Are you over 18 and want to see adult content?
More Annotations
A complete backup of http://nitrocode.ir/
Are you over 18 and want to see adult content?
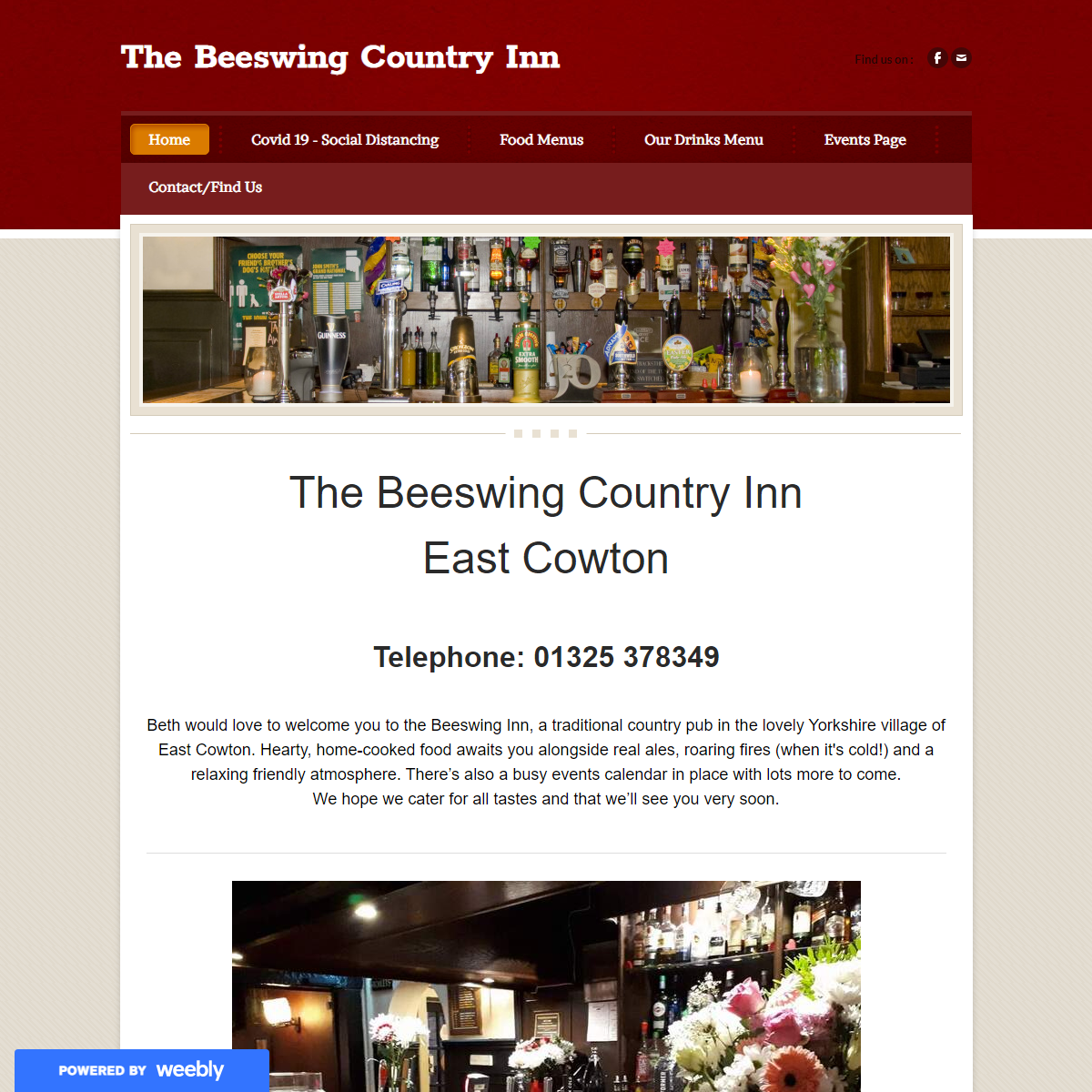
A complete backup of https://thebeeswing.weebly.com/
Are you over 18 and want to see adult content?
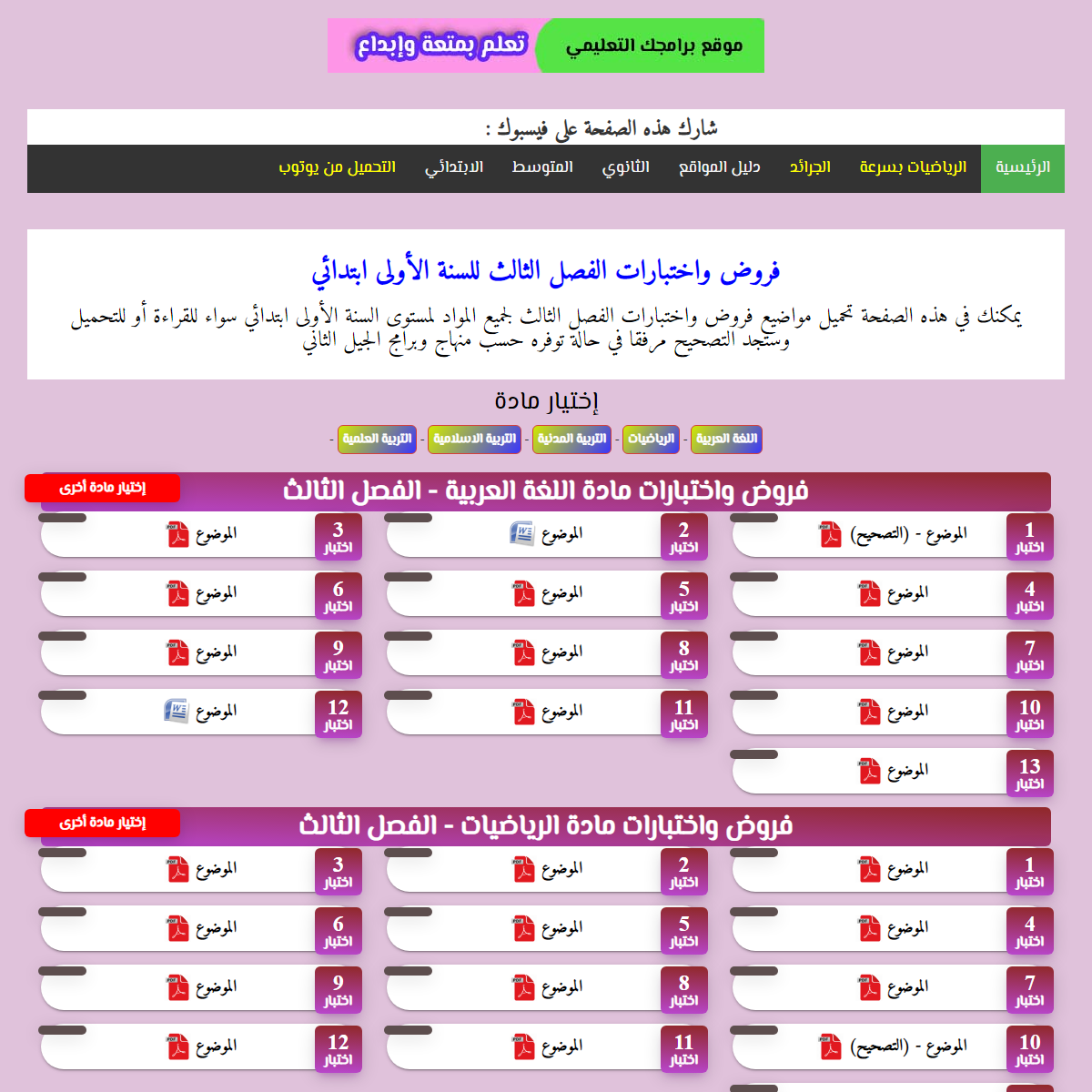
A complete backup of https://baramjak.com/prim1-exam3.php
Are you over 18 and want to see adult content?
