Are you over 18 and want to see adult content?
More Annotations

A complete backup of ent.ltn.com.tw/news/breakingnews/3079352
Are you over 18 and want to see adult content?
A complete backup of www.indiatvnews.com/entertainment/tv/indian-idol-11-grand-finale-live-update-streaming-aditya-narayan-neha-
Are you over 18 and want to see adult content?

A complete backup of www.nownews.com/news/20200225/3954652/
Are you over 18 and want to see adult content?

A complete backup of www.blick.ch/people-tv/international/jury-einigt-sich-im-vergewaltigungsprozess-auf-urteil-harvey-weinstein
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of everyonedoesit.co.uk
Are you over 18 and want to see adult content?

A complete backup of bambinischoolng.com
Are you over 18 and want to see adult content?

A complete backup of taxsavingislove.com
Are you over 18 and want to see adult content?

A complete backup of royalwolf.com.au
Are you over 18 and want to see adult content?
A complete backup of hannover-rueck.de
Are you over 18 and want to see adult content?
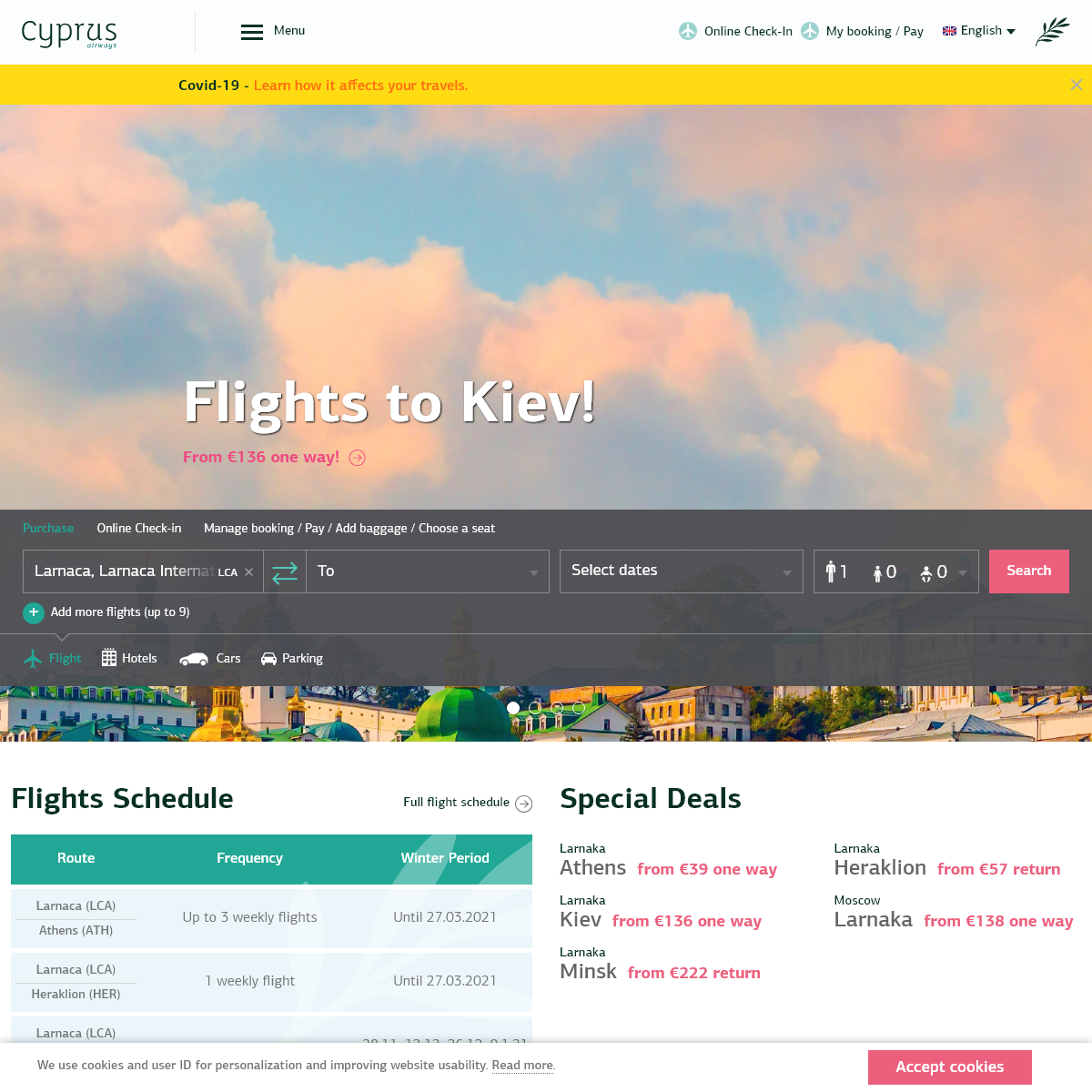
A complete backup of cyprusairways.com
Are you over 18 and want to see adult content?
Text
I-MAB BIOPHARMA
I-Mab to Hold Investor Call to Present In-Depth Phase 1 Clinical Data on Highly Differentiated CD73 Antibody Uliledlimab . I-Mab today announced that it will hold a call with investors on Monday, June 7 at 8:00 a.m. ET to provide an in-depth clinical data analysis of its U.S. phase 1 study of uliledlimab in combination with atezolizumab in patients with advanced cancers.I-MAB BIOPHARMA
I-Mab is a clinical stage biopharmaceutical company committed to the discovery, development and commercialization of first-in-class and best-in-class biologics to treat cancers and autoimmune diseases.I-MAB BIOPHARMA
I-Mab’s competitive pipeline and dynamic business strategy makes it perfectly positioned to capitalize on unique business conditions in China: a growing talent pool of professionals with both global and local R&D expertise; a critical mass of CRO services operating according to global standards; a rapidly improving regulatory and drug-innovation environment; and large capital resources. I-MAB | INVESTOR RELATIONS The Investor Relations website contains information about I-Mab's business for stockholders, potential investors, and financialanalysts.
