Are you over 18 and want to see adult content?
More Annotations

A complete backup of https://balkanje.com/turske-serije/gresnik-2014/
Are you over 18 and want to see adult content?

A complete backup of https://balkanje.com/latino-serije/kandidat-2020/
Are you over 18 and want to see adult content?

A complete backup of https://balkanje.com/turske-serije/bella-2017/
Are you over 18 and want to see adult content?
A complete backup of https://balkanje.com/turske-serije/izmedju-nas-2021/
Are you over 18 and want to see adult content?

A complete backup of https://balkanje.com/turske-serije/page/3/
Are you over 18 and want to see adult content?

A complete backup of https://balkanje.com/latino-serije/bela-robinja-2016/
Are you over 18 and want to see adult content?

A complete backup of https://balkanje.com/turske-serije/crna-guja-2008/
Are you over 18 and want to see adult content?

A complete backup of https://balkanje.com/zakletva-epizoda-394/
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of hudvardskompaniet.se
Are you over 18 and want to see adult content?

A complete backup of rochesterjazz.com
Are you over 18 and want to see adult content?
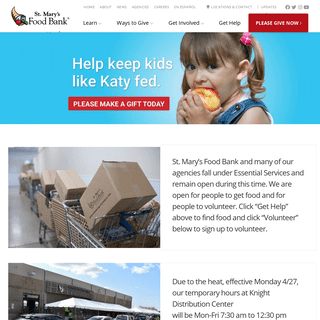
A complete backup of firstfoodbank.org
Are you over 18 and want to see adult content?

A complete backup of nationalinfantrymuseum.org
Are you over 18 and want to see adult content?

A complete backup of onehanesplace.com
Are you over 18 and want to see adult content?
Text
route.
RECALL OF HAND SANITISERS (FEB TO APRIL 2021) Wholesale-level recall of hand sanitisers due to detection of acetaldehyde and/or methanol at levels above the pharmaceutical pharmacopeia limit. Date of Recall: Feb to Apr 2021. Products: Asepso™ Professional Hand Sanitiser (Batch: B42M) Cuticura Hand Sanitizer (all batches) Epi KleenGel Hand Sanitizer Gel (all batches) HSA | COSMETIC PRODUCT NOTIFICATION Submit notification and re-notification Find out how to submit a cosmetic product notification before you sell your cosmetic product in Singapore. Update or cancel notification Find out how to update or cancel a notification after it has already been submitted. Last updated: 25 Sep 2019. HSA | GOOD DISTRIBUTION PRACTICE FOR MEDICAL DEVICES Good Distribution Practice for Medical Devices. Dealers must have a Quality Management System that meets requirements to ensure the safety, quality and performance of medical devices they are dealing in. Importers and wholesalers dealing with medical devices in Singapore should develop a Quality Management System (QMS) thatconforms to the
HSA | RISK CLASSIFICATION OF MEDICAL DEVICESSEE MORE ON HSA.GOV.SG GUIDE TO SUBMISSION OF UPDATE TO CLASS A MEDICAL … GUIDE TO SUBMISSION OF UPDATE TO CLASS A MEDICAL DEVICE EXEMPTION LIST Last update: August 2018 Page 5 of 6 Note 1. The excel file cannot contain any blank cells within the columns B – columns J and shouldnot
HEALTH SCIENCES AUTHORITY (HSA)E-SERVICESREPORT ADVERSE EVENTSBLOOD DONATIONLAB SERVICESWHO WE ARE HSA is the national authority regulating health products; managing the national blood bank, transfusion medicine and forensic medicine expertise; and providing HSA | INFOSEARCH FOR HEALTH PRODUCTS AND SERVICES Illegal health products found in Singapore. If you encounter technical issues for the e-services, e-mail the HSA helpdesk or call 6776 0168 (from 7.00 am to midnight daily) for assistance. Last updated: 19 Apr 2021. Content in this section show. E-services. Infosearch. HSA | MEDICAL DEVICE INFORMATION COMMUNICATION SYSTEM (MEDICS) E-services. Ensure you have the following credentials before you access the MEDICS e-service: CRIS company account. CorpPass. If you encounter technical issues for the e-services, e-mail the HSA helpdesk or call 6776 0168 (from 7.00 am to midnight daily) for assistance. apply@medics. apply@medics. E-services. HSA | SPECIAL ACCESS ROUTES Special Access Routes (SAR) In specific situations, we may allow the import or the supply of therapeutic products via Special Access Routes (SAR). Import and supply of unregistered TP for patient's use Hospitals or clinics need approval from us before importing unregistered therapeutic products for HSA | REGISTRATION OVERVIEW OF MEDICAL DEVICES Class B, C and D registration. Refer to the table below for an overview of the evaluation routes for Class B, C and D medical devices. A medical device without prior approval from any of our overseas reference regulatory agencies must undergo a full evaluation. The following medical devices cannot take the expedited Class C (ECR)route.