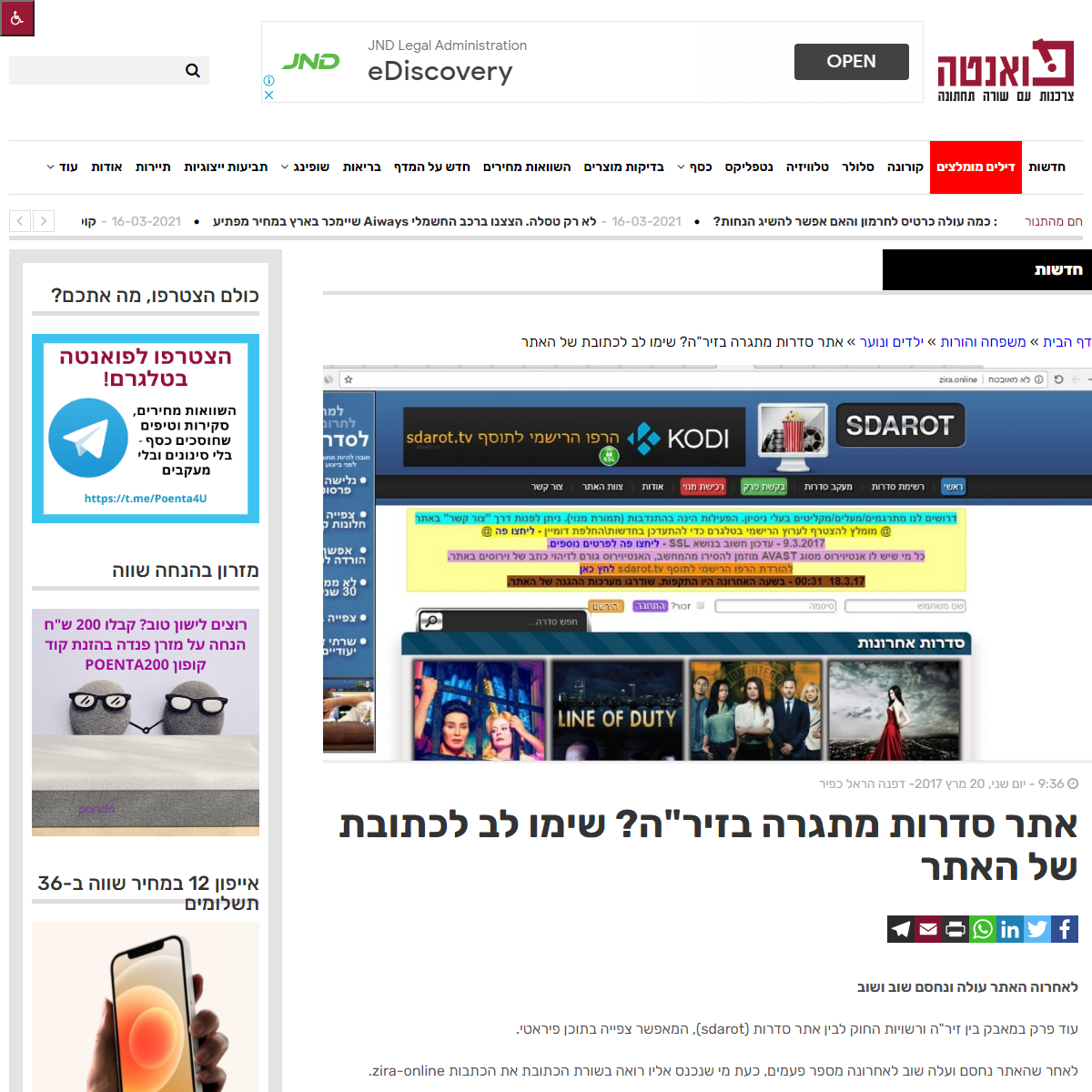
A complete backup of https://www.poenta.co.il/article/sdarot-zira/
Are you over 18 and want to see adult content?
A complete backup of https://kannadakamataana.blogspot.com/2016/06/3.html
Are you over 18 and want to see adult content?

A complete backup of http://www.kurtvip.com/genc-manita-takipcimizden/
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of lasmorfianapoletana.com
Are you over 18 and want to see adult content?
A complete backup of danicasills.storenvy.com
Are you over 18 and want to see adult content?
A complete backup of laindustriainc.com
Are you over 18 and want to see adult content?

A complete backup of redondounion.org
Are you over 18 and want to see adult content?
Text
management
REGULATORY AFFAIRS MANAGEMENT SUITE Regulatory Affairs Management Suite. RAMS adds Mexico Smart Builders and more new features. Experience the new free Global Fee Finder tool, three Mexico Equivalency Route Smart Builders, and IVDR Regulatory Essentials reports. Log in or register. 2:21. HEALTH CANADA REGULATORY APPROVAL PROCESS FOR MEDICAL DEVICES Health Canada will invoice fees over $5000. Step 6. Health Canada reviews MDL application (Class II, III and IV) and Premarket Review Document (Class III and IV only). Step 7. For Class I devices, approved applications will be posted on the Health Canada website and CANADIAN REGULATORY AMENDMENTS INTRODUCE EXPANDED POST Canadian regulatory amendments introduce expanded post-market surveillance provisions. Jan 13, 2021. Health Canada (HC) published SOR/2020-262 in the Canada Gazette, consisting of significant amendments to the Food and Drug Regulations and the Medical Devices Regulations (CMDR). The amendments aim to strengthen post-marketsurveillance for
NEW THAI MEDICAL DEVICE REGULATIONS INCREASE TECHNICAL Thailand's Ministry of Public Health, which includes the Thai Food and Drug Administration (Thai FDA or FDA Thailand), issued new medical device regulations in February that increase technical data submission requirements and application fees. Emergo by UL has details about thenew regulations.
