Are you over 18 and want to see adult content?
More Annotations
Sunshine Joy 3D Tapestries and Hippie Merchandise Wholesale and Retail
Are you over 18 and want to see adult content?

ITGalaxy.ro - Magazin electronice si IT. Calculatoare online.
Are you over 18 and want to see adult content?

English Language School Bournemouth | ETC International College
Are you over 18 and want to see adult content?
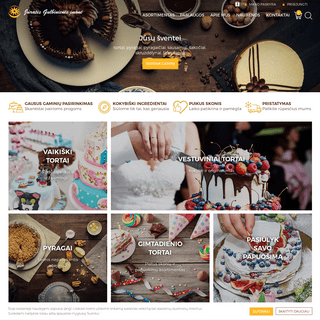
Tortai, pyragai, šakočiai | J. Gulbinienės įmonė
Are you over 18 and want to see adult content?
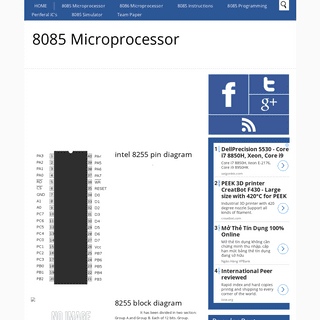
8085 Microprocessor
Are you over 18 and want to see adult content?
تحقیق مقاله پروژه کتاب پایان نامه پیام نور آزاد علمی کاربردی
Are you over 18 and want to see adult content?

MM88BET เว็บแทงบอลออนไลน์ คาสิโนออนไลน์ บาคาร่าออนไลน์ เว็บตรงMM88
Are you over 18 and want to see adult content?

Mix any number of RSS feeds into one unique new feed! | RSS Mix
Are you over 18 and want to see adult content?

IPEYE - Онлайн видеонаблюдение через Интернет
Are you over 18 and want to see adult content?

SERGUR RAYON 116 UNIV. JEMBER – Meningkatkan Guru Profesional yang Bermartabat
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of schlachthof-bremen.de
Are you over 18 and want to see adult content?

A complete backup of espacestemps.net
Are you over 18 and want to see adult content?
A complete backup of themeparks.com.au
Are you over 18 and want to see adult content?

A complete backup of naturalresources.wales
Are you over 18 and want to see adult content?
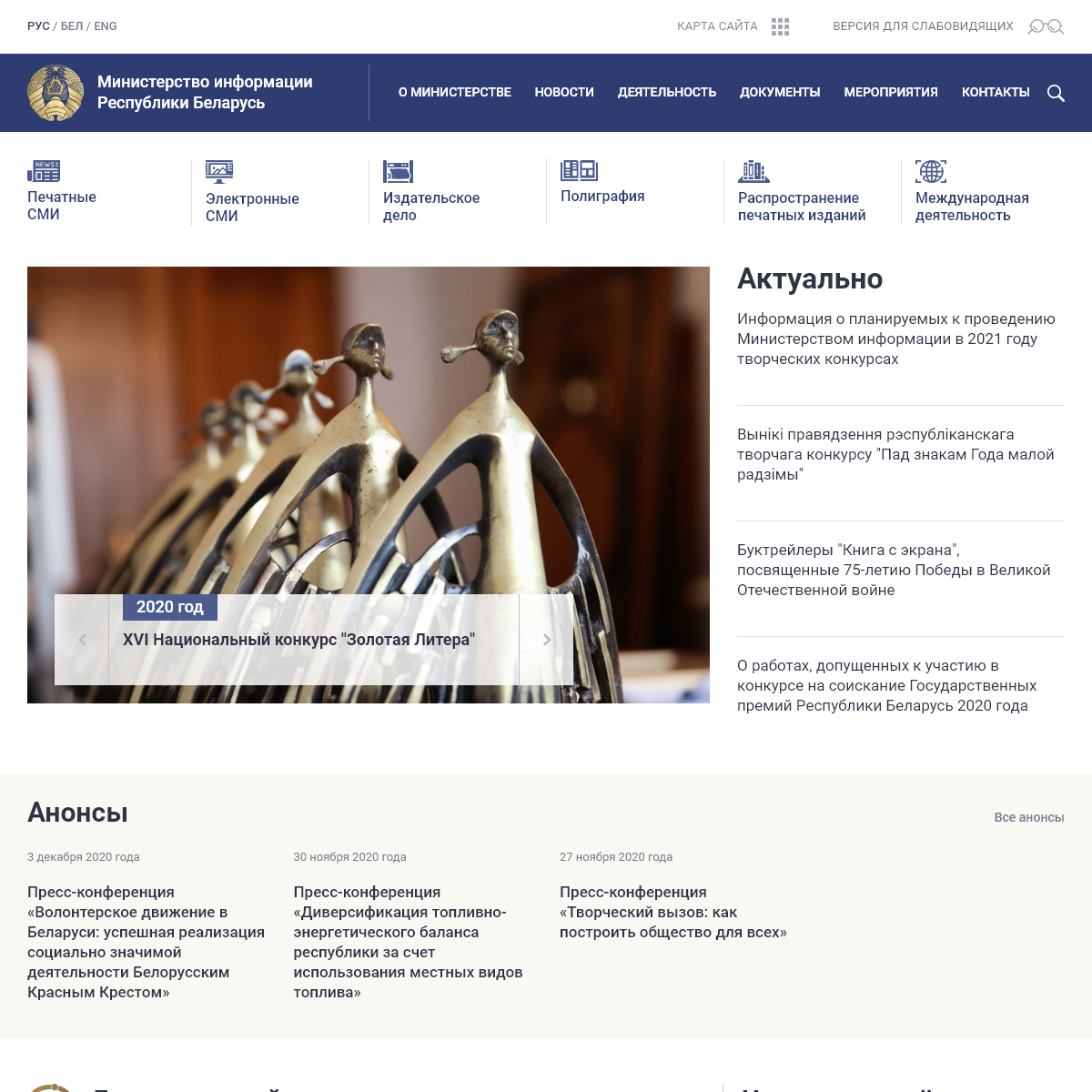
A complete backup of mininform.gov.by
Are you over 18 and want to see adult content?
Text
MONEAD STUDY
MONEAD study participants receive a free iPod touch pre-loaded with a user-friendly, interactive electronic diary that was developed specifically for the MONEAD study. This app provides the participant with a useful tool to keep track of important aspects of their own clinical care including, but not limited to, seizure and medicationinformation.