RECALL OF HAND SANITISERS (FEB TO APRIL 2021) Wholesale-level recall of hand sanitisers due to detection of acetaldehyde and/or methanol at levels above the pharmaceutical pharmacopeia limit. Date of Recall: Feb to Apr 2021. Products: Asepso™ Professional Hand Sanitiser (Batch: B42M) Cuticura Hand Sanitizer (all batches) Epi KleenGel Hand Sanitizer Gel (all batches)HSA | E-SERVICES
Update changes to your business or company information such as company name, company address, contact details and other related information. If you encounter technical issues for the e-services, e-mail the HSA helpdesk or call 6776 0168 (from 7.00 am to midnight daily) for HSA | GUIDANCE DOCUMENTS FOR THERAPEUTIC PRODUCTS Guidance documents for therapeutic products. Here is the list of guidance documents with relevant forms and templates to help you meet the regulatory requirements for dealing in therapeutic products. 8. Guideline on the Registration of Human Plasma-derived TherapeuticProducts 79 KB. 9.
HSA | GUIDANCE DOCUMENTS FOR CLINICAL TRIALS Clinical trial submissions Clinical trial submissions . Guidance on determination of whether a clinical trial requires a Clinical Trial Authorisation, Clinical Trial Notification or Clinical Trial Certificate 498 KB (updated 1 Mar 2021); Guidance on regulatory requirements for new applications and subsequent submissions 452 KB (updated 28 Apr 2021) HSA GRANTS PROVISIONAL AUTHORISATION FOR ‘BREFENCE … hsa grants provisional authorisation for ‘brefence go covid-19 breath test system’ and ‘traciex breathalyser’ for detection ofcovid-19 infection
SPECIAL ACCESS ROUTE FOR UNAUTHORISED COVID-19 VACCINES This Special Access Route (SAR) applies only to COVID-19 vaccines that are granted Emergency Use List (EUL) by the World Health Organization (WHO) and have not been authorised under the Pandemic Special Access Route (PSAR) by HSA, i.e. non-PSAR authorised vaccines. GUIDANCE ON THE IMPORT OF HAND SANITISERS, MASKS Guidance on the Import of Hand Sanitisers, Masks, Thermometers and Protective Gear. Hand Sanitisers Hand sanitisers do NOT need approval from the Health Sciences Authority (HSA) before they are brought intoSingapore for sale.