AUSTRALIA WILL DEVELOP UDI SYSTEM FOR MEDICAL DEVICES An amendment to Australia's Therapeutic Goods Act 1989 calls for the creation of a unique device identification (UDI) system for medical devices. Also included among the amendment's provisions is language that could be used to restrict international commerce in therapeutic goods in line with international agreements. Emergo by UL has the details that medical device companies TFDA RELEASES DETAILS ABOUT TRANSITION TO NEW TAIWANESE The Taiwan Food and Drug Administration (TFDA) has published documents explaining aspects of the transition to new medical device regulations, including guidelines for appointment of Technical Personnel, Good Distribution Practice (GDP) licensing requirements, and exemptions from quality management system (QMS) certification. Emergo by UL has the details about how BRAZIL'S MEDICAL DEVICE REGULATORY UPDATE In recent years, ANVISA released numerous regulatory updates to make Brazil a more accessible medical device market. Currently, the medical device industry has more transparency about the requirements for selling in Brazil. Medical device companies can expect several new regulations in the coming years, including new registrationrequirements.
RAMS
We're sorry, but this browser is not supported by RAMS Application. To get the best experience while using this Application, we recommend that you use any of the BRAZILIAN HEALTH SURVEILLANCE AGENCY COLLEGIATE … XXVII - in vitro diagnostic products: reagents, calibrators, patterns, control s, sample collectors, materials and instruments, used individually or in combination, with the intended use determined by the manufacturer, for in vitro analysis of samples derived from the human body, solely or principally to provide information for diagnostic, monitoring or screening purposes or to determine the GLOBAL MEDICAL DEVICE CONSULTING Use regulatory guidance on digital health, wireless compliance, software as medical device (SaMD), and security best practices to succeed with emerging technologies. Assess clinical data requirements for your device and design a clinical strategy that supports your commercialization and product goals. Leverage design review, qualitymanagement
REGULATORY AFFAIRS MANAGEMENT SUITE Regulatory Affairs Management Suite. RAMS adds Mexico Smart Builders and more new features. Experience the new free Global Fee Finder tool, three Mexico Equivalency Route Smart Builders, and IVDR Regulatory Essentials reports. Log in or register. 2:21. HEALTH CANADA REGULATORY APPROVAL PROCESS FOR MEDICAL DEVICES Health Canada will invoice fees over $5000. Step 6. Health Canada reviews MDL application (Class II, III and IV) and Premarket Review Document (Class III and IV only). Step 7. For Class I devices, approved applications will be posted on the Health Canada website and CANADIAN REGULATORY AMENDMENTS INTRODUCE EXPANDED POST Canadian regulatory amendments introduce expanded post-market surveillance provisions. Jan 13, 2021. Health Canada (HC) published SOR/2020-262 in the Canada Gazette, consisting of significant amendments to the Food and Drug Regulations and the Medical Devices Regulations (CMDR). The amendments aim to strengthen post-marketsurveillance for
NEW THAI MEDICAL DEVICE REGULATIONS INCREASE TECHNICAL Thailand's Ministry of Public Health, which includes the Thai Food and Drug Administration (Thai FDA or FDA Thailand), issued new medical device regulations in February that increase technical data submission requirements and application fees. Emergo by UL has details about thenew regulations.
AUSTRALIA WILL DEVELOP UDI SYSTEM FOR MEDICAL DEVICES An amendment to Australia's Therapeutic Goods Act 1989 calls for the creation of a unique device identification (UDI) system for medical devices. Also included among the amendment's provisions is language that could be used to restrict international commerce in therapeutic goods in line with international agreements. Emergo by UL has the details that medical device companies TFDA RELEASES DETAILS ABOUT TRANSITION TO NEW TAIWANESE The Taiwan Food and Drug Administration (TFDA) has published documents explaining aspects of the transition to new medical device regulations, including guidelines for appointment of Technical Personnel, Good Distribution Practice (GDP) licensing requirements, and exemptions from quality management system (QMS) certification. Emergo by UL has the details about how BRAZIL'S MEDICAL DEVICE REGULATORY UPDATE In recent years, ANVISA released numerous regulatory updates to make Brazil a more accessible medical device market. Currently, the medical device industry has more transparency about the requirements for selling in Brazil. Medical device companies can expect several new regulations in the coming years, including new registrationrequirements.
RAMS
We're sorry, but this browser is not supported by RAMS Application. To get the best experience while using this Application, we recommend that you use any of the BRAZILIAN HEALTH SURVEILLANCE AGENCY COLLEGIATE … XXVII - in vitro diagnostic products: reagents, calibrators, patterns, control s, sample collectors, materials and instruments, used individually or in combination, with the intended use determined by the manufacturer, for in vitro analysis of samples derived from the human body, solely or principally to provide information for diagnostic, monitoring or screening purposes or to determine the HEALTH CANADA 2021 MDAP UPDATE INCLUDES COVID-19 Health Canada's Medical Device Action Plan progress report covers efforts to improve device safety and effectiveness, post-market surveillance and monitoring and better public access to medical device information. Learn more at Emergo by UL. KEY DIFFERENCES BETWEEN IVD CLINICAL STUDY REQUIREMENTS Part One of our series on IVD devices examines the differences between performance evaluation (PE) study requirements under the IVDD and clinical performance studies under the IVDR in Europe. Learn more atEmergo by UL.
NEW THAI MEDICAL DEVICE REGULATIONS INCREASE TECHNICAL Thailand's Ministry of Public Health, which includes the Thai Food and Drug Administration (Thai FDA or FDA Thailand), issued new medical device regulations in February that increase technical data submission requirements and application fees. Emergo by UL has details about thenew regulations.
AUSTRALIAN CONFORMITY ASSESSMENT BODY (AU CAB Australia's Therapeutic Goods Administration (TGA) has announced that corporations inside the country may apply starting on July 1, 2021 for Australian Conformity Assessment Body (AU CAB) designation. This initiative is intended to allow for quicker and more flexible approvals of medical devices by expanding the range of entities qualified to issue conformity assessment PUBLIC CONSULTATION OPEN FOR SWISS IVD ORDINANCE The Swiss Federal Office of Public Health (FOPH) announced the opening of a public consultation on the draft In Vitro Diagnostics Ordinance (IvDO) and amendments to the Ordinance on Clinical Trials for Medical Devices (ClinO-MD). These are meant to provide the country's new regulatory framework for IVD products harmonized with the EU IVDR when the latter goes into effect on PREPARING FOR THE EU MDR DATE OF APPLICATION The EU Medical Devices Regulation (MDR) Date of Application (DoA) is imminent. Do you understand what your regulatory obligations will be as of May 26, 2021? This webinar will define the different categories of products covered under MDR and will outline noteworthy requirementsand deadlines.