ADVANTAGE ECLINICAL
Notice: To protect the sensitive data this system handles, your browser connection will be automatically terminated if you remain on the same page longer than 15 minutes without an action that submits the page to the server. Upon termination, all data modifications to that page will be lost.SPLIT - EMMES
Notice: To protect the sensitive data this system handles, your browser connection will be automatically terminated if you remain on the same page longer than 20 minutes without an action that submits the page to the server.Upon termination, all data modifications to that page will be lost. Elapsed time on a page will be indicated by a timer in the browser's status bar. CAROLINAS CORD BLOOD BANK DATA ENTRY SYSTEM Welcome to the Carolinas' Cord Blood Bank. The Carolinas' Cord Blood Bank Data Entry System allows entry of data to the medical coordinating center securely and directly via the World Wide Web. The bank will be able to access and review their data directly.BPCA DATA ENTRY
BPCA Data Entry - Login. AdvantageEDC SM Login Screen. Notice: To protect the sensitive data this system handles, your browser connection will be automatically terminated if you remain on the same page longer than 20 minutes without an action that submits the page to the server. Upon termination, all data modifications to that page willbe lost.
BMTCTN WEBSITE
BMT CTN 1801 Co-Chair: Leslie S. Kean, MD, PhD Leslie.Kean@childrens.harvard.edu. BMT CTN 1801 Co-Chair: Miguel-Angel Perales, MD peralesm@mskcc.org. BMT CTN 1801 Protocol Officer: Wael Saber, MD, MS wsaber@mcw.edu. Protocol Coordinator: Kristy Applegate, MBA kapplegate@emmes.com. The BMT CTN is committed to including widespread transplant COMM-QA-002 FRM4 SERVICE PROVIDER QUALITY AGREEMENT (5DUKE MASTERCONTROLCRID EMMESDUKE BLOOD BANKDUKE NORTH PERIOPDUKE CORD BLOOD BANKDUKE NORTH PAVILION MAP Durham, N ORAQ-MC3 Quality@duke.edu 2400 Pratt Street 9th floor DUMC Box 3850 Rev. 02 Effective Date: 08 Jan 2021 services provided, IScritical.