COVID-19 ANTIGEN RAPID TESTS (ART) SELF-TEST KITS The Health Sciences Authority (HSA) is committed to ensuring the availability of COVID-19 antigen rapid test (ART) self-test kits that meet the required safety, efficacy and quality standards, for individuals to administer on their own. HSA GRANTS INTERIM AUTHORISATION FOR FOUR COVID-19 ANTIGEN Name of antigen rapid test self-test kit. Name of distributor. Authorisation date. Abbott PanBio TM COVID-19 Antigen Self-Test. Unison Collaborative Pte Ltd. 31 May 2021. Quidel QuickVue At-Home OTCCOVID-19 Test
HSA | IMPORTER, WHOLESALER AND MANUFACTURERS' LICENSING Apply for a licence You need to use MEDICS to apply for a dealer's licence. This includes the manufacturer, importer and wholesaler's licences. Amend a licence You must notify us of changes to your dealer's licence on details such as medical device risk classification, QMS certification and company details. Renew or cancel a licence Renew your dealer's licence on time to continue dealing in HSA UNCOVERS ILLEGAL SURGICAL MASK MANUFACTURING AND HSA Uncovers Illegal Surgical Mask Manufacturing and Repackaging Facility, Consumers Advised to Stop Using Masks from Vision EmpireHealthcare
HSA | E-SERVICES
Update changes to your business or company information such as company name, company address, contact details and other related information. If you encounter technical issues for the e-services, e-mail the HSA helpdesk or call 6776 0168 (from 7.00 am to midnight daily) for HSA | INFOSEARCH FOR HEALTH PRODUCTS AND SERVICES Illegal health products found in Singapore. If you encounter technical issues for the e-services, e-mail the HSA helpdesk or call 6776 0168 (from 7.00 am to midnight daily) for assistance. Last updated: 19 Apr 2021. Content in this section show. E-services. Infosearch. HSA | MEDICAL DEVICE INFORMATION COMMUNICATION SYSTEM (MEDICS) E-services. Ensure you have the following credentials before you access the MEDICS e-service: CRIS company account. CorpPass. If you encounter technical issues for the e-services, e-mail the HSA helpdesk or call 6776 0168 (from 7.00 am to midnight daily) for assistance. apply@medics. apply@medics. E-services. RECALL OF HAND SANITISERS (FEB TO APRIL 2021) Wholesale-level recall of hand sanitisers due to detection of acetaldehyde and/or methanol at levels above the pharmaceutical pharmacopeia limit. Date of Recall: Feb to Apr 2021. Products: Asepso™ Professional Hand Sanitiser (Batch: B42M) Cuticura Hand Sanitizer (all batches) Epi KleenGel Hand Sanitizer Gel (all batches) HSA | GUIDANCE DOCUMENTS FOR CTGTP Guidance documents for CTGTP. Here is the list of guidance documents with relevant forms and templates to help you meet the regulatory requirements for dealing in cell, tissue or gene therapy products. Product Defect Reporting and Recall Procedures for Therapeutic Products and Cells, Tissue and Gene Therapy Products 408 KB. HSA | REGULATORY OVERVIEW OF CELL, TISSUE OR GENE THERAPY Legislation. We regulate therapeutic products in Singapore under the Health Products Act (HPA) and its regulations, including the Health Products (Cell, Tissue and Gene Therapy Products) Regulations 2021. minimally manipulated, i.e. biological characteristics or functions of the cell or the structural properties of the tissue are not altered. HSA | IMPORTER, WHOLESALER AND MANUFACTURERS' LICENSING Apply for a licence You need to use MEDICS to apply for a dealer's licence. This includes the manufacturer, importer and wholesaler's licences. Amend a licence You must notify us of changes to your dealer's licence on details such as medical device risk classification, QMS certification and company details. Renew or cancel a licence Renew your dealer's licence on time to continue dealing in HSA | IMPORT AND SUPPLY OF UNREGISTERED THERAPEUTICSEE MORE ONHSA.GOV.SG
HSA | OVERVIEW OF VARIATION APPLICATION TYPES There are two types of variation applications of existing registered therapeutic products: Major variation application (MAV) Minor variation application (MIV) Application type. Conditions. MAV-1. Any variation to the approved indication, dosing regimen or patient group (s). Inclusion of clinical information extending the usage of theproduct
HSA | GOOD DISTRIBUTION PRACTICE FOR MEDICAL DEVICES Good Distribution Practice for Medical Devices. Dealers must have a Quality Management System that meets requirements to ensure the safety, quality and performance of medical devices they are dealing in. Importers and wholesalers dealing with medical devices in Singapore should develop a Quality Management System (QMS) thatconforms to the