I-MAB BIOPHARMA
SHANGHAI, China, and GAITHERSBURG, MD – April 26, 2021 --I-Mab (the “Company”) (Nasdaq: IMAB), a clinical-stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, today announced positive topline results from its regional multi-center, randomized, double-blind and placebo-controlled phase 2 study (NCT03235752) I-MAB | I-MAB REPORTS FINANCIAL RESULTS FOR FULL YEAR OF 18 significant clinical milestones achieved since the Company’s IPO in January 2020, with multiple important data readouts for lemzoparlimab (TJC4), uliledlimab (TJD5) and other clinical assets expected in 2021 New drug application (NDA) submission for felzartamab (TJ202) CD38 antibody forI-MAB BIOPHARMA
Jul 26, 2018, I-Mab Biopharma (“I-Mab”), a company focusing on innovative biologics in immuno-oncology and immuno-inflammation, and ABL Bio Corporation (“ABL Bio”), a South Korean biotechnology company, today jointly announced that two companies had entered into a strategic partnering agreement for ABL Bio to in-license the global rights excluding Greater China to I-Mab’s bispecific 天境生物-I-MAB-医药研发-双靶点-独角兽企业-肿瘤自 …TRANSLATE THIS PAGE 天境生物是一家聚焦于肿瘤免疫和自身免疫领域的创新生物药企业,公司高度专注于在全球范围内开发具有“全球首创”和“同类最优”潜力的创新生物药。我们的使命是研制具有突破性的创新生物药,填补临床治疗需求的空白,并且切实提高全球患者的生存质量。I-MAB BIOPHARMA
A first-in-patient study of lemzoparlimab, a differentiated anti-CD47 antibody, in subjects with relapsed/refractory malignancy: initialmonotherapy results
I-MAB
table of contents united states securities and exchange commission washington, d.c. 20549 form 20-f (mark one) ☐ registration statement pursuant to section 12(b) or 12(g) of the securitiesI-MAB BIOPHARMA
I-Mab to Hold Investor Call to Present In-Depth Phase 1 Clinical Data on Highly Differentiated CD73 Antibody Uliledlimab . I-Mab today announced that it will hold a call with investors on Monday, June 7 at 8:00 a.m. ET to provide an in-depth clinical data analysis of its U.S. phase 1 study of uliledlimab in combination with atezolizumab in patients with advanced cancers.I-MAB BIOPHARMA
I-Mab is a clinical stage biopharmaceutical company committed to the discovery, development and commercialization of first-in-class and best-in-class biologics to treat cancers and autoimmune diseases.I-MAB BIOPHARMA
I-Mab’s competitive pipeline and dynamic business strategy makes it perfectly positioned to capitalize on unique business conditions in China: a growing talent pool of professionals with both global and local R&D expertise; a critical mass of CRO services operating according to global standards; a rapidly improving regulatory and drug-innovation environment; and large capital resources. I-MAB | INVESTOR RELATIONS The Investor Relations website contains information about I-Mab's business for stockholders, potential investors, and financialanalysts.
I-MAB BIOPHARMA
SHANGHAI, China, and GAITHERSBURG, MD – April 26, 2021 --I-Mab (the “Company”) (Nasdaq: IMAB), a clinical-stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, today announced positive topline results from its regional multi-center, randomized, double-blind and placebo-controlled phase 2 study (NCT03235752) I-MAB | I-MAB REPORTS FINANCIAL RESULTS FOR FULL YEAR OF 18 significant clinical milestones achieved since the Company’s IPO in January 2020, with multiple important data readouts for lemzoparlimab (TJC4), uliledlimab (TJD5) and other clinical assets expected in 2021 New drug application (NDA) submission for felzartamab (TJ202) CD38 antibody forI-MAB BIOPHARMA
Jul 26, 2018, I-Mab Biopharma (“I-Mab”), a company focusing on innovative biologics in immuno-oncology and immuno-inflammation, and ABL Bio Corporation (“ABL Bio”), a South Korean biotechnology company, today jointly announced that two companies had entered into a strategic partnering agreement for ABL Bio to in-license the global rights excluding Greater China to I-Mab’s bispecific 天境生物-I-MAB-医药研发-双靶点-独角兽企业-肿瘤自 …TRANSLATE THIS PAGE 天境生物是一家聚焦于肿瘤免疫和自身免疫领域的创新生物药企业,公司高度专注于在全球范围内开发具有“全球首创”和“同类最优”潜力的创新生物药。我们的使命是研制具有突破性的创新生物药,填补临床治疗需求的空白,并且切实提高全球患者的生存质量。I-MAB BIOPHARMA
A first-in-patient study of lemzoparlimab, a differentiated anti-CD47 antibody, in subjects with relapsed/refractory malignancy: initialmonotherapy results
I-MAB
table of contents united states securities and exchange commission washington, d.c. 20549 form 20-f (mark one) ☐ registration statement pursuant to section 12(b) or 12(g) of the securitiesI-MAB BIOPHARMA
I-Mab Presents Phase 1 Data on Highly Differentiated CD73 Antibody Uliledlimab at ASCO 2021 . I-Mab today announced that an abstract detailing clinical data from its U.S. phase 1 study of uliledlimab in combination with atezolizumab in patients with advanced cancer will be presented at the 2021 ASCO Annual Meeting, taking place June 4-8,2021.