EMMES IS A FULL SERVICE CLINICAL RESEARCH ORGANIZATION (CRO) Welcome to Emmes, a global full service clinical research organization (CRO) dedicated to helping private sector, government, non-profit and academic partners achieve their biopharmaceutical development and human health goals. Committed to scientific excellence, collaboration, and problem-solving, we work closely with clients to generate high EMMES CAREERS, JOB OPENINGS AND WORKPLACE AWARDS The Emmes Company, LLC is dedicated to hiring an inclusive workforce. If you seek to request a reasonable accommodation during the application process, call Emmes at 301-251-1161 and request to speak with human resources. Our EEO Coordinator looks forward to engaging in an interactive process to provide reasonable accommodations. ANCHOR - LOGIN - EMMES Notice: To protect the sensitive data this system handles, your browser connection will be automatically terminated if you remain on the same page longer than 20 minutes without an action that submits the page to the server.Upon termination, all data modifications to that page will be lost. Elapsed time on a page will be indicated by a timer in the browser's status bar.MONEAD STUDY
MONEAD study participants receive a free iPod touch pre-loaded with a user-friendly, interactive electronic diary that was developed specifically for the MONEAD study. This app provides the participant with a useful tool to keep track of important aspects of their own clinical care including, but not limited to, seizure and medicationinformation.
ADVANTAGE ECLINICAL
Notice: To protect the sensitive data this system handles, your browser connection will be automatically terminated if you remain on the same page longer than 15 minutes without an action that submits the page to the server. Upon termination, all data modifications to that page will be lost.SPLIT - EMMES
Notice: To protect the sensitive data this system handles, your browser connection will be automatically terminated if you remain on the same page longer than 20 minutes without an action that submits the page to the server.Upon termination, all data modifications to that page will be lost. Elapsed time on a page will be indicated by a timer in the browser's status bar. CAROLINAS CORD BLOOD BANK DATA ENTRY SYSTEM Welcome to the Carolinas' Cord Blood Bank. The Carolinas' Cord Blood Bank Data Entry System allows entry of data to the medical coordinating center securely and directly via the World Wide Web. The bank will be able to access and review their data directly.BPCA DATA ENTRY
BPCA Data Entry - Login. AdvantageEDC SM Login Screen. Notice: To protect the sensitive data this system handles, your browser connection will be automatically terminated if you remain on the same page longer than 20 minutes without an action that submits the page to the server. Upon termination, all data modifications to that page willbe lost.
BMTCTN WEBSITE
BMT CTN 1801 Co-Chair: Leslie S. Kean, MD, PhD Leslie.Kean@childrens.harvard.edu. BMT CTN 1801 Co-Chair: Miguel-Angel Perales, MD peralesm@mskcc.org. BMT CTN 1801 Protocol Officer: Wael Saber, MD, MS wsaber@mcw.edu. Protocol Coordinator: Kristy Applegate, MBA kapplegate@emmes.com. The BMT CTN is committed to including widespread transplant COMM-QA-002 FRM4 SERVICE PROVIDER QUALITY AGREEMENT (5DUKE MASTERCONTROLCRID EMMESDUKE BLOOD BANKDUKE NORTH PERIOPDUKE CORD BLOOD BANKDUKE NORTH PAVILION MAP Durham, N ORAQ-MC3 Quality@duke.edu 2400 Pratt Street 9th floor DUMC Box 3850 Rev. 02 Effective Date: 08 Jan 2021 services provided, IScritical.
ABOUT US - EMMES
About Us. Welcome to Emmes, a global full service clinical research organization (CRO) dedicated to supporting the advancement of public health and biopharmaceutical innovation. Founded in 1977, Emmes has conducted over 2000 clinical studies across a broad range of therapeutic areas, with significant expertise in infectious diseases,vaccines
EMMES CULTURE GUIDED BY A QUEST FOR TRUTH, IMPROVING HUMAN Emmes is guided and motivated by an unwavering quest for TRUTH. We believe our longevity and success are a direct result of the honest and transparent relationships we cultivate with organizations like yours. As a global full-service CRO, we appreciate the opportunity to help bring innovative therapies to the world and improve publichealth.