HSA | E-SERVICES
Update changes to your business or company information such as company name, company address, contact details and other related information. If you encounter technical issues for the e-services, e-mail the HSA helpdesk or call 6776 0168 (from 7.00 am to midnight daily) for HSA | INFOSEARCH FOR HEALTH PRODUCTS AND SERVICES Illegal health products found in Singapore. If you encounter technical issues for the e-services, e-mail the HSA helpdesk or call 6776 0168 (from 7.00 am to midnight daily) for assistance. Last updated: 19 Apr 2021. Content in this section show. E-services. Infosearch. HSA | MEDICAL DEVICE INFORMATION COMMUNICATION SYSTEM (MEDICS) E-services. Ensure you have the following credentials before you access the MEDICS e-service: CRIS company account. CorpPass. If you encounter technical issues for the e-services, e-mail the HSA helpdesk or call 6776 0168 (from 7.00 am to midnight daily) for assistance. apply@medics. apply@medics. E-services. RECALL OF HAND SANITISERS (FEB TO APRIL 2021) Wholesale-level recall of hand sanitisers due to detection of acetaldehyde and/or methanol at levels above the pharmaceutical pharmacopeia limit. Date of Recall: Feb to Apr 2021. Products: Asepso™ Professional Hand Sanitiser (Batch: B42M) Cuticura Hand Sanitizer (all batches) Epi KleenGel Hand Sanitizer Gel (all batches) HSA | GUIDANCE DOCUMENTS FOR CTGTP Guidance documents for CTGTP. Here is the list of guidance documents with relevant forms and templates to help you meet the regulatory requirements for dealing in cell, tissue or gene therapy products. Product Defect Reporting and Recall Procedures for Therapeutic Products and Cells, Tissue and Gene Therapy Products 408 KB. HSA | REGULATORY OVERVIEW OF CELL, TISSUE OR GENE THERAPY Legislation. We regulate therapeutic products in Singapore under the Health Products Act (HPA) and its regulations, including the Health Products (Cell, Tissue and Gene Therapy Products) Regulations 2021. minimally manipulated, i.e. biological characteristics or functions of the cell or the structural properties of the tissue are not altered. HSA | IMPORTER, WHOLESALER AND MANUFACTURERS' LICENSING Apply for a licence You need to use MEDICS to apply for a dealer's licence. This includes the manufacturer, importer and wholesaler's licences. Amend a licence You must notify us of changes to your dealer's licence on details such as medical device risk classification, QMS certification and company details. Renew or cancel a licence Renew your dealer's licence on time to continue dealing in HSA | IMPORT AND SUPPLY OF UNREGISTERED THERAPEUTICSEE MORE ONHSA.GOV.SG
HSA | OVERVIEW OF VARIATION APPLICATION TYPES There are two types of variation applications of existing registered therapeutic products: Major variation application (MAV) Minor variation application (MIV) Application type. Conditions. MAV-1. Any variation to the approved indication, dosing regimen or patient group (s). Inclusion of clinical information extending the usage of theproduct
HSA | GOOD DISTRIBUTION PRACTICE FOR MEDICAL DEVICES Good Distribution Practice for Medical Devices. Dealers must have a Quality Management System that meets requirements to ensure the safety, quality and performance of medical devices they are dealing in. Importers and wholesalers dealing with medical devices in Singapore should develop a Quality Management System (QMS) thatconforms to the
HEALTH SCIENCES AUTHORITY (HSA) HSA is the national authority regulating health products; managing the national blood bank, transfusion medicine and forensic medicine expertise; and providingHSA | E-SERVICES
Update changes to your business or company information such as company name, company address, contact details and other related information. If you encounter technical issues for the e-services, e-mail the HSA helpdesk or call 6776 0168 (from 7.00 am to midnight daily) for HSA'S PANDEMIC SPECIAL ACCESS ROUTE Facilitates Singapore's access to critical novel vaccines, medicines and medical devices. The Health Sciences Authority (HSA) has introduced a Pandemic Special Access Route (PSAR) to facilitate early access to critical novel vaccines, medicines and medical devices during a pandemic, such as the current COVID-19 pandemic. HSA REMOVES OVER 700 LISTINGS OF ILLEGAL HEALTH PRODUCTS HSA Removes Over 700 Listings of Illegal Health Products from Local E-Commerce Platforms During Week of Global Intensified Surveillance. The Health Sciences Authority (HSA) has removed more than 3,200 product listings from local e-commerce platforms from January to May2021.