I-MAB BIOPHARMA
I-Mab is a clinical stage biopharmaceutical company committed to the discovery, development and commercialization of first-in-class and best-in-class biologics to treat cancers and autoimmune diseases.I-MAB BIOPHARMA
SHANGHAI, China, and GAITHERSBURG, MD – April 26, 2021 --I-Mab (the “Company”) (Nasdaq: IMAB), a clinical-stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, today announced positive topline results from its regional multi-center, randomized, double-blind and placebo-controlled phase 2 study (NCT03235752) I-MAB | I-MAB TO HOLD INVESTOR CALL TO PRESENT IN-DEPTH SHANGHAI and GAITHERSBURG, Md., June 2, 2021 /PRNewswire/ -- I-Mab (the "Company") (Nasdaq: IMAB), a clinical stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, today announced that it will hold a call with investors on Monday, June 7 at 8:00 a.m. ET to provide an in-depth clinical data analysis of its U.S. phase 1 study ofI-MAB BIOPHARMA
Jul 26, 2018, I-Mab Biopharma (“I-Mab”), a company focusing on innovative biologics in immuno-oncology and immuno-inflammation, and ABL Bio Corporation (“ABL Bio”), a South Korean biotechnology company, today jointly announced that two companies had entered into a strategic partnering agreement for ABL Bio to in-license the global rights excluding Greater China to I-Mab’s bispecificI-MAB BIOPHARMA
I-Mab Biopharma (“I-Mab”), a China-based clinical stage biopharmaceutical company exclusively focused on the development of innovative biologics in immuno-oncology and autoimmune diseases, announces on Jan. 25, 2019 that its IND application for TJC4, has been approved by the US Food & Drug Administration (FDA). I-MAB | I-MAB ULILEDLIMAB PHASE 1 DATA CONFERENCE CALL I-Mab Uliledlimab Phase 1 Data Conference Call. Jun 7, 2021 at 8:00 AM EDT Meeting ID: 992 9308 1819 Password: 942873 I-MAB | I-MAB PRESENTS PHASE 1 DATA ON HIGHLY Uliledlimab is a highly differentiated CD73 antibody with a unique epitope that confers pharmacological advantages Data from U.S. phase 1 dose escalation trial demonstrates favorable safety and tolerability with no dose-limiting toxicities; preliminary clinical activity of uliledlimab has beenI-MAB | SEC FILING
Exhibit 99.1 . I-Mab Announces Positive Topline Phase 2 Results for Olamkicept in Ulcerative Colitis . SHANGHAI, China, and GAITHERSBURG, MD — April 26, 2021 — I-Mab (the “Company”) (Nasdaq: IMAB), a clinical-stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, today announced positive topline results from its regionalI-MAB BIOPHARMA
A first-in-patient study of lemzoparlimab, a differentiated anti-CD47 antibody, in subjects with relapsed/refractory malignancy: initialmonotherapy results
I-MAB BIOPHARMA
I-Mab to Hold Investor Call to Present In-Depth Phase 1 Clinical Data on Highly Differentiated CD73 Antibody Uliledlimab . I-Mab today announced that it will hold a call with investors on Monday, June 7 at 8:00 a.m. ET to provide an in-depth clinical data analysis of its U.S. phase 1 study of uliledlimab in combination with atezolizumab in patients with advanced cancers. I-MAB | INVESTOR RELATIONS May 27, 2021. SHANGHAI and GAITHERSBURG, Md. , May 27, 2021 /PRNewswire/ -- I-Mab (the "Company") (Nasdaq: IMAB ), a clinical-stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, today announced its participation in the following conferences in June. I-Mab PresentsPhase 1 Data on
I-MAB BIOPHARMA
Our discovery team has deep expertise in functional assays across a wide range of immune cells, including T cells, B cells and macrophages. By designing robust, high-throughput assays, we significantly increase screening efficiency and the probability of finding differentiated molecules. Our discovery team is passionate about, and has excelledI-MAB BIOPHARMA
This is a strong testament to our discovery capability in target biology, antibody engineering, translational medicine and antibody CMC. Bi-specific antibody panel. Despite the recent success of checkpoint inhibitors, it is estimated that more than 60% of cancerpatients
I-MAB BIOPHARMA
These in-licensing deals enable us to acquire multiple novel or highly differentiated clinical-stage assets with favorable clinical safety and early efficacy data. We have quickly built our China Portfolio through in-licensing deals with global biotech partners, including MorphoSys, Genexine, MacroGenics and Ferring, and is the reason we areI-MAB BIOPHARMA
raven.lin@i-mabbiopharma.com. About ABL Bio: ABL Bio is a privately held South Korean biotechnology company developing antibody therapeutics for immuno-oncology and neurodegenerative disease. ABL Bio was founded in 2016 and recently completed series C fund raising of USD 65 million in 2018 after successful series A and B financing in2016 and 2017.
I-MAB | I-MAB AND ABL BIO TO PRESENT PRECLINICAL DATA FOR SHANGHAI and GAITHERSBURG, Md., June 12, 2020 /PRNewswire/ -- I-Mab (the "Company") (Nasdaq: IMAB), a clinical stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, will be presenting preclinical data on a newly developed, novel asset TJ-CD4B, at the American Association for Cancer Research (AACR) Virtual Annual Meeting II on 天境生物-I-MAB-医药研发-双靶点-独角兽企业-肿瘤自 …TRANSLATE THIS PAGE 天境生物是一家聚焦于肿瘤免疫和自身免疫领域的创新生物药企业,公司高度专注于在全球范围内开发具有“全球首创”和“同类最优”潜力的创新生物药。我们的使命是研制具有突破性的创新生物药,填补临床治疗需求的空白,并且切实提高全球患者的生存质量。I-MAB BIOPHARMA
I-Mab Biopharma (“I-Mab”), a China-based clinical stage biopharmaceutical company exclusively focused on the development of innovative biologics in immuno-oncology and autoimmune diseases, announces on Jan. 25, 2019 that its IND application for TJC4, has been approved by the US Food & Drug Administration (FDA).I-MAB BIOPHARMA
A first-in-patient study of lemzoparlimab, a differentiated anti-CD47 antibody, in subjects with relapsed/refractory malignancy: initialmonotherapy results
I-MAB BIOPHARMA
I-Mab to Hold Investor Call to Present In-Depth Phase 1 Clinical Data on Highly Differentiated CD73 Antibody Uliledlimab . I-Mab today announced that it will hold a call with investors on Monday, June 7 at 8:00 a.m. ET to provide an in-depth clinical data analysis of its U.S. phase 1 study of uliledlimab in combination with atezolizumab in patients with advanced cancers. I-MAB | INVESTOR RELATIONS May 27, 2021. SHANGHAI and GAITHERSBURG, Md. , May 27, 2021 /PRNewswire/ -- I-Mab (the "Company") (Nasdaq: IMAB ), a clinical-stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, today announced its participation in the following conferences in June. I-Mab PresentsPhase 1 Data on
I-MAB BIOPHARMA
Our discovery team has deep expertise in functional assays across a wide range of immune cells, including T cells, B cells and macrophages. By designing robust, high-throughput assays, we significantly increase screening efficiency and the probability of finding differentiated molecules. Our discovery team is passionate about, and has excelledI-MAB BIOPHARMA
These in-licensing deals enable us to acquire multiple novel or highly differentiated clinical-stage assets with favorable clinical safety and early efficacy data. We have quickly built our China Portfolio through in-licensing deals with global biotech partners, including MorphoSys, Genexine, MacroGenics and Ferring, and is the reason we areI-MAB BIOPHARMA
I-Mab (Nasdaq: IMAB) is a dynamic, global biotech company exclusively focused on discovery, development and soon commercialization of novel or highly differentiated biologics in the therapeutic areas of immuno-oncology and autoimmune diseases. The Company’s mission is to bring transformational medicines to patients around the world throughI-MAB BIOPHARMA
raven.lin@i-mabbiopharma.com. About ABL Bio: ABL Bio is a privately held South Korean biotechnology company developing antibody therapeutics for immuno-oncology and neurodegenerative disease. ABL Bio was founded in 2016 and recently completed series C fund raising of USD 65 million in 2018 after successful series A and B financing in2016 and 2017.