EMMES BIOINFORMATICS, BIOSTATISTICS AND BIG DATA SOLUTIONS Emmes supports your clinical research by providing a collaborative and reproducible approach to large-scale bioinformatics analysis. We help you integrate and analyze data across various high-throughput technologies, including next-generation sequencing (NGS), microarrays, mass spectrometry and others that measure “-omics” outcomes.CONNECT WITH US
We know that successful partnerships begin at the very first interaction with you and are fostered by an undivided focus on your needs. We look forward to talking with you, learning more about your specific goals and determining how we can help you. Headquarters. 401 North Washington Street. Suite 700. Rockville, MD 20850. Phone301.251.1161.
EMMES LEADERSHIP, LEADERS, EMPLOYEES. COLLABORATIVE CULTURE Emmes Leaders. At Emmes, we value a management style that is inclusive, not authoritarian. We set high standards and empower our people to take initiative and follow-through. We respect our employees, who are lifelong learners and make significant contributions to our work and mission. We’re professional, yet encourage an informal and GAMIDA CELL CLINICAL TRIALS DATABASE Notice: To protect the sensitive data this system handles, your browser connection will be automatically terminated if you remain on the same page longer than 20 minutes without an action that submits the page to the server.Upon termination, all data modifications to that page will be lost. Elapsed time on a page will be indicated by a timer in the browser's status bar.BMTCTN WEBSITE
Welcome. The Blood and Marrow Transplant Clinical Trials Network (BMT CTN) was established in October 2001 to conduct large multi-institutional clinical trials. The trials will address important issues in hematopoietic stem cell transplantation (HSCT), thereby furthering understanding of the best possible treatment approaches.BPCA DATA ENTRY
BPCA Data Entry - Login. AdvantageEDC SM Login Screen. Notice: To protect the sensitive data this system handles, your browser connection will be automatically terminated if you remain on the same page longer than 20 minutes without an action that submits the page to the server. Upon termination, all data modifications to that page willbe lost.
DR. ANNE LINDBLAD PROMOTED TO PRESIDENT/CEO OF THE … EMMES is a professional partner to clinicians, scientists, and program leaders around the world. Founded in 1977, the company provides scientific and technical support for clinical research studiesBMTCTN WEBSITE
BMT CTN Protocol 1904 . BMT CTN 1904 - Hematopoietic Cell Transplantation Using Treosulfan-Based Conditioning for the Treatment of Bone Marrow Failure Diseases EMMES IS A FULL SERVICE CLINICAL RESEARCH ORGANIZATION (CRO) Advancing human health through scientific rigor and integrity. Welcome to Emmes, a global full service clinical research organization (CRO) dedicated to helping private sector, government, non-profit and academic partners achieve their biopharmaceutical development andhuman health goals.
EMMES CAREERS, JOB OPENINGS AND WORKPLACE AWARDS Emmes is a CRO offering career opportunities for professional development, outstanding benefits packages, and a flexible, casual, collaborative workplace ANCHOR - LOGIN - EMMES Notice: To protect the sensitive data this system handles, your browser connection will be automatically terminated if you remain on the same page longer than 20 minutes without an action that submits the page to the server.Upon termination, all data modifications to that page will be lost. Elapsed time on a page will be indicated by a timer in the browser's status bar.ADVANTAGE ECLINICAL
Notice: To protect the sensitive data this system handles, your browser connection will be automatically terminated if you remain on the same page longer than 15 minutes without an action that submits the page to the server. Upon termination, all data modifications to that page will be lost.SPLIT - EMMES
Notice: To protect the sensitive data this system handles, your browser connection will be automatically terminated if you remain on the same page longer than 20 minutes without an action that submits the page to the server.Upon termination, all data modifications to that page will be lost. Elapsed time on a page will be indicated by a timer in the browser's status bar.MONEAD STUDY
Maternal Outcomes and Neurodevelopmental Effects of Antiepileptic Drugs (MONEAD) Why is this study important? Most Women with epilepsy have normal pregnancies, but appear to be at risk for problems during pregnancy (e.g., seizures, change in medications, depression, c-sections) and adverse outcomes in their children (e.g., thinking or behavioral problems). CAROLINAS CORD BLOOD BANK DATA ENTRY SYSTEM The Carolinas' Cord Blood Bank Data Entry System allows entry of data to the medical coordinating center securely and directly via the WorldWide Web.