NMPA PUBLISHES REVISED CHINESE MEDICAL DEVICE REGULATIONS China’s National Medical Products Administration (NMPA) has published (links in Chinese) the revised Regulations on the Supervision and Administration of Medical Devices, which introduces changes to the existing Order No. 650.The updated regulatory framework is slated to take effect on June 1, 2021. When the draft version of this regulatory update was published in June 2018, we reported that US FDA TARGETS ISO 13485 HARMONIZATION, DE NOVO The US Food and Drug Administration plans a final rule for De Novo medical device classification as well as a proposed rule for closer harmonization to ISO 13485:2016 quality management system requirements for later in 2020. Learn more about UK REGULATORS CLARIFY ROLE OF IMPORTERS UNDER MDR, IVDR The United Kingdom has left the European Union, but for Northern Ireland the situation is different. In order to maintain an open border between the Irish Republic and Northern Ireland, the Northern Ireland Protocol arranges for the European product legislation toremain applicable.
BRAZIL ANVISA REGULATORY APPROVAL PROCESS FOR IVDS Manufacturers need to obtain ANVISA ( Agência Nacional de Vigilância Sanitária) approval prior to selling their products in Brazil.Brazil has four classes of IVDs with ascending risk, and multiple registration pathways. Cadastro and Registro.The Cadastro registration route is for lower-risk IVDs, has a simplified application, and typically takes less time than Registro reviews. GLOBAL MEDICAL DEVICE CONSULTING Use regulatory guidance on digital health, wireless compliance, software as medical device (SaMD), and security best practices to succeed with emerging technologies. Assess clinical data requirements for your device and design a clinical strategy that supports your commercialization and product goals. Leverage design review, qualitymanagement
REGULATORY AFFAIRS MANAGEMENT SUITE Regulatory Affairs Management Suite. RAMS adds Mexico Smart Builders and more new features. Experience the new free Global Fee Finder tool, three Mexico Equivalency Route Smart Builders, and IVDR Regulatory Essentials reports. Log in or register. 2:21. HEALTH CANADA REGULATORY APPROVAL PROCESS FOR MEDICAL DEVICES Health Canada will invoice fees over $5000. Step 6. Health Canada reviews MDL application (Class II, III and IV) and Premarket Review Document (Class III and IV only). Step 7. For Class I devices, approved applications will be posted on the Health Canada website and CANADIAN REGULATORY AMENDMENTS INTRODUCE EXPANDED POST Canadian regulatory amendments introduce expanded post-market surveillance provisions. Jan 13, 2021. Health Canada (HC) published SOR/2020-262 in the Canada Gazette, consisting of significant amendments to the Food and Drug Regulations and the Medical Devices Regulations (CMDR). The amendments aim to strengthen post-marketsurveillance for
NEW THAI MEDICAL DEVICE REGULATIONS INCREASE TECHNICAL Thailand's Ministry of Public Health, which includes the Thai Food and Drug Administration (Thai FDA or FDA Thailand), issued new medical device regulations in February that increase technical data submission requirements and application fees. Emergo by UL has details about thenew regulations.
AUSTRALIA WILL DEVELOP UDI SYSTEM FOR MEDICAL DEVICES An amendment to Australia's Therapeutic Goods Act 1989 calls for the creation of a unique device identification (UDI) system for medical devices. Also included among the amendment's provisions is language that could be used to restrict international commerce in therapeutic goods in line with international agreements. Emergo by UL has the details that medical device companies TFDA RELEASES DETAILS ABOUT TRANSITION TO NEW TAIWANESE The Taiwan Food and Drug Administration (TFDA) has published documents explaining aspects of the transition to new medical device regulations, including guidelines for appointment of Technical Personnel, Good Distribution Practice (GDP) licensing requirements, and exemptions from quality management system (QMS) certification. Emergo by UL has the details about how BRAZIL'S MEDICAL DEVICE REGULATORY UPDATE In recent years, ANVISA released numerous regulatory updates to make Brazil a more accessible medical device market. Currently, the medical device industry has more transparency about the requirements for selling in Brazil. Medical device companies can expect several new regulations in the coming years, including new registrationrequirements.
RAMS
We're sorry, but this browser is not supported by RAMS Application. To get the best experience while using this Application, we recommend that you use any of the BRAZILIAN HEALTH SURVEILLANCE AGENCY COLLEGIATE … XXVII - in vitro diagnostic products: reagents, calibrators, patterns, control s, sample collectors, materials and instruments, used individually or in combination, with the intended use determined by the manufacturer, for in vitro analysis of samples derived from the human body, solely or principally to provide information for diagnostic, monitoring or screening purposes or to determine the GLOBAL MEDICAL DEVICE CONSULTING Use regulatory guidance on digital health, wireless compliance, software as medical device (SaMD), and security best practices to succeed with emerging technologies. Assess clinical data requirements for your device and design a clinical strategy that supports your commercialization and product goals. Leverage design review, qualitymanagement
REGULATORY AFFAIRS MANAGEMENT SUITE Regulatory Affairs Management Suite. RAMS adds Mexico Smart Builders and more new features. Experience the new free Global Fee Finder tool, three Mexico Equivalency Route Smart Builders, and IVDR Regulatory Essentials reports. Log in or register. 2:21. HEALTH CANADA REGULATORY APPROVAL PROCESS FOR MEDICAL DEVICES Health Canada will invoice fees over $5000. Step 6. Health Canada reviews MDL application (Class II, III and IV) and Premarket Review Document (Class III and IV only). Step 7. For Class I devices, approved applications will be posted on the Health Canada website and CANADIAN REGULATORY AMENDMENTS INTRODUCE EXPANDED POST Canadian regulatory amendments introduce expanded post-market surveillance provisions. Jan 13, 2021. Health Canada (HC) published SOR/2020-262 in the Canada Gazette, consisting of significant amendments to the Food and Drug Regulations and the Medical Devices Regulations (CMDR). The amendments aim to strengthen post-marketsurveillance for
NEW THAI MEDICAL DEVICE REGULATIONS INCREASE TECHNICAL Thailand's Ministry of Public Health, which includes the Thai Food and Drug Administration (Thai FDA or FDA Thailand), issued new medical device regulations in February that increase technical data submission requirements and application fees. Emergo by UL has details about thenew regulations.
