United States
_ Close Icon _
Americas
Argentina
Spanish
Brazil
Portuguese
Canada
English French
Chile
Spanish
Colombia
Spanish
Mexico
Spanish
Peru
Spanish
United States
English
Venezuela
Spanish
Asia Pacific
Australia
English
China
Simplified Chinese
India
English
Japan
Japanese
Korea
Korean
New Zealand
English
Taiwan
Traditional Chinese
Europe
Austria
German
Belgium
English Dutch FrenchCzech Republic
English
Denmark
Danish
Finland
Finnish
France
French
Germany
German
Greece
Greek
Hungary
Hungarian
Ireland
English
Italy
Italian
Luxembourg
English
Netherlands
Dutch
Norway
Norwegian
Poland
Polish
Portugal
English
Romania
Romanian
Russia
Russian
Spain
Spanish
Sweden
Swedish
Switzerland
German French
United Kingdom
English
Middle East
Saudi Arabia
English Arabic
Turkey
Turkish
United Arab EmiratesEnglish Arabic
Other
Other Markets
Distributor Markets
_ __ _
_ _
_ Previous Page Icon _ _ Previous Page Icon _ Patients & Caregivers_ Next Page Icon _
Our Medicines
Get Help Paying For Your Medicine Risk Evaluation and Mitigation Strategies (REMS)FEATURED
* Get Help Paying For Your Medicine Healthcare Providers_ Next Page Icon _
Pre-Approval Access to Investigational Medicines_ Next Page Icon _
Investigational drugs availableFEATURED
* COVID-19 Updates
Researchers
_ Next Page Icon _
Areas of Focus
In the Pipeline
Our R&D Team
_ Next Page Icon _
Researcher Bios
Research Publications "Golden Ticket" Contests Translational Medicine Clinical Trials & Research_ Next Page Icon _
Disclosure Commitment Independent Research_ Next Page Icon _
Investigator Sponsored Research Portal Areas of Interest & Competitive Research Grants Compound & Technology Requests Data Sharing Request ProcessFEATURED
* Area of Focus
Investors
_ Next Page Icon _
Stock information
_ Next Page Icon _
Dividend history
Stock split history
Investment return calculatorFinancial reporting
_ Next Page Icon _
SEC filings
Quarterly Results
Product Sales summaryAnnual Reports
Analyst coverage
Events and presentations Shareholder Services_ Next Page Icon _
Annual Meeting of Shareholders Cost Basis calculator Acquisition FAQs for Celgene Shareholders Acquisition-related information for Celgene shareholders_ Next Page Icon _
Tax Implications of Celgene Acquisition Acquisition FAQs for Celgene ShareholdersInvestor contacts
_ Next Page Icon _
Investor Alerts Subscribe Investor Alerts UnsubscribeRSS feeds
FEATURED
* Diversity and Inclusion Report Business Development_ Next Page Icon _
Our Partnerships
Why Partner With Us
Areas of Interest
Business Development Leadership TeamBegin a Discussion
FEATURED
* Diversity and Inclusion ReportNews
_ Next Page Icon _
Press Releases
_ Next Page Icon _
RSS Feeds
Unsubscribe to News Email Alerts - Bristol Myers Squibb Subscribe to New Email Alerts - Bristol Myers Squibb Life & Science Stories_ Next Page Icon _
All
People
Science
News & Perspectives
Medical Meetings
_ Next Page Icon _
BMS at EULAR 2021 Congress BMS at Digestive Disease Week® (DDW) 2021 BMS at ASCO and EHA 2021BMS at AAD VMX 2021
Media Library
_ Next Page Icon _
Scientific Media Resources Disease State InfographicsMedia Contacts
FEATURED
* BMS at ASCO and EHA 2021About Us
_ Next Page Icon _
Our Company
_ Next Page Icon _
Our Mission, Vision, Values & CommitmentOur Principles
History Timeline
Survivorship Today
Committed to Creating a Better Future for Cancer PatientsGlobal Patient Week
Patient & Employee StoriesGovernance
Doing Business with Us as a SupplierAchievements
Worldwide FacilitiesCorporate Brand
Leadership
_ Next Page Icon _
Leadership Team
Board of Directors
People & Business Resource Groups Leadership Team Global Diversity & Inclusion_ Next Page Icon _
Our Commitments
Global Diversity and Inclusion Report People & Business Resource Groups Learning & Development Programs Our Commitment to Equal Employment Opportunity Workforce Statistics Giovanni Caforio reflects to recent Asian American hate crimesResponsibility