BPCA DATA ENTRY
Notice: To protect the sensitive data this system handles, your browser connection will be automatically terminated if you remain on the same page longer than 20 minutes without an action that submits the page to the server.Upon termination, all data modifications to that page will be lost. Elapsed time on a page will be indicated by a timer in the browser's status bar.BMTCTN WEBSITE
This Project has been funded in whole or in part with Federal funds from the NHLBI, NIH, and NCI under Grant No. U01-HL069294 COMM-QA-002 FRM4 SERVICE PROVIDER QUALITY AGREEMENT (5DUKE MASTERCONTROLCRID EMMESDUKE BLOOD BANKDUKE NORTH PERIOPDUKE CORD BLOOD BANKDUKE NORTH PAVILION MAP Durham, N ORAQ-MC3 Quality@duke.edu 2400 Pratt Street 9th floor DUMC Box 3850 Rev. 02 Effective Date: 08 Jan 2021 services provided, IScritical.
EMMES IS A FULL SERVICE CLINICAL RESEARCH ORGANIZATION (CRO) Advancing human health through scientific rigor and integrity. Welcome to Emmes, a global full service clinical research organization (CRO) dedicated to helping private sector, government, non-profit and academic partners achieve their biopharmaceutical development andhuman health goals.
EMMES CAREERS, JOB OPENINGS AND WORKPLACE AWARDS Emmes is a CRO offering career opportunities for professional development, outstanding benefits packages, and a flexible, casual, collaborative workplace ANCHOR - LOGIN - EMMES Notice: To protect the sensitive data this system handles, your browser connection will be automatically terminated if you remain on the same page longer than 20 minutes without an action that submits the page to the server.Upon termination, all data modifications to that page will be lost. Elapsed time on a page will be indicated by a timer in the browser's status bar.ADVANTAGE ECLINICAL
Notice: To protect the sensitive data this system handles, your browser connection will be automatically terminated if you remain on the same page longer than 15 minutes without an action that submits the page to the server. Upon termination, all data modifications to that page will be lost.SPLIT - EMMES
Notice: To protect the sensitive data this system handles, your browser connection will be automatically terminated if you remain on the same page longer than 20 minutes without an action that submits the page to the server.Upon termination, all data modifications to that page will be lost. Elapsed time on a page will be indicated by a timer in the browser's status bar.MONEAD STUDY
Maternal Outcomes and Neurodevelopmental Effects of Antiepileptic Drugs (MONEAD) Why is this study important? Most Women with epilepsy have normal pregnancies, but appear to be at risk for problems during pregnancy (e.g., seizures, change in medications, depression, c-sections) and adverse outcomes in their children (e.g., thinking or behavioral problems). CAROLINAS CORD BLOOD BANK DATA ENTRY SYSTEM The Carolinas' Cord Blood Bank Data Entry System allows entry of data to the medical coordinating center securely and directly via the WorldWide Web.
BPCA DATA ENTRY
Notice: To protect the sensitive data this system handles, your browser connection will be automatically terminated if you remain on the same page longer than 20 minutes without an action that submits the page to the server.Upon termination, all data modifications to that page will be lost. Elapsed time on a page will be indicated by a timer in the browser's status bar.BMTCTN WEBSITE
This Project has been funded in whole or in part with Federal funds from the NHLBI, NIH, and NCI under Grant No. U01-HL069294 COMM-QA-002 FRM4 SERVICE PROVIDER QUALITY AGREEMENT (5DUKE MASTERCONTROLCRID EMMESDUKE BLOOD BANKDUKE NORTH PERIOPDUKE CORD BLOOD BANKDUKE NORTH PAVILION MAP Durham, N ORAQ-MC3 Quality@duke.edu 2400 Pratt Street 9th floor DUMC Box 3850 Rev. 02 Effective Date: 08 Jan 2021 services provided, IScritical.