AUSTRALIA WILL DEVELOP UDI SYSTEM FOR MEDICAL DEVICES An amendment to Australia's Therapeutic Goods Act 1989 calls for the creation of a unique device identification (UDI) system for medical devices. Also included among the amendment's provisions is language that could be used to restrict international commerce in therapeutic goods in line with international agreements. Emergo by UL has the details that medical device companies TFDA RELEASES DETAILS ABOUT TRANSITION TO NEW TAIWANESE The Taiwan Food and Drug Administration (TFDA) has published documents explaining aspects of the transition to new medical device regulations, including guidelines for appointment of Technical Personnel, Good Distribution Practice (GDP) licensing requirements, and exemptions from quality management system (QMS) certification. Emergo by UL has the details about how BRAZIL'S MEDICAL DEVICE REGULATORY UPDATE In recent years, ANVISA released numerous regulatory updates to make Brazil a more accessible medical device market. Currently, the medical device industry has more transparency about the requirements for selling in Brazil. Medical device companies can expect several new regulations in the coming years, including new registrationrequirements.
RAMS
We're sorry, but this browser is not supported by RAMS Application. To get the best experience while using this Application, we recommend that you use any of the BRAZILIAN HEALTH SURVEILLANCE AGENCY COLLEGIATE … XXVII - in vitro diagnostic products: reagents, calibrators, patterns, control s, sample collectors, materials and instruments, used individually or in combination, with the intended use determined by the manufacturer, for in vitro analysis of samples derived from the human body, solely or principally to provide information for diagnostic, monitoring or screening purposes or to determine the HEALTH CANADA 2021 MDAP UPDATE INCLUDES COVID-19 Health Canada's Medical Device Action Plan progress report covers efforts to improve device safety and effectiveness, post-market surveillance and monitoring and better public access to medical device information. Learn more at Emergo by UL. KEY DIFFERENCES BETWEEN IVD CLINICAL STUDY REQUIREMENTS Part One of our series on IVD devices examines the differences between performance evaluation (PE) study requirements under the IVDD and clinical performance studies under the IVDR in Europe. Learn more atEmergo by UL.
NEW THAI MEDICAL DEVICE REGULATIONS INCREASE TECHNICAL Thailand's Ministry of Public Health, which includes the Thai Food and Drug Administration (Thai FDA or FDA Thailand), issued new medical device regulations in February that increase technical data submission requirements and application fees. Emergo by UL has details about thenew regulations.
AUSTRALIAN CONFORMITY ASSESSMENT BODY (AU CAB Australia's Therapeutic Goods Administration (TGA) has announced that corporations inside the country may apply starting on July 1, 2021 for Australian Conformity Assessment Body (AU CAB) designation. This initiative is intended to allow for quicker and more flexible approvals of medical devices by expanding the range of entities qualified to issue conformity assessment PUBLIC CONSULTATION OPEN FOR SWISS IVD ORDINANCE The Swiss Federal Office of Public Health (FOPH) announced the opening of a public consultation on the draft In Vitro Diagnostics Ordinance (IvDO) and amendments to the Ordinance on Clinical Trials for Medical Devices (ClinO-MD). These are meant to provide the country's new regulatory framework for IVD products harmonized with the EU IVDR when the latter goes into effect on PREPARING FOR THE EU MDR DATE OF APPLICATION The EU Medical Devices Regulation (MDR) Date of Application (DoA) is imminent. Do you understand what your regulatory obligations will be as of May 26, 2021? This webinar will define the different categories of products covered under MDR and will outline noteworthy requirementsand deadlines.
NMPA PUBLISHES REVISED CHINESE MEDICAL DEVICE REGULATIONS China’s National Medical Products Administration (NMPA) has published (links in Chinese) the revised Regulations on the Supervision and Administration of Medical Devices, which introduces changes to the existing Order No. 650.The updated regulatory framework is slated to take effect on June 1, 2021. When the draft version of this regulatory update was published in June 2018, we reported that US FDA TARGETS ISO 13485 HARMONIZATION, DE NOVO The US Food and Drug Administration plans a final rule for De Novo medical device classification as well as a proposed rule for closer harmonization to ISO 13485:2016 quality management system requirements for later in 2020. Learn more about UK REGULATORS CLARIFY ROLE OF IMPORTERS UNDER MDR, IVDR The United Kingdom has left the European Union, but for Northern Ireland the situation is different. In order to maintain an open border between the Irish Republic and Northern Ireland, the Northern Ireland Protocol arranges for the European product legislation toremain applicable.
BRAZIL ANVISA REGULATORY APPROVAL PROCESS FOR IVDS Manufacturers need to obtain ANVISA ( Agência Nacional de Vigilância Sanitária) approval prior to selling their products in Brazil.Brazil has four classes of IVDs with ascending risk, and multiple registration pathways. Cadastro and Registro.The Cadastro registration route is for lower-risk IVDs, has a simplified application, and typically takes less time than Registro reviews. GLOBAL MEDICAL DEVICE CONSULTING Use regulatory guidance on digital health, wireless compliance, software as medical device (SaMD), and security best practices to succeed with emerging technologies. Assess clinical data requirements for your device and design a clinical strategy that supports your commercialization and product goals. Leverage design review, qualitymanagement
REGULATORY AFFAIRS MANAGEMENT SUITE Regulatory Affairs Management Suite. RAMS adds Mexico Smart Builders and more new features. Experience the new free Global Fee Finder tool, three Mexico Equivalency Route Smart Builders, and IVDR Regulatory Essentials reports. Log in or register. 2:21. NEW THAI MEDICAL DEVICE REGULATIONS INCREASE TECHNICAL Thailand's Ministry of Public Health, which includes the Thai Food and Drug Administration (Thai FDA or FDA Thailand), issued new medical device regulations in February that increase technical data submission requirements and application fees. Emergo by UL has details about thenew regulations.