I-MAB | I-MAB AND ABL BIO TO PRESENT PRECLINICAL DATA FOR SHANGHAI and GAITHERSBURG, Md., June 12, 2020 /PRNewswire/ -- I-Mab (the "Company") (Nasdaq: IMAB), a clinical stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, will be presenting preclinical data on a newly developed, novel asset TJ-CD4B, at the American Association for Cancer Research (AACR) Virtual Annual Meeting II onI-MAB BIOPHARMA
I-Mab Biopharma (“I-Mab”), a China-based clinical stage biopharmaceutical company exclusively focused on the development of innovative biologics in immuno-oncology and autoimmune diseases, announces on Jan. 25, 2019 that its IND application for TJC4, has been approved by the US Food & Drug Administration (FDA). 天境生物-I-MAB-医药研发-双靶点-独角兽企业-肿瘤自 …TRANSLATE THIS PAGE 天境生物是一家聚焦于肿瘤免疫和自身免疫领域的创新生物药企业,公司高度专注于在全球范围内开发具有“全球首创”和“同类最优”潜力的创新生物药。我们的使命是研制具有突破性的创新生物药,填补临床治疗需求的空白,并且切实提高全球患者的生存质量。I-MAB BIOPHARMA
A first-in-patient study of lemzoparlimab, a differentiated anti-CD47 antibody, in subjects with relapsed/refractory malignancy: initialmonotherapy results
I-MAB BIOPHARMA
These in-licensing deals enable us to acquire multiple novel or highly differentiated clinical-stage assets with favorable clinical safety and early efficacy data. We have quickly built our China Portfolio through in-licensing deals with global biotech partners, including MorphoSys, Genexine, MacroGenics and Ferring, and is the reason we areI-MAB BIOPHARMA
This is a strong testament to our discovery capability in target biology, antibody engineering, translational medicine and antibody CMC. Bi-specific antibody panel. Despite the recent success of checkpoint inhibitors, it is estimated that more than 60% of cancerpatients
I-MAB BIOPHARMA
I-Mab (Nasdaq: IMAB) is an innovation-driven global biotech company focusing on discovery, development and soon commercialization of novel and highly differentiated biologics in immuno-oncology therapeutic area. The Company's mission is to bring transformational medicines to patients around the world through drug innovation.I-MAB BIOPHARMA
I-Mab (Nasdaq: IMAB) is a dynamic, global biotech company exclusively focused on discovery, development and soon commercialization of novel or highly differentiated biologics in the therapeutic areas of immuno-oncology and autoimmune diseases. The Company’s mission is to bring transformational medicines to patients around the world throughI-MAB BIOPHARMA
raven.lin@i-mabbiopharma.com. About ABL Bio: ABL Bio is a privately held South Korean biotechnology company developing antibody therapeutics for immuno-oncology and neurodegenerative disease. ABL Bio was founded in 2016 and recently completed series C fund raising of USD 65 million in 2018 after successful series A and B financing in2016 and 2017.
I-MAB | ANNUAL REPORTS Apr 28, 2021. Annual Report 2020 3.4 MB. Apr 29, 2020 I-MAB | I-MAB TO HOST 2021 R&D DAY Mandarin session on April 7, 2021 and English session on April 26, 2021 for investors and research analysts. SHANGHAI, China and GAITHERSBURG, Md., March 24, 2021 (GLOBE NEWSWIRE) -- I-Mab (the “Company”) (Nasdaq: IMAB), a clinical stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, today announced that it will host its I-MAB | I-MAB ULILEDLIMAB PHASE 1 DATA CONFERENCE CALL I-Mab Uliledlimab Phase 1 Data Conference Call. Jun 7, 2021 at 8:00 AM EDT Meeting ID: 992 9308 1819 Password: 942873 I-MAB | ABBVIE AND I-MAB ENTER INTO GLOBAL STRATEGIC NORTH CHICAGO, Illinois and SHANGHAI, Sept. 4, 2020 /PRNewswire/ -- AbbVie (NYSE: ABBV) and I-Mab (Nasdaq: IMAB) announced today that AbbVie and I-Mab have signed a broad, global collaboration agreement for the development and commercialization of lemzoparlimab (also known as TJC4), an innovative anti-CD47 monoclonal antibody internally discovered and developed by I-Mab forI-MAB BIOPHARMA
“I-Mab is very excited about our strategic partnership with such a strong and integrated pharmaceutical group like CSPC pharma, During the negotiation, we were highly impressed by CSPC’s professional and innovative team.” said Zheru Zhang, Ph.D., President of I-Mab “I-Mab is a company dedicated to providing patients with innovative biological treatments among which TG103 is strikinglyI-MAB BIOPHARMA
I-Mab to Hold Investor Call to Present In-Depth Phase 1 Clinical Data on Highly Differentiated CD73 Antibody Uliledlimab . I-Mab today announced that it will hold a call with investors on Monday, June 7 at 8:00 a.m. ET to provide an in-depth clinical data analysis of its U.S. phase 1 study of uliledlimab in combination with atezolizumab in patients with advanced cancers. I-MAB | INVESTOR RELATIONS May 27, 2021. SHANGHAI and GAITHERSBURG, Md. , May 27, 2021 /PRNewswire/ -- I-Mab (the "Company") (Nasdaq: IMAB ), a clinical-stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, today announced its participation in the following conferences in June. I-Mab PresentsPhase 1 Data on
I-MAB BIOPHARMA
Our discovery team has deep expertise in functional assays across a wide range of immune cells, including T cells, B cells and macrophages. By designing robust, high-throughput assays, we significantly increase screening efficiency and the probability of finding differentiated molecules. Our discovery team is passionate about, and has excelledI-MAB BIOPHARMA
This is a strong testament to our discovery capability in target biology, antibody engineering, translational medicine and antibody CMC. Bi-specific antibody panel. Despite the recent success of checkpoint inhibitors, it is estimated that more than 60% of cancerpatients
I-MAB BIOPHARMA
These in-licensing deals enable us to acquire multiple novel or highly differentiated clinical-stage assets with favorable clinical safety and early efficacy data. We have quickly built our China Portfolio through in-licensing deals with global biotech partners, including MorphoSys, Genexine, MacroGenics and Ferring, and is the reason we areI-MAB BIOPHARMA
raven.lin@i-mabbiopharma.com. About ABL Bio: ABL Bio is a privately held South Korean biotechnology company developing antibody therapeutics for immuno-oncology and neurodegenerative disease. ABL Bio was founded in 2016 and recently completed series C fund raising of USD 65 million in 2018 after successful series A and B financing in2016 and 2017.