ABOUT US - EMMES
Emmes is a CRO with strengths in biostatistics, bioinformatics, clinical trials management, infectious diseases, vaccines, ophthalmology, hematology, rare diseases EMMES CULTURE GUIDED BY A QUEST FOR TRUTH, IMPROVING HUMAN Integrity: We are united in our devotion to our values.We seek decency, truth, honesty, and transparency in our relationships with our colleagues, clients, and collaborators. Agility: We are quick to action, resourceful, innovative, flexible, and adaptable to change.. Passion for Excellence: We seek continuous improvement and high quality in all aspects of our work. EMMES BIOINFORMATICS, BIOSTATISTICS AND BIG DATA SOLUTIONS Emmes supports your clinical research by providing a collaborative and reproducible approach to large-scale bioinformatics analysis. We help you integrate and analyze data across various high-throughput technologies, including next-generation sequencing (NGS), microarrays, mass spectrometry and others that measure “-omics” outcomes. EMMES LEADERSHIP, LEADERS, EMPLOYEES. COLLABORATIVE CULTURE Emmes management are inclusive, empowering, informal, and maintain a friendly environment where employees can be lifelong learners andcollaborative
CONNECT WITH US
We know that successful partnerships begin at the very first interaction with you and are fostered by an undivided focus on your needs. We look forward to talking with you, learning more about your specific goals and determining how we can help you. GAMIDA CELL CLINICAL TRIALS DATABASE Notice: To protect the sensitive data this system handles, your browser connection will be automatically terminated if you remain on the same page longer than 20 minutes without an action that submits the page to the server.Upon termination, all data modifications to that page will be lost. Elapsed time on a page will be indicated by a timer in the browser's status bar.BPCA DATA ENTRY
Notice: To protect the sensitive data this system handles, your browser connection will be automatically terminated if you remain on the same page longer than 20 minutes without an action that submits the page to the server.Upon termination, all data modifications to that page will be lost. Elapsed time on a page will be indicated by a timer in the browser's status bar.BMTCTN WEBSITE
Welcome. The Blood and Marrow Transplant Clinical Trials Network (BMT CTN) was established in October 2001 to conduct large multi-institutional clinical trials. DR. ANNE LINDBLAD PROMOTED TO PRESIDENT/CEO OF THE … EMMES is a professional partner to clinicians, scientists, and program leaders around the world. Founded in 1977, the company provides scientific and technical support for clinical research studiesBMTCTN WEBSITE
BMT CTN Protocol 1904 . BMT CTN 1904 - Hematopoietic Cell Transplantation Using Treosulfan-Based Conditioning for the Treatment of Bone Marrow Failure Diseases YOUR BROWSER DOES NOT SUPPORT JAVASCRIPT! Skip to main contentToggle navigation
* Connect with Us
* News
* Careers
SEARCH FORM
Search
* About
* About us
* Emmes Brand Story
* Our People
* Emmes Leaders
* Our Culture
* Emmes by the Numbers* Clients
* Emmes Locations
* Therapeutic Areas
* Emmes Therapeutic Areas * Vaccines and Infectious Diseases* Ophthalmology
* Hematology
* Maternal and Child Health* Neuroscience
* Rare Diseases
* Services
* Emmes Services Overview * Clinical Research Services* Biostatistics
* Bioinformatics
* Advantage eClinical * Clinical Safety and Pharmacovigilance * Real World Data and Real World Evidence * Experience and Publications* Our Work
* Experience
* Publications
* Clinical Research Spotlight*
*
*
*
*
*
EMMES ACQUIRES ORPHAN REACH FURTHER EXPANDING GLOBAL PRESENCELearn More
WELCOME RHONDA HENRY, PRESIDENT OF EMMES BIOPHARMALearn More
SELECTED AS ONE OF THE BEST PLACES TO WORK IN GREATER WASHINGTON AREALearn More
Q&A WITH EMMES CEO FEATURED IN THE WASHINGTON BUSINESS JOURNALLearn More
EMMES RELEASES AVIAN FLU RESEARCH VIDEOLearn More
EMMES’ CO-AUTHORS SUBSTANCE USE STUDY REPORT IN NEJMLearn More
ADVANCING HUMAN HEALTH THROUGH SCIENTIFIC RIGOR AND INTEGRITY Welcome to Emmes, a global full service clinical research organization (CRO) dedicated to helping private sector, government, non-profit and academic partners achieve their biopharmaceutical development and human health goals. Committed to scientific excellence, collaboration, and problem-solving, we work closely with clients to generate high quality data and make better treatments available to patients.WHO WE ARE
With offices throughout the US, Europe, Canada, and India, Emmes supports the advancement of global public health and biopharmaceutical innovation through disciplined science, rigorous research, fact-based decision-making, and operational excellence. Founded in 1977, we are a diverse, inclusive, innovative CRO with experience managing studies in75+ nations.