HEALTH CANADA REGULATORY APPROVAL PROCESS FOR MEDICAL DEVICES Health Canada will invoice fees over $5000. Step 6. Health Canada reviews MDL application (Class II, III and IV) and Premarket Review Document (Class III and IV only). Step 7. For Class I devices, approved applications will be posted on the Health Canada website and NMPA PUBLISHES REVISED CHINESE MEDICAL DEVICE REGULATIONS China’s National Medical Products Administration (NMPA) has published (links in Chinese) the revised Regulations on the Supervision and Administration of Medical Devices, which introduces changes to the existing Order No. 650.The updated regulatory framework is slated to take effect on June 1, 2021. When the draft version of this regulatory update was published in June 2018, we reported that AUSTRALIA WILL DEVELOP UDI SYSTEM FOR MEDICAL DEVICES An amendment to Australia's Therapeutic Goods Act 1989 calls for the creation of a unique device identification (UDI) system for medical devices. Also included among the amendment's provisions is language that could be used to restrict international commerce in therapeutic goods in line with international agreements. Emergo by UL has the details that medical device companies EUROPEAN MDR CLINICAL INVESTIGATION Q&A DOCUMENT FORSEE MORE ONEMERGOBYUL.COM
TFDA RELEASES DETAILS ABOUT TRANSITION TO NEW TAIWANESE The Taiwan Food and Drug Administration (TFDA) has published documents explaining aspects of the transition to new medical device regulations, including guidelines for appointment of Technical Personnel, Good Distribution Practice (GDP) licensing requirements, and exemptions from quality management system (QMS) certification. Emergo by UL has the details about howRAMS
We're sorry, but this browser is not supported by RAMS Application. To get the best experience while using this Application, we recommend that you use any of the BRAZILIAN HEALTH SURVEILLANCE AGENCY COLLEGIATE … XXVII - in vitro diagnostic products: reagents, calibrators, patterns, control s, sample collectors, materials and instruments, used individually or in combination, with the intended use determined by the manufacturer, for in vitro analysis of samples derived from the human body, solely or principally to provide information for diagnostic, monitoring or screening purposes or to determine the GLOBAL MEDICAL DEVICE CONSULTING Use regulatory guidance on digital health, wireless compliance, software as medical device (SaMD), and security best practices to succeed with emerging technologies. Assess clinical data requirements for your device and design a clinical strategy that supports your commercialization and product goals. Leverage design review, qualitymanagement
REGULATORY AFFAIRS MANAGEMENT SUITE Regulatory Affairs Management Suite. RAMS adds Mexico Smart Builders and more new features. Experience the new free Global Fee Finder tool, three Mexico Equivalency Route Smart Builders, and IVDR Regulatory Essentials reports. Log in or register. 2:21. NEW THAI MEDICAL DEVICE REGULATIONS INCREASE TECHNICAL Thailand's Ministry of Public Health, which includes the Thai Food and Drug Administration (Thai FDA or FDA Thailand), issued new medical device regulations in February that increase technical data submission requirements and application fees. Emergo by UL has details about thenew regulations.
HEALTH CANADA REGULATORY APPROVAL PROCESS FOR MEDICAL DEVICES Health Canada will invoice fees over $5000. Step 6. Health Canada reviews MDL application (Class II, III and IV) and Premarket Review Document (Class III and IV only). Step 7. For Class I devices, approved applications will be posted on the Health Canada website and NMPA PUBLISHES REVISED CHINESE MEDICAL DEVICE REGULATIONS China’s National Medical Products Administration (NMPA) has published (links in Chinese) the revised Regulations on the Supervision and Administration of Medical Devices, which introduces changes to the existing Order No. 650.The updated regulatory framework is slated to take effect on June 1, 2021. When the draft version of this regulatory update was published in June 2018, we reported that AUSTRALIA WILL DEVELOP UDI SYSTEM FOR MEDICAL DEVICES An amendment to Australia's Therapeutic Goods Act 1989 calls for the creation of a unique device identification (UDI) system for medical devices. Also included among the amendment's provisions is language that could be used to restrict international commerce in therapeutic goods in line with international agreements. Emergo by UL has the details that medical device companies EUROPEAN MDR CLINICAL INVESTIGATION Q&A DOCUMENT FORSEE MORE ONEMERGOBYUL.COM
TFDA RELEASES DETAILS ABOUT TRANSITION TO NEW TAIWANESE The Taiwan Food and Drug Administration (TFDA) has published documents explaining aspects of the transition to new medical device regulations, including guidelines for appointment of Technical Personnel, Good Distribution Practice (GDP) licensing requirements, and exemptions from quality management system (QMS) certification. Emergo by UL has the details about howRAMS
We're sorry, but this browser is not supported by RAMS Application. To get the best experience while using this Application, we recommend that you use any of the BRAZILIAN HEALTH SURVEILLANCE AGENCY COLLEGIATE … XXVII - in vitro diagnostic products: reagents, calibrators, patterns, control s, sample collectors, materials and instruments, used individually or in combination, with the intended use determined by the manufacturer, for in vitro analysis of samples derived from the human body, solely or principally to provide information for diagnostic, monitoring or screening purposes or to determine the HEALTH CANADA 2021 MDAP UPDATE INCLUDES COVID-19 Health Canada's Medical Device Action Plan progress report covers efforts to improve device safety and effectiveness, post-market surveillance and monitoring and better public access to medical device information. Learn more at Emergo by UL. KEY DIFFERENCES BETWEEN IVD CLINICAL STUDY REQUIREMENTS Part One of our series on IVD devices examines the differences between performance evaluation (PE) study requirements under the IVDD and clinical performance studies under the IVDR in Europe. Learn more atEmergo by UL.
AUSTRALIAN CONFORMITY ASSESSMENT BODY (AU CAB Australia's Therapeutic Goods Administration (TGA) has announced that corporations inside the country may apply starting on July 1, 2021 for Australian Conformity Assessment Body (AU CAB) designation. This initiative is intended to allow for quicker and more flexible approvals of medical devices by expanding the range of entities qualified to issue conformity assessment PREPARING FOR THE EU MDR DATE OF APPLICATION The EU Medical Devices Regulation (MDR) Date of Application (DoA) is imminent. Do you understand what your regulatory obligations will be as of May 26, 2021? This webinar will define the different categories of products covered under MDR and will outline noteworthy requirementsand deadlines.
US FDA TARGETS MULTIPLE CLASS I AND CLASS II MEDICAL Types of devices targeted by the permanent 510 (k) exemption proposal include a wide range of products and systems: home-use ventilators, airway monitoring systems, OTC electrocardiograph software, air purifiers and cleaners, medical device cleaners, N95 respirators and oxygenators. Table 6 of the FDA notice provides a full list of ClassII and
US FDA TARGETS ISO 13485 HARMONIZATION, DE NOVO The US Food and Drug Administration plans a final rule for De Novo medical device classification as well as a proposed rule for closer harmonization to ISO 13485:2016 quality management system requirements for later in 2020. Learn more about TAIWAN FDA SETS MAY 2021 DEADLINE FOR NEW MEDICAL DEVICE The Taiwanese government plans to launch new medical device regulations on May 1, 2021, ushering in new requirements for unique device identification (UDI), distribution and other processes. The Medical Devices Act (link in Chinese) establishes a new medical device regulatory framework in Taiwan via a phased implementation schedule. SWITZERLAND APPROVES MEDDO REVISION AND MOVES TOWARD MDR Switzerland approves MedDO revision and moves toward MDR harmonization. Jul 21, 2020. The Swiss Federal Council has approved a thorough revision of the Medical Devices Ordinance ( MedDO ), along with the new Ordinance on Clinical Trials for Medical Devices ( ClinO-MD ). (The ordinances are abbreviated as MepV and KlinV-Meprespectively in the
UK REGULATORS CLARIFY ROLE OF IMPORTERS UNDER MDR, IVDR The United Kingdom has left the European Union, but for Northern Ireland the situation is different. In order to maintain an open border between the Irish Republic and Northern Ireland, the Northern Ireland Protocol arranges for the European product legislation toremain applicable.