NEW INITIATIVES FOR REGISTRATION OF THERAPEUTIC PRODUCTS New Initiatives for Registration of Therapeutic Products 14 May 2021. Enhanced Dossier Clarification Supplement on Chemistry, Manufacturing & Controls (CMC) for New Drug Applications and Generic DrugApplications
HSA | GUIDANCE DOCUMENTS FOR CTGTP Guidance documents for CTGTP. Here is the list of guidance documents with relevant forms and templates to help you meet the regulatory requirements for dealing in cell, tissue or gene therapy products. Product Defect Reporting and Recall Procedures for Therapeutic Products and Cells, Tissue and Gene Therapy Products 408 KB. HSA | MEDICAL DEVICE REGISTRATION Registration overview Understand the evaluation routes, fees and turn-around-times applicable when you register your medical device. Is it a medical device Tool Check if your device is considered a medical device in Singapore. Risk classification rules and factors Understand the general risk classification system for medical devices, as well as the rules and factors that determine risk class. HSA | IMPORTER, WHOLESALER AND MANUFACTURERS' LICENSING Apply for a licence You need to use MEDICS to apply for a dealer's licence. This includes the manufacturer, importer and wholesaler's licences. Amend a licence You must notify us of changes to your dealer's licence on details such as medical device risk classification, QMS certification and company details. Renew or cancel a licence Renew your dealer's licence on time to continue dealing in NEW POST-APPROVAL INITIATIVES FOR THERAPEUTIC PRODUCTS New post-approval initiatives for therapeutic products. The Therapeutic Products Branch (TPB), Health Products Regulation Group, is pleased to share the following new post-approval initiatives. HSA | GOOD DISTRIBUTION PRACTICE FOR MEDICAL DEVICES Good Distribution Practice for Medical Devices. Dealers must have a Quality Management System that meets requirements to ensure the safety, quality and performance of medical devices they are dealing in. Importers and wholesalers dealing with medical devices in Singapore should develop a Quality Management System (QMS) thatconforms to the
Skip to main content A Singapore Government Agency Website Products regulation Blood donation Lab services Who we are E-services Decrease font size Increase font size Searchhide
×
search
Close
* E-services
* Product Regulations* Medical devices
* Therapeutic products * Health supplements * Traditional medicines * Chinese proprietary medicines* Cosmetic products
* Tobacco regulation* CTGTP
* More Regulations
* Clinical trials
* Controlled drugs and psychotropic substances* Poisons
* Homoeopathic medicines * Medicated oils and balms * Topical antiseptics * International collaboration * Report adverse events* Blood donation
* Lab services
* Who we are
Medical devices
* Regulatory overview * Register your device * Changes to registered devices* Dealer's licence
* Free Sales Certificate and Export Certificate * Report adverse events * Field Safety Corrective Action reporting* Clinical trials
* Consultation schemes * Advertisements and promotions * Bringing personal medical devices into Singapore * Guidance documents * Fees and turnaround time * COVID-19: Standards Resources for Essential Medical Devices* Digital Health
* International collaboration Therapeutic products * Regulatory overview * Register your product * Apply for post-approval variation * Reclassify your product * Dealer's licensing and certification * Report adverse events * Report or recall defective products * Retail pharmacy licensing* Clinical trials
* Product consultation * Advertisements and promotions* CPP and SLS
* International collaboration * Guidance documents * Fees and turnaround timeHealth supplements
* Regulatory overview * Health supplement claims * Advertisements and promotions * Report adverse events * International collaboration Traditional medicines * Regulatory overview * Advertisements and promotions * Report adverse events * Traditional medicinal materials * International collaboration Chinese proprietary medicines * Regulatory overview* Product listing