Learn More
THERAPEUTIC AREA EXPERTISE From comprehensive support of our clients’ pandemic research to groundbreaking work in ophthalmology, cell & gene therapy and rare diseases, Emmes has collaborated on clinical research across a wide array of disease states. We possess a broad range of therapeutic experience in common and rare diseases, supported by specialized expertise in biostatistics, bioinformatics and e-clinicaltechnologies.
Learn More
EMMES SERVICES
As a strategic collaborator, we believe in expanding the services Emmes offers to best meet the changing needs of our clients. Ongoing investment in global clinical research capabilities, innovative data management platforms, biostatistics/bioinformatics expertise, and our people has enabled Emmes to provide a multi-faceted approach to help you meet your research objectives. Explore Emmes servicesEMMES INSIGHTS
We have been fortunate to collaborate on over 2,000 clinical trials and 2,575+ publications that advance the understanding of medicine andpublic health.
Today’s insight: Getting FDA Fast Track approval for your therapy. Explore Emmes experience and publicationsJOIN THE EMMES TEAM
Are you seeking an opportunity to leverage your background, experience, knowledge and heart to advance clinical research? Do you wish to work for a company whose culture is embodied in the phrase Emmes Cares? We want to meet you. Explore careers at EmmesASK AN ADVISOR
START A CONVERSATION WITH US How can we help you achieve your specific goals? Contact us today to start a dialogue. We look forward to talking with you.Contact Emmes
INVESTIGATORS: PARTICIPATE IN A TRIAL Are you an investigator looking to participate in studies conducted by Emmes? Do you have a passion for advancing existing knowledge of diseases and improving the lives of patients?Contact us today
EMMES BY THE NUMBERS65%
Client Retention Rate1963
Start of our longest-running client engagement800+
Phase I, II, III, and IV clinical trials in 75+ countries Explore Emmes by the NumbersHeadquarters
401 North Washington StreetSuite 700
Rockville, MD 20850
Phone
301.251.1161
* About
* About us
* Emmes Brand Story
* Our People
* Emmes Leaders
* Our Culture
* Emmes by the Numbers* Clients
* Emmes Locations
* Therapeutic Areas
* Emmes Therapeutic Areas * Vaccines and Infectious Diseases* Ophthalmology
* Hematology
* Maternal and Child Health* Neuroscience
* Rare Diseases
* Services
* Emmes Services Overview * Clinical Research Services* Biostatistics
* Bioinformatics
* Advantage eClinical * Clinical Safety and Pharmacovigilance * Real World Data and Real World Evidence * Experience and Publications* Our Work
* Experience
* Publications
* Clinical Research Spotlight Thank you for visiting our site and taking an interest in our products & services. Please feel free to contact us if you wish to receive moreinformation.
For employment-related inquiries, please view our Careers Section. Alternatively, please feel free to contact us via email using theform.
Contact Us
Thank you for your interest in working with Emmes. REASON YOU ARE CONTACTING EMMES - Select -Request Additional Information on ProjectOngoing ProjectsWebsite ProblemsPrivacy StatementOther (Non-employment)Investigator Submission Select the most appropriate reason you are requesting follow-up fromEmmes.
YOUR FULL NAME
Your full name including your credentials if you prefer.YOUR EMAIL ADDRESS
Should be a properly formatted email address where you would like tobe contacted.
COMMENTS
Please provide any additional information that may help us assist with your comments/request.SUBMIT* Cookie Policy
* FCOI
* GDPR
* Privacy Shield Policy * Purchasing Terms and Conditions Copyright © 2021, The Emmes Company, LLC Back to Top YOUR BROWSER DOES NOT SUPPORT JAVASCRIPT!COOKIE POLICY
Our site uses cookies for analytics and to improve our site and services. You can manage these cookies via your browser settings at any time. By continuing to use our site, you agree to our use of cookies. For more information, including how to change your settings, see our Cookie Policy. OK, I agree Cookie PolicyDetails
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0