I-MAB | I-MAB AND ABL BIO TO PRESENT PRECLINICAL DATA FOR SHANGHAI and GAITHERSBURG, Md., June 12, 2020 /PRNewswire/ -- I-Mab (the "Company") (Nasdaq: IMAB), a clinical stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, will be presenting preclinical data on a newly developed, novel asset TJ-CD4B, at the American Association for Cancer Research (AACR) Virtual Annual Meeting II on 天境生物-I-MAB-医药研发-双靶点-独角兽企业-肿瘤自 …TRANSLATE THIS PAGE 天境生物是一家聚焦于肿瘤免疫和自身免疫领域的创新生物药企业,公司高度专注于在全球范围内开发具有“全球首创”和“同类最优”潜力的创新生物药。我们的使命是研制具有突破性的创新生物药,填补临床治疗需求的空白,并且切实提高全球患者的生存质量。I-MAB BIOPHARMA
I-Mab Biopharma (“I-Mab”), a China-based clinical stage biopharmaceutical company exclusively focused on the development of innovative biologics in immuno-oncology and autoimmune diseases, announces on Jan. 25, 2019 that its IND application for TJC4, has been approved by the US Food & Drug Administration (FDA).I-MAB BIOPHARMA
A first-in-patient study of lemzoparlimab, a differentiated anti-CD47 antibody, in subjects with relapsed/refractory malignancy: initialmonotherapy results
I-MAB BIOPHARMA
I-Mab to Hold Investor Call to Present In-Depth Phase 1 Clinical Data on Highly Differentiated CD73 Antibody Uliledlimab . I-Mab today announced that it will hold a call with investors on Monday, June 7 at 8:00 a.m. ET to provide an in-depth clinical data analysis of its U.S. phase 1 study of uliledlimab in combination with atezolizumab in patients with advanced cancers. I-MAB | INVESTOR RELATIONS May 27, 2021. SHANGHAI and GAITHERSBURG, Md. , May 27, 2021 /PRNewswire/ -- I-Mab (the "Company") (Nasdaq: IMAB ), a clinical-stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, today announced its participation in the following conferences in June. I-Mab PresentsPhase 1 Data on
I-MAB BIOPHARMA
Our discovery team has deep expertise in functional assays across a wide range of immune cells, including T cells, B cells and macrophages. By designing robust, high-throughput assays, we significantly increase screening efficiency and the probability of finding differentiated molecules. Our discovery team is passionate about, and has excelledI-MAB BIOPHARMA
This is a strong testament to our discovery capability in target biology, antibody engineering, translational medicine and antibody CMC. Bi-specific antibody panel. Despite the recent success of checkpoint inhibitors, it is estimated that more than 60% of cancerpatients
I-MAB BIOPHARMA
These in-licensing deals enable us to acquire multiple novel or highly differentiated clinical-stage assets with favorable clinical safety and early efficacy data. We have quickly built our China Portfolio through in-licensing deals with global biotech partners, including MorphoSys, Genexine, MacroGenics and Ferring, and is the reason we areI-MAB BIOPHARMA
raven.lin@i-mabbiopharma.com. About ABL Bio: ABL Bio is a privately held South Korean biotechnology company developing antibody therapeutics for immuno-oncology and neurodegenerative disease. ABL Bio was founded in 2016 and recently completed series C fund raising of USD 65 million in 2018 after successful series A and B financing in2016 and 2017.
I-MAB | I-MAB AND ABL BIO TO PRESENT PRECLINICAL DATA FOR SHANGHAI and GAITHERSBURG, Md., June 12, 2020 /PRNewswire/ -- I-Mab (the "Company") (Nasdaq: IMAB), a clinical stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, will be presenting preclinical data on a newly developed, novel asset TJ-CD4B, at the American Association for Cancer Research (AACR) Virtual Annual Meeting II on 天境生物-I-MAB-医药研发-双靶点-独角兽企业-肿瘤自 …TRANSLATE THIS PAGE 天境生物是一家聚焦于肿瘤免疫和自身免疫领域的创新生物药企业,公司高度专注于在全球范围内开发具有“全球首创”和“同类最优”潜力的创新生物药。我们的使命是研制具有突破性的创新生物药,填补临床治疗需求的空白,并且切实提高全球患者的生存质量。I-MAB BIOPHARMA
I-Mab Biopharma (“I-Mab”), a China-based clinical stage biopharmaceutical company exclusively focused on the development of innovative biologics in immuno-oncology and autoimmune diseases, announces on Jan. 25, 2019 that its IND application for TJC4, has been approved by the US Food & Drug Administration (FDA).I-MAB BIOPHARMA
A first-in-patient study of lemzoparlimab, a differentiated anti-CD47 antibody, in subjects with relapsed/refractory malignancy: initialmonotherapy results
I-MAB BIOPHARMA
This is a strong testament to our discovery capability in target biology, antibody engineering, translational medicine and antibody CMC. Bi-specific antibody panel. Despite the recent success of checkpoint inhibitors, it is estimated that more than 60% of cancerpatients
I-MAB BIOPHARMA
These in-licensing deals enable us to acquire multiple novel or highly differentiated clinical-stage assets with favorable clinical safety and early efficacy data. We have quickly built our China Portfolio through in-licensing deals with global biotech partners, including MorphoSys, Genexine, MacroGenics and Ferring, and is the reason we areI-MAB BIOPHARMA
I-Mab (Nasdaq: IMAB) is an innovation-driven global biotech company focusing on discovery, development and soon commercialization of novel and highly differentiated biologics in immuno-oncology therapeutic area. The Company's mission is to bring transformational medicines to patients around the world through drug innovation.I-MAB BIOPHARMA
I-Mab (Nasdaq: IMAB) is a dynamic, global biotech company exclusively focused on discovery, development and soon commercialization of novel or highly differentiated biologics in the therapeutic areas of immuno-oncology and autoimmune diseases. The Company’s mission is to bring transformational medicines to patients around the world throughI-MAB BIOPHARMA
raven.lin@i-mabbiopharma.com. About ABL Bio: ABL Bio is a privately held South Korean biotechnology company developing antibody therapeutics for immuno-oncology and neurodegenerative disease. ABL Bio was founded in 2016 and recently completed series C fund raising of USD 65 million in 2018 after successful series A and B financing in2016 and 2017.