BRAZIL ANVISA REGULATORY APPROVAL PROCESS FOR IVDS Manufacturers need to obtain ANVISA ( Agência Nacional de Vigilância Sanitária) approval prior to selling their products in Brazil.Brazil has four classes of IVDs with ascending risk, and multiple registration pathways. Cadastro and Registro.The Cadastro registration route is for lower-risk IVDs, has a simplified application, and typically takes less time than Registro reviews. Skip to main content__
SEARCH FORM
Search
Close
__
*
* Industry Focus__
INDUSTRY FOCUS
The healthcare industry is changing and we have the breadth of expertise to help you evolve with it. View All* MedTech
* HealthTech
* Pharmaceuticals / BioTech * Healthcare Providers * Health & Wellness Retail We have deep expertise with a range of product types, including combination and borderline products.* Services __
SERVICES
Comprehensive service offerings at every point in the product lifecycle.
* Market Access
* Digital Health & Cybersecurity * Business Process Services * Human Factors Research & Design* Clinical Research
* Digital Applications: RAMS A platform of digital products to improve, simplify and automate RA/QAactivities
LEARN MORE
* Blog__
BLOG
The latest industry news and insights from our global team. View All* Market Access
* Digital Health & Cybersecurity * Business Process Services * Human Factors Research & Design* Clinical Research
FEATURED
*
A PERSONAL TESTAMENT TO THE IMPORTANCE OF MEDICAL TECHNOLOGY Human factors engineer Emilee Stanczyk offers a personal reflection on how wearable device design and...*
CHOOSING AN OPTIMAL FORMATIVE USABILITY TEST SAMPLE SIZE Considerations for ensuring that formative usability tests for medical products include appropriate numbers of subjects.* Resources __
RESOURCES
Resources and tools tailored to medical device professionals. View All* Market Data
* Webinars
* White Papers
* Process Charts
* Videos
* Device Regulations* Tools
MDR RESOURCE CENTER
THE KNOWLEDGE YOU NEED FOR MDR IMPLEMENTATION Europe's Medical Devices Regulation (MDR) goes into effect in May 2020, and we want you to be prepared. Meet our MDR team and get free educational resources on the MDR.* Events__
EVENTS
Learn from our experts through live events. View AllTYPES
* Webinars
* Workshops
* Tradeshows
* Other
RELATED SERVICE
* Market Access
* Digital Health & Cybersecurity * Human Factors Research & DesignUPCOMING
*
BRAZIL'S MEDICAL DEVICE REGULATORY UPDATEJune 24, 2021
Get more details__
* About __
ABOUT
Our global consulting team works from 20+ offices on six continents.View All
* News
* Certifications
*
* Careers
* Locations
US LOCATION
* Emergo by UL Global Headquarters * 2500 Bee Cave Road Building 1, Suite 300Austin Texas 78746
+1 512 222 0262
* ALL LOCATIONS__
* Contact
* Subscribe
* __
SEARCH FORM
Search
Close
__
COVID-19 RESOURCES, INSIGHTS, AND NEWSFEATURED RESOURCE
Get insights from our human factors experts Subscribe to TalkingPoints, a monthly round-up of news, insights, and resources about human factors and usability for medical devices, IVDs, combination products, and more.SUBSCRIBE
FEATURED SERVICE
Updated RAMS features offer EU IVDR support Discover the new PEP and PER Smart Builders, and utilize the Product Classification service for IVDR kits.Learn more
FEATURED SERVICE
Navigate the post-Brexit regulatory environment Emergo by UL can provide UK Responsible Person (UKRP) services to allow your company to distribute medical devices, IVDs, or combination products in the United Kingdom.Learn more
FEATURED RESOURCE
Expert resources to plan your EU IVDR transition The EU IVDR deadline is less than two years away. Visit our IVDR Resource Center for the latest resources, tools, and news from our regulatory and human factors experts.Learn more
FEATURED RESOURCE
Get insights from our human factors experts Subscribe to TalkingPoints, a monthly round-up of news, insights, and resources about human factors and usability for medical devices, IVDs, combination products, and more.SUBSCRIBE
FEATURED SERVICE
Updated RAMS features offer EU IVDR support Discover the new PEP and PER Smart Builders, and utilize the Product Classification service for IVDR kits.Learn more
__
__
OUR SERVICES
Our experienced staff and global network will provide the support youneed.
Home Discovery Human factors research & design Regulatory strategy & gap analysis Software compliance & security Clinical research Risk management Quality management system support Technical file & dossier development In-country representation Post-market complianceDiscovery
We can help you explore unmet market needs, innovate with full consideration of commercial goals, and enhance the user experience.Learn more
Human factors research & design Apply user-centered design principles through user research, adverse event analysis, usability testing, cognitive walkthroughs, staff training sessions, and more.Learn more
Regulatory strategy & gap analysis Plan for rapid global market access with product classification, regulatory pathway selection, technical documentation, and qualitymanagement.
Learn more
Software compliance & security Use regulatory guidance on digital health, wireless compliance, software as medical device (SaMD), and security best practices to succeed with emerging technologies.Learn more
Clinical research
Assess clinical data requirements for your device and design a clinical strategy that supports your commercialization and productgoals.
Learn more
Risk management
Leverage design review, quality management, and clinical data to ensure product safety in line with ISO 14971 and ISO 14155.Learn more
Quality management system support Implement and maintain a streamlined QMS in compliance with ISO 13485:2016 and local standards, with internal and gap audits to MDSAPrequirements.
Learn more
Technical file & dossier development Get expert help compiling technical and design documentation tailored to requirements for European CE marking, US FDA 510(k) clearance, and access to global markets.Learn more
In-country representation Employ local regulatory expertise to secure market access, conduct clinical trials, and maintain control of your medical deviceregistrations.
Learn more
Post-market compliance Maintain your position on market with post-market clinical follow-up (PMCF), root cause analysis, incident reporting, and global vigilancesupport.
Learn more
OUR SERVICES
Our experienced staff and global network will provide the support youneed.