* Dealer's licensing and certification * Free Sale Certificate * Advertisements and promotions * Report adverse events * Traditional medicinal materials * Fees and Turnaround time of Chinese Proprietary Medicines * International collaborationCosmetic products
* Regulatory overview * Cosmetic product notification * ASEAN Cosmetic Directive * Good Manufacturing Practice certification for cosmetic products * Report adverse events * Fees and turnaround timeTobacco regulation
* Overview of tobacco control* Tobacco licences
* List of suspended and revoked licences * Report tobacco-related offences * Fees and turnaround time * Tobacco retailer educational video and quizCTGTP
* Regulatory overview * Class 1 CTGTP notification * Register a Class 2 CTGTP * Variation applications* Dealer's notice
* Dealer's licensing and certification * Report adverse events * Report or recall defective products* Clinical trials
* Product consultation * Advertisements and promotions * Certificate of a Pharmaceutical Product * Guidance documents * Fees and turnaround time* Register of CTGTP
* Register of CTGTP Licensees Bringing personal medications into Singapore * Regulations for bringing in personal medications * Check medication requirements * Apply for approvalClinical trials
* Regulatory overview * CTA, CTN or CTC submissions * Submit a CRM notification * Good Clinical Practice Inspections * Report adverse events* Innovation Office
* Conducting clinical trials * Participating in clinical trials * Guidance documents * Clinical Trials Register * Clinical trials statistics Controlled drugs and psychotropic substances * Controlled drugs * Psychotropic substances * Restricted substances * Fees and turnaround timePoisons
* Overview of Form A Poisons Licence * Apply for a licence* Amend a licence
* Renew or cancel a licence * Fees and turnaround timeTopical antiseptics
* Regulatory overview * Topical antiseptics classification guide * Guidance for suppliers of hand sanitisers International collaboration * Therapeutic products* Medical devices
* Complementary health products Complementary health products* Access
* ASEAN TMHS
E-services
* Infosearch
* HSA PIN
Consumer safety
* Articles
* Make a report
Why join HSA
* Apply for a job
* Sponsorship for Diploma in Nursing * Employment benefits* Articles
* Forensic Odontology Scholarship Register your device * Registration overview * Is it a medical device Tool * Risk classification rules and factors * Risk classification tool Tool * Registration and licensing requirements Tool * Medical device grouping tool Tool * Grouping medical devices * Registration guides * Priority Review Scheme * Special Access RoutesDealer's licence
* Apply for a licence* Amend a licence
* Renew or cancel a licence * Good Distribution Practice for Medical Devices Field Safety Corrective Action reporting* When to report
* How to report
* Risk management
International collaboration* IMDRF
* AMDC
Register your product * Registration overview * Registration guides for therapeutic products * Summary Reports of Benefit-Risk Assessment * Special Access Routes (SAR) * GMP conformity assessment of overseas manufacturers Apply for post-approval variation * Overview of variation application types * Major variation (MAV-1) application * Major variation (MAV-2) application * Minor variation (MIV) application Reclassify your product * Apply for reclassification * Preparations allowed as General Sale List * List of reclassified medicines Dealer's licensing and certification * Overview of dealer's licences * Apply for a licence* Amend a licence
* Renew or cancel a licence * Good Manufacturing Practice and Good Distribution PracticeStandards
* Certification of local dealers * Handling of Applications and Conduct of Inspections DuringCOVID-19
Retail pharmacy licensing * Overview of retail pharmacy licence * Apply for a retail pharmacy licence * Amend a retail pharmacy licence * Renew or cancel a retail pharmacy licence * Compounding of therapeutic products * Supply GSLs via vending machines * Supply of registered therapeutic products through e-pharmacyClinical trials
* Regulatory overview * New CTA or CTN submission * Subsequent submissions * Good Clinical Practice Inspections * Report adverse events * Product consultation * Conducting clinical trials * Clinical Trials Register * Clinical trials statisticsCPP and SLS