_ Next Page Icon _
COVID-19 Updates
Bristol Myers Squibb FoundationCorporate Giving
Independent Medical Education Supporting Our Communities Access to Medicines in the Developing World Position on Key IssuesSustainability
_ Next Page Icon _
Goals & Key Indicators Economic ResponsibilityGovernance
Social Progress
Environmental Performance Global Reporting Initiative Sustainability ResourcesSubject Index
Contact Us
_ Next Page Icon _
Social
FEATURED
* Diversity and Inclusion Report_ _
Search
Medicines > Careers >Press Releases >
Medical Information > BRISTOL MYERS SQUIBB - GLOBAL BIOPHARMACEUTICAL COMPANYHER PERSPECTIVE
Tackling Mount Kilimanjaro and your career journeyLearn More
IN THIS TOGETHER:
Our Commitment to Global Diversity and InclusionLearn More
1 / 2
NEXT
In This Together:
NEW FDA APPROVAL >
Bristol Myers Squibb’s new oral treatment option is now approved for appropriate patients with ulcerative colitis.OUR R&D TEAM >
Learn more about the backgrounds, scientific pursuits and personal interests of members of our research team. PATIENT & EMPLOYEE STORIES > We invite you to meet some of our people — employees who embody and give expression to our mission, and patients whose determination fuels and energizes our work. Their words are personal and powerful, and together, they help tell the Bristol Myers Squibbstory.
LIFE & SCIENCE STORIES A subset of BMS.com, Life & Science Stories is where we share the work our global community of colleagues are doing around the world to help patients and advance scientific innovation.Learn More >
May 26, 2021 World Multiple Sclerosis Day 2021: a Q&A with Cathy Trzaskawka and Richard Nieman _ _ May 25, 2021 Harnessing translational medicine to guide drug discovery and development _ _ May 19, 2021 Fighting the stigma: How candid conversations may help men facing cancer _ _ May 19, 2021 News & Perspectives Are you familiar with AFib symptoms and the increased risk of stroke associated with thecondition? _ _
May 19, 2021 Science Understanding the potential of LAG-3 _ _ May 18, 2021 News & Perspectives Patient engagement and diversity in clinical trials _ _SURVIVORSHIP TODAY
Emmy winner Sterling K. Brown explores cancer survivorship during aglobal pandemic.
Learn More
_ Play Icon _
* Bin’s journey during COVID-19_ Play Icon _
* AJ’s day in the life_ Play Icon _
* Relationships afterwardsMOVING FORWARD.
TOGETHER.
Careers
We’re looking for people who want to change lives. Join us as we transform patients’ lives through science.* Learn More
Investors
Learn more about how we strive to produce sustained strong performance and shareholder value.* Learn More
LOOKING FOR ANSWERS? Quick links to helpful resources. ------------------------- Clinical Trial Information for Researchers Clinical trials and research are a critical part of bringing new medicines to patients.* Learn More
* Find a Clinical Trial ------------------------- Medical Information for Healthcare Providers Information and resources to assist with caring for patients. * Resources for U.S. Healthcare Providers * Global Medical Information ------------------------- Help Paying for Your Medicines If you are a patient in the U.S. struggling to understand your coverage or pay for your medication, we may be able to help.* Find Out How
QUICK LINKS
* Home
* Patients & Caregivers * Healthcare Providers * Researchers & Partners* Investors
* Careers
* News
RESOURCES
* Our Medicines
* Life & Science Stories* About Us
* UK-CA Slavery and Human Trafficking Statement (PDF)FOLLOW US
*
*
*
*
*
* Legal Notice
* Privacy Policy
* Trademarks
* Forward-Looking Statement * Worldwide Locations* Contact Us
*
May 28, 2021
� 2021 Bristol-Myers Squibb CompanyDetails
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0