HUMAN FACTORS RESEARCH & DESIGN Helping clients bring products to market and ensuring best-in-classuser experiences.
MARKET ACCESS
A leading consultancy with more than 2,800 medical device and IVDclients worldwide.
CLINICAL RESEARCH
Emergo's CRO team is 100% focused on clinical research for medicaldevices.
DIGITAL HEALTH & CYBERSECURITY Meet regulator expectations and reduce your risk of cyber threats with early cybersecurity assessments.____
PHARMACEUTICALS / BIOTECH The demand for combination products continues to grow. We partner with combination product developers to overcome regulatory challenges as well as design safer, more effective products that help deliver more personalized and precise care.LEARN HOW WE HELP
MDR
RESOURCE CENTER
Europe's Medical Devices Regulation (MDR) goes into effect in May 2021, and we want you to be prepared. Meet our MDR team and get free educational resources on the MDR.START GETTING READY
IVDR
RESOURCE CENTER
Europe’s In Vitro Diagnostic Devices Regulation (IVDR) will bring big changes in May 2022, so it is imperative that you start getting ready. Meet our IVDR experts and find related news and resources.START GETTING READY
LATIN AMERICA
RESOURCE CENTER
Explore the medical device and IVD regulatory systems of Brazil and Mexico, and access growing markets throughout the region. Find out how we can help you expand your global footprint.LEARN MORE
BLOG
The latest industry news and insights from our global team. View All ------------------------- A PERSONAL TESTAMENT TO THE IMPORTANCE OF MEDICAL TECHNOLOGY Human factors engineer Emilee Stanczyk offers a personal reflection on how wearable device design and... CHOOSING AN OPTIMAL FORMATIVE USABILITY TEST SAMPLE SIZE Considerations for ensuring that formative usability tests for medical products include appropriate numbers of subjects. ESTIMATING THE LIKELIHOOD OF A USE ERROR: PART 1 Developing appropriate and accurate tools for estimating likelihood of use errors for medical devices US FDA CONSIDERS UPDATING LABELING REQUIREMENTS FOR MEDICAL IMPLANTS WITH METAL COMPONENTS FDA to revise labeling and product information requirements for metal implants using metal components to...EVENTS
Learn from our experts through live webinars, workshops, and tradeshows. View All -------------------------Webinar
Market Access
BRAZIL'S MEDICAL DEVICE REGULATORY UPDATEThu Jun 24
NEWS
Get updates about Emergo by UL in the field and in the press. View All ------------------------- RAMS PLATFORM UPDATE EXPANDS MEDICAL DEVICE AND IVD REGULATORYTOOLKITS
The newest version of the RAMS SaaS provides new Smart Builder, Regulatory Intelligence and related QA/RA... EMERGO BY UL AND CMTC (CHINA) TO COLLABORATE ON HUMAN FACTORS RESEARCH& DESIGN
Partnership advances human factors engineering and usability of medical devices in China RAMS 2.5 LAUNCHES, EXPANDING AUTOMATED RA/QA CAPABILITIES FOR MEDICAL DEVICE AND IVD COMPANIES RAMS 2.5 offers expanded medical device and IVD market coverage, plus new Smart Builder tools... HAVE QUESTIONS ON WHAT EMERGO CAN DO FOR YOU?CONTACT US
Industry Focus
* MedTech
* HealthTech
* Pharmaceuticals / BioTech * Healthcare Providers * Health & Wellness RetailServices
* Market Access
* Human Factors Research & Design * Digital Health & Cybersecurity* Clinical Research
* Business Process Services * Digital Applications: RAMS* Blog
* Resources
* About
* Office Locations
* Let's connect
* Language
*
English日本語한국어简体中文English
* Subscribe
* Join the RADAR Newsletter * Join the TalkingPoints Newsletter � 2021 EMERGO BY UL. ALL RIGHTS RESERVED. PRIVACY TERMS OF USE DATA SUBJECT ACCESS REQUEST PORTAL Within the UL family of companies we provide a broad portfolio of offerings to all the medical device industries. This includes certification, Notified Body and consultancy services. In order to protect and prevent any conflict of interest, perception of conflict of interest and protection of both our brand and our customers brands, UL is unable to provide consultancy services to Notified Body or MDSAP customers. UL has processes in place to identify and manage any potential conflicts of interest and maintain impartiality. PRIVACY PREFERENCE CENTER*
YOUR PRIVACY
*
STRICTLY NECESSARY COOKIES*
PERFORMANCE COOKIES
*
FUNCTIONAL COOKIES
*
TARGETING COOKIES
YOUR PRIVACY
When you visit any website, it may store or retrieve information on your browser, mostly in the form of cookies. This information might be about you, your preferences or your device and is mostly used to make the site work as you expect it to. The information does not usually directly identify you, but it can give you a more personalized web experience. Because we respect your right to privacy, you can choose not to allow some types of cookies. Click on the different category headings to find out more and change our default settings. However, blocking some types of cookies may impact your experience of the site and the services we are able to offer. Learn More STRICTLY NECESSARY COOKIESAlways Active
These cookies are necessary for the website to function and cannot be switched off in our systems. They are usually only set in response to actions made by you which amount to a request for services, such as setting your privacy preferences, logging in or filling in forms. You can set your browser to block or alert you about these cookies, but some parts of the site will not then work. These cookies do not store any personally identifiable information.PERFORMANCE COOKIES
Performance Cookies
These cookies allow us to count visits and traffic sources so we can measure and improve the performance of our site. They help us to know which pages are the most and least popular and see how visitors move around the site. All information these cookies collect is aggregated and therefore anonymous. If you do not allow these cookies we will not know when you have visited our site, and will not be able to monitor its performance.FUNCTIONAL COOKIES
Functional Cookies
These cookies enable the website to provide enhanced functionality and personalisation. They may be set by us or by third party providers whose services we have added to our pages. If you do not allow these cookies then some or all of these services may not functionproperly.
TARGETING COOKIES
Targeting Cookies
These cookies may be set through our site by our advertising partners. They may be used by those companies to build a profile of your interests and show you relevant adverts on other sites. They do not store directly personal information, but are based on uniquely identifying your browser and internet device. If you do not allow these cookies, you will experience less targeted advertising.BACK BUTTON BACK
Vendor Search
Filter Button
Consent Leg.Interest checkbox label label checkbox label label checkbox label labelClear
checkbox label labelApply Cancel
Confirm My Choices
Allow All
COOKIES ON UL
We use cookies to personalize content and ads, to provide social media features and to analyze our traffic. We also share information about your use of our site with our social media, advertising and analyticspartners.Learn more
Cookies Settings Accept All CookiesDetails
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0