I-MAB | I-MAB TO HOST 2021 R&D DAY Mandarin session on April 7, 2021 and English session on April 26, 2021 for investors and research analysts. SHANGHAI, China and GAITHERSBURG, Md., March 24, 2021 (GLOBE NEWSWIRE) -- I-Mab (the “Company”) (Nasdaq: IMAB), a clinical stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, today announced that it will host its I-MAB | I-MAB ULILEDLIMAB PHASE 1 DATA CONFERENCE CALL I-Mab Uliledlimab Phase 1 Data Conference Call. Jun 7, 2021 at 8:00 AM EDT Meeting ID: 992 9308 1819 Password: 942873 I-MAB | ABBVIE AND I-MAB ENTER INTO GLOBAL STRATEGIC NORTH CHICAGO, Illinois and SHANGHAI, Sept. 4, 2020 /PRNewswire/ -- AbbVie (NYSE: ABBV) and I-Mab (Nasdaq: IMAB) announced today that AbbVie and I-Mab have signed a broad, global collaboration agreement for the development and commercialization of lemzoparlimab (also known as TJC4), an innovative anti-CD47 monoclonal antibody internally discovered and developed by I-Mab for I-MAB APPOINTS RUYI HE AND RONG SHAO TO BOARD OF DIRECTORS I-Mab Appoints Ruyi He and Rong Shao to Board of Directors June 1, 2021 SHANGHAI and GAITHERSBURG, Md. , June 1, 2021 /PRNewswire/ -- I-Mab (the "Company") (Nasdaq: IMAB ), a clinical stagebiopharmaceutical
I-MAB | I-MAB PRESENTS PHASE 1 DATA ON HIGHLY Uliledlimab is a highly differentiated CD73 antibody with a unique epitope that confers pharmacological advantages; Data from U.S. phase 1 dose escalation trial demonstrates favorable safety and tolerability with no dose-limiting toxicities; preliminary clinical activity of uliledlimab has been observed in patients with advanced cancers; ASCO abstract among Top 12 selected for posterI-MAB BIOPHARMA
I-Mab to Hold Investor Call to Present In-Depth Phase 1 Clinical Data on Highly Differentiated CD73 Antibody Uliledlimab . I-Mab today announced that it will hold a call with investors on Monday, June 7 at 8:00 a.m. ET to provide an in-depth clinical data analysis of its U.S. phase 1 study of uliledlimab in combination with atezolizumab in patients with advanced cancers. I-MAB | INVESTOR RELATIONS May 27, 2021. SHANGHAI and GAITHERSBURG, Md. , May 27, 2021 /PRNewswire/ -- I-Mab (the "Company") (Nasdaq: IMAB ), a clinical-stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, today announced its participation in the following conferences in June. I-Mab PresentsPhase 1 Data on
I-MAB BIOPHARMA
Our discovery team has deep expertise in functional assays across a wide range of immune cells, including T cells, B cells and macrophages. By designing robust, high-throughput assays, we significantly increase screening efficiency and the probability of finding differentiated molecules. Our discovery team is passionate about, and has excelledI-MAB BIOPHARMA
This is a strong testament to our discovery capability in target biology, antibody engineering, translational medicine and antibody CMC. Bi-specific antibody panel. Despite the recent success of checkpoint inhibitors, it is estimated that more than 60% of cancerpatients
I-MAB BIOPHARMA
These in-licensing deals enable us to acquire multiple novel or highly differentiated clinical-stage assets with favorable clinical safety and early efficacy data. We have quickly built our China Portfolio through in-licensing deals with global biotech partners, including MorphoSys, Genexine, MacroGenics and Ferring, and is the reason we areI-MAB BIOPHARMA
raven.lin@i-mabbiopharma.com. About ABL Bio: ABL Bio is a privately held South Korean biotechnology company developing antibody therapeutics for immuno-oncology and neurodegenerative disease. ABL Bio was founded in 2016 and recently completed series C fund raising of USD 65 million in 2018 after successful series A and B financing in2016 and 2017.