* Certificate of a Pharmaceutical Product * Statement of Licensing Status International collaboration* Access
* ICH
* ICMRA
* NPRA
Product listing
* Apply for new product listing * Amend or cancel product listing * Transfer product listing * Responsibilities of a CPM dealer Dealer's licensing and certification * Overview of dealer's licences * Apply for a Chinese Proprietary Medicine dealer's licence * Amend a Chinese Proprietary Medicine dealer's licence * Renew or cancel a licence * Good Manufacturing Practice and Good Distribution Practice * Certification of local dealers * Import for re-export of Chinese Proprietary Medicines * Handling of Applications and Conduct of Inspections DuringCOVID-19
Cosmetic product notification * Submit notification and re-notification * Update or cancel notificationTobacco licences
* Tobacco retail licence * Tobacco import and wholesale licence Register a Class 2 CTGTP * Registration overview * Registration guides for Class 2 CTGTP * Special Access Routes (SAR) * GMP conformity assessment of overseas manufacturers Variation applications * Overview of variation applications * MAV -1 application * MIV-1 and MIV-2 applications Dealer's licensing and certification * Overview of dealer's licences * Apply for a licence* Amend a licence
* Renew or cancel a licence * Good Manufacturing Practice and Good Distribution PracticeStandards
* Certification of local dealersClinical trials
* Regulatory overview * New CTA or CTN submission * Subsequent submissions * Good Clinical Practice Inspections * Report adverse events * Product consultation * Conducting clinical trials * Clinical Trials Register * Clinical trials statistics CTA, CTN or CTC submissions * New CTA or CTN submission * New CTC submission * Subsequent submissions Conducting clinical trials* Study staff
* Sponsor
* Informed consent * Investigational and auxiliary products * Investigator Site File* Template forms
Controlled drugs
* Apply for a controlled drugs licence * Amend licence to wholesale or manufacture controlled drugs * Renew or cancel a licence Therapeutic products* Access
* ICH
* ICMRA
* NPRA
Medical devices
* IMDRF
* AMDC
Articles
* Details
Articles
* Details
Grouping medical devices * Dental Grouping Terms (DGT)* Hearing aids
* Immunohistochemistry (IHC) IVD reagents * Fluorescence in situ hybridisation (FISH) probes IVD* IVD analysers
* In vitro fertilisation (IVF) media* Family
* System
* Group
* Single
* IVD Test Kit
* IVD cluster
Registration guides
* Class B full registration * Class B abridged registration * Class B immediate registration * Class C full registration * Class C abridged registration * Class C expedited registration * Class C immediate registration * Class D full registration * Class D abridged registration * Class D expedited registration * Class D with a registrable drug in a secondary role fullregistration
* Class D with registrable drug in secondary role abridgedregistration
Special Access Routes * Import for re-export of unregistered devices * Unregistered devices for non-clinical purposes * Registered medical devices on consignment basis * Unregistered devices requested by qualified practitioners * Unregistered devices requested by licensed PHMC * Custom-made medical devices * Import of unregistered medical devices for exhibition * PSAR for Supply of Emergency Medical Devices Registration overview * Registration overview* Application types
* Evaluation Routes
* Screening and evaluation stages * Application dossier * Retain, cancel or transfer registrations Registration guides for therapeutic products * New drug application * Generic drug application * Biosimilar product application * Submission of Drug Master File * Submission of Plasma Master File Special Access Routes (SAR) * Import and supply of unregistered TP for patient's use * Import and supply of registered drug on consignment basis * Special consignment by a product registrant * Pandemic Special Access Route for Supply of Emergency TP Major variation (MAV-1) application * Overview of MAV-1 application * Full evaluation route for MAV-1 application * Abridged evaluation route for MAV-1 application * Verification evaluation route for MAV-1 application Minor variation (MIV) application* Overview
* Submit MIV applications Certification of local dealers * Good Manufacturing Practice certificate for local manufacturers * Good Distribution Practice certificate for local dealers Certification of local dealers * Good Manufacturing Practice certificate for local manufacturers * Good Distribution Practice certificate for local dealers Tobacco retail licence * Apply for a tobacco retail licence * Amend tobacco retail licence * Renew tobacco retail licence * Cancel tobacco retail licence Tobacco import and wholesale licence * Apply for tobacco import and wholesale licence * Amend tobacco import and wholesale licence * Renew tobacco import and wholesale licence * Cancel tobacco import and wholesale licence Registration overview * Registration process * Pre-submission consultation * Application submission and screening * Evaluation and regulatory decision * Retain, cancel or transfer registrations Registration guides for Class 2 CTGTP * Full evaluation route * Abridged evaluation route Special Access Routes (SAR) * Import and supply of unregistered Class 2 CTGTP * Import and supply of CTGTP on consignment basisMAV -1 application
* Full evaluation route * Abridged evaluation route Certification of local dealers * Good Manufacturing Practice certificate for local manufacturers * Good Distribution Practice certificate for local dealersBrowse
Decrease font size Increase font size Search BackNotice
The blood donor carpark is closed. Donors can park at the Health Promotion Board basement carpark. Find out more here.
WELCOME TO THE HEALTH SCIENCES AUTHORITY We regulate health products, serve the administration of justice, secure the nation's blood supply, and safeguard the public's health. HEALTH PRODUCTS REGULATIONSMedical devices
Registration, licensing, change notification, adverse events, FSCA, advertisements, product consultation, digital health Therapeutic products Registration, variations, reclassification, licensing, advertisements, product consultationHealth supplements
Safety and quality standards, claims, contaminants Chinese Proprietary Medicines Product listing, licensing, advertisements Traditional medicines Labelling, ingredients, contaminants, advertisementsCosmetic products
Classification, notification, ASEAN Cosmetic DirectiveTobacco regulation
Licences, suspended and revoked licences, report offences Cell, tissue and gene therapy products Registration, variations, notifications, licensing, advertisement, product consultation ------------------------- Report adverse eventsGo to E-services
Be a responsible blood donor Check your eligibility and donate Bringing personal medication into Singapore Check approval requirementsANNOUNCEMENTS
Press Releases | 25 May 2021 HSA Approves New Storage Condition for the Pfizer-Biontech Covid-19 Vaccine at Refrigerator Temperature... Press Releases | 18 May 2021 HSA Extends the Use of Pfizer-BioNTech COVID-19 Vaccine to Adolescents of Ages 12... Press Releases | 21 Apr 2021 Joint Operation Against Online Sales of Suspected Counterfeit RespiratorsAll announcements
CONSUMER SAFETY
Dubious weight loss products sold online can harm your health Pain relief products purchased from dubious sources lead to serioushealth problems
The dangers of adulterated, counterfeit and substandard healthproducts
Stay cautious, stay safe - 'whitening' and 'anti-aging' products found to contain mercury and prohibited ingredients Go to consumer safetyABOUT HSA
Applied sciences
We serve the administration of justice with the use of forensic medicine, forensic science and analytical chemistry testing.Blood services
We secure the nation's blood supply by ensuring a safe and adequate blood supply for public and private hospitals. Health products regulation We regulate health products to meet standards of safety, quality andefficacy.
Health Sciences AuthorityWho we are
Announcements
Careers
Contact us
Feedback
------------------------- Report VulnerabilityPrivacy Statement
Terms of Use
Sitemap
HSA Data Protection Policy Share your views @ REACH � 2021 Government of Singapore Last Updated 25 May 2021Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0