I-MAB | I-MAB AND ABL BIO TO PRESENT PRECLINICAL DATA FOR SHANGHAI and GAITHERSBURG, Md., June 12, 2020 /PRNewswire/ -- I-Mab (the "Company") (Nasdaq: IMAB), a clinical stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, will be presenting preclinical data on a newly developed, novel asset TJ-CD4B, at the American Association for Cancer Research (AACR) Virtual Annual Meeting II on 天境生物-I-MAB-医药研发-双靶点-独角兽企业-肿瘤自 …TRANSLATE THIS PAGE 天境生物是一家聚焦于肿瘤免疫和自身免疫领域的创新生物药企业,公司高度专注于在全球范围内开发具有“全球首创”和“同类最优”潜力的创新生物药。我们的使命是研制具有突破性的创新生物药,填补临床治疗需求的空白,并且切实提高全球患者的生存质量。I-MAB BIOPHARMA
I-Mab Biopharma (“I-Mab”), a China-based clinical stage biopharmaceutical company exclusively focused on the development of innovative biologics in immuno-oncology and autoimmune diseases, announces on Jan. 25, 2019 that its IND application for TJC4, has been approved by the US Food & Drug Administration (FDA).I-MAB BIOPHARMA
A first-in-patient study of lemzoparlimab, a differentiated anti-CD47 antibody, in subjects with relapsed/refractory malignancy: initialmonotherapy results
I-MAB BIOPHARMA
I-Mab to Hold Investor Call to Present In-Depth Phase 1 Clinical Data on Highly Differentiated CD73 Antibody Uliledlimab . I-Mab today announced that it will hold a call with investors on Monday, June 7 at 8:00 a.m. ET to provide an in-depth clinical data analysis of its U.S. phase 1 study of uliledlimab in combination with atezolizumab in patients with advanced cancers. I-MAB | INVESTOR RELATIONS May 27, 2021. SHANGHAI and GAITHERSBURG, Md. , May 27, 2021 /PRNewswire/ -- I-Mab (the "Company") (Nasdaq: IMAB ), a clinical-stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, today announced its participation in the following conferences in June. I-Mab PresentsPhase 1 Data on
I-MAB BIOPHARMA
Our discovery team has deep expertise in functional assays across a wide range of immune cells, including T cells, B cells and macrophages. By designing robust, high-throughput assays, we significantly increase screening efficiency and the probability of finding differentiated molecules. Our discovery team is passionate about, and has excelledI-MAB BIOPHARMA
This is a strong testament to our discovery capability in target biology, antibody engineering, translational medicine and antibody CMC. Bi-specific antibody panel. Despite the recent success of checkpoint inhibitors, it is estimated that more than 60% of cancerpatients
I-MAB BIOPHARMA
These in-licensing deals enable us to acquire multiple novel or highly differentiated clinical-stage assets with favorable clinical safety and early efficacy data. We have quickly built our China Portfolio through in-licensing deals with global biotech partners, including MorphoSys, Genexine, MacroGenics and Ferring, and is the reason we areI-MAB BIOPHARMA
raven.lin@i-mabbiopharma.com. About ABL Bio: ABL Bio is a privately held South Korean biotechnology company developing antibody therapeutics for immuno-oncology and neurodegenerative disease. ABL Bio was founded in 2016 and recently completed series C fund raising of USD 65 million in 2018 after successful series A and B financing in2016 and 2017.
I-MAB | I-MAB AND ABL BIO TO PRESENT PRECLINICAL DATA FOR SHANGHAI and GAITHERSBURG, Md., June 12, 2020 /PRNewswire/ -- I-Mab (the "Company") (Nasdaq: IMAB), a clinical stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, will be presenting preclinical data on a newly developed, novel asset TJ-CD4B, at the American Association for Cancer Research (AACR) Virtual Annual Meeting II on 天境生物-I-MAB-医药研发-双靶点-独角兽企业-肿瘤自 …TRANSLATE THIS PAGE 天境生物是一家聚焦于肿瘤免疫和自身免疫领域的创新生物药企业,公司高度专注于在全球范围内开发具有“全球首创”和“同类最优”潜力的创新生物药。我们的使命是研制具有突破性的创新生物药,填补临床治疗需求的空白,并且切实提高全球患者的生存质量。I-MAB BIOPHARMA
I-Mab Biopharma (“I-Mab”), a China-based clinical stage biopharmaceutical company exclusively focused on the development of innovative biologics in immuno-oncology and autoimmune diseases, announces on Jan. 25, 2019 that its IND application for TJC4, has been approved by the US Food & Drug Administration (FDA).I-MAB BIOPHARMA
A first-in-patient study of lemzoparlimab, a differentiated anti-CD47 antibody, in subjects with relapsed/refractory malignancy: initialmonotherapy results
I-MAB BIOPHARMA
This is a strong testament to our discovery capability in target biology, antibody engineering, translational medicine and antibody CMC. Bi-specific antibody panel. Despite the recent success of checkpoint inhibitors, it is estimated that more than 60% of cancerpatients
I-MAB BIOPHARMA
These in-licensing deals enable us to acquire multiple novel or highly differentiated clinical-stage assets with favorable clinical safety and early efficacy data. We have quickly built our China Portfolio through in-licensing deals with global biotech partners, including MorphoSys, Genexine, MacroGenics and Ferring, and is the reason we areI-MAB BIOPHARMA
I-Mab (Nasdaq: IMAB) is an innovation-driven global biotech company focusing on discovery, development and soon commercialization of novel and highly differentiated biologics in immuno-oncology therapeutic area. The Company's mission is to bring transformational medicines to patients around the world through drug innovation.I-MAB BIOPHARMA
I-Mab (Nasdaq: IMAB) is a dynamic, global biotech company exclusively focused on discovery, development and soon commercialization of novel or highly differentiated biologics in the therapeutic areas of immuno-oncology and autoimmune diseases. The Company’s mission is to bring transformational medicines to patients around the world throughI-MAB BIOPHARMA
raven.lin@i-mabbiopharma.com. About ABL Bio: ABL Bio is a privately held South Korean biotechnology company developing antibody therapeutics for immuno-oncology and neurodegenerative disease. ABL Bio was founded in 2016 and recently completed series C fund raising of USD 65 million in 2018 after successful series A and B financing in2016 and 2017.
I-MAB | I-MAB TO HOST 2021 R&D DAY Mandarin session on April 7, 2021 and English session on April 26, 2021 for investors and research analysts. SHANGHAI, China and GAITHERSBURG, Md., March 24, 2021 (GLOBE NEWSWIRE) -- I-Mab (the “Company”) (Nasdaq: IMAB), a clinical stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, today announced that it will host its I-MAB | I-MAB ULILEDLIMAB PHASE 1 DATA CONFERENCE CALL I-Mab Uliledlimab Phase 1 Data Conference Call. Jun 7, 2021 at 8:00 AM EDT Meeting ID: 992 9308 1819 Password: 942873 I-MAB | ABBVIE AND I-MAB ENTER INTO GLOBAL STRATEGIC NORTH CHICAGO, Illinois and SHANGHAI, Sept. 4, 2020 /PRNewswire/ -- AbbVie (NYSE: ABBV) and I-Mab (Nasdaq: IMAB) announced today that AbbVie and I-Mab have signed a broad, global collaboration agreement for the development and commercialization of lemzoparlimab (also known as TJC4), an innovative anti-CD47 monoclonal antibody internally discovered and developed by I-Mab for I-MAB APPOINTS RUYI HE AND RONG SHAO TO BOARD OF DIRECTORS I-Mab Appoints Ruyi He and Rong Shao to Board of Directors June 1, 2021 SHANGHAI and GAITHERSBURG, Md. , June 1, 2021 /PRNewswire/ -- I-Mab (the "Company") (Nasdaq: IMAB ), a clinical stagebiopharmaceutical
I-MAB | I-MAB PRESENTS PHASE 1 DATA ON HIGHLY Uliledlimab is a highly differentiated CD73 antibody with a unique epitope that confers pharmacological advantages; Data from U.S. phase 1 dose escalation trial demonstrates favorable safety and tolerability with no dose-limiting toxicities; preliminary clinical activity of uliledlimab has been observed in patients with advanced cancers; ASCO abstract among Top 12 selected for posterI-MAB BIOPHARMA
I-Mab to Hold Investor Call to Present In-Depth Phase 1 Clinical Data on Highly Differentiated CD73 Antibody Uliledlimab . I-Mab today announced that it will hold a call with investors on Monday, June 7 at 8:00 a.m. ET to provide an in-depth clinical data analysis of its U.S. phase 1 study of uliledlimab in combination with atezolizumab in patients with advanced cancers. I-MAB | INVESTOR RELATIONS May 27, 2021. SHANGHAI and GAITHERSBURG, Md. , May 27, 2021 /PRNewswire/ -- I-Mab (the "Company") (Nasdaq: IMAB ), a clinical-stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, today announced its participation in the following conferences in June. I-Mab PresentsPhase 1 Data on
I-MAB BIOPHARMA
Our discovery team has deep expertise in functional assays across a wide range of immune cells, including T cells, B cells and macrophages. By designing robust, high-throughput assays, we significantly increase screening efficiency and the probability of finding differentiated molecules. Our discovery team is passionate about, and has excelledI-MAB BIOPHARMA
This is a strong testament to our discovery capability in target biology, antibody engineering, translational medicine and antibody CMC. Bi-specific antibody panel. Despite the recent success of checkpoint inhibitors, it is estimated that more than 60% of cancerpatients
I-MAB BIOPHARMA
These in-licensing deals enable us to acquire multiple novel or highly differentiated clinical-stage assets with favorable clinical safety and early efficacy data. We have quickly built our China Portfolio through in-licensing deals with global biotech partners, including MorphoSys, Genexine, MacroGenics and Ferring, and is the reason we areI-MAB BIOPHARMA
raven.lin@i-mabbiopharma.com. About ABL Bio: ABL Bio is a privately held South Korean biotechnology company developing antibody therapeutics for immuno-oncology and neurodegenerative disease. ABL Bio was founded in 2016 and recently completed series C fund raising of USD 65 million in 2018 after successful series A and B financing in2016 and 2017.
I-MAB | I-MAB AND ABL BIO TO PRESENT PRECLINICAL DATA FOR SHANGHAI and GAITHERSBURG, Md., June 12, 2020 /PRNewswire/ -- I-Mab (the "Company") (Nasdaq: IMAB), a clinical stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, will be presenting preclinical data on a newly developed, novel asset TJ-CD4B, at the American Association for Cancer Research (AACR) Virtual Annual Meeting II on 天境生物-I-MAB-医药研发-双靶点-独角兽企业-肿瘤自 …TRANSLATE THIS PAGE 天境生物是一家聚焦于肿瘤免疫和自身免疫领域的创新生物药企业,公司高度专注于在全球范围内开发具有“全球首创”和“同类最优”潜力的创新生物药。我们的使命是研制具有突破性的创新生物药,填补临床治疗需求的空白,并且切实提高全球患者的生存质量。I-MAB BIOPHARMA
I-Mab Biopharma (“I-Mab”), a China-based clinical stage biopharmaceutical company exclusively focused on the development of innovative biologics in immuno-oncology and autoimmune diseases, announces on Jan. 25, 2019 that its IND application for TJC4, has been approved by the US Food & Drug Administration (FDA).I-MAB BIOPHARMA
A first-in-patient study of lemzoparlimab, a differentiated anti-CD47 antibody, in subjects with relapsed/refractory malignancy: initialmonotherapy results
I-MAB BIOPHARMA
I-Mab to Hold Investor Call to Present In-Depth Phase 1 Clinical Data on Highly Differentiated CD73 Antibody Uliledlimab . I-Mab today announced that it will hold a call with investors on Monday, June 7 at 8:00 a.m. ET to provide an in-depth clinical data analysis of its U.S. phase 1 study of uliledlimab in combination with atezolizumab in patients with advanced cancers. I-MAB | INVESTOR RELATIONS May 27, 2021. SHANGHAI and GAITHERSBURG, Md. , May 27, 2021 /PRNewswire/ -- I-Mab (the "Company") (Nasdaq: IMAB ), a clinical-stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, today announced its participation in the following conferences in June. I-Mab PresentsPhase 1 Data on
I-MAB BIOPHARMA
Our discovery team has deep expertise in functional assays across a wide range of immune cells, including T cells, B cells and macrophages. By designing robust, high-throughput assays, we significantly increase screening efficiency and the probability of finding differentiated molecules. Our discovery team is passionate about, and has excelledI-MAB BIOPHARMA
This is a strong testament to our discovery capability in target biology, antibody engineering, translational medicine and antibody CMC. Bi-specific antibody panel. Despite the recent success of checkpoint inhibitors, it is estimated that more than 60% of cancerpatients
I-MAB BIOPHARMA
These in-licensing deals enable us to acquire multiple novel or highly differentiated clinical-stage assets with favorable clinical safety and early efficacy data. We have quickly built our China Portfolio through in-licensing deals with global biotech partners, including MorphoSys, Genexine, MacroGenics and Ferring, and is the reason we areI-MAB BIOPHARMA
raven.lin@i-mabbiopharma.com. About ABL Bio: ABL Bio is a privately held South Korean biotechnology company developing antibody therapeutics for immuno-oncology and neurodegenerative disease. ABL Bio was founded in 2016 and recently completed series C fund raising of USD 65 million in 2018 after successful series A and B financing in2016 and 2017.
I-MAB | I-MAB AND ABL BIO TO PRESENT PRECLINICAL DATA FOR SHANGHAI and GAITHERSBURG, Md., June 12, 2020 /PRNewswire/ -- I-Mab (the "Company") (Nasdaq: IMAB), a clinical stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, will be presenting preclinical data on a newly developed, novel asset TJ-CD4B, at the American Association for Cancer Research (AACR) Virtual Annual Meeting II on 天境生物-I-MAB-医药研发-双靶点-独角兽企业-肿瘤自 …TRANSLATE THIS PAGE 天境生物是一家聚焦于肿瘤免疫和自身免疫领域的创新生物药企业,公司高度专注于在全球范围内开发具有“全球首创”和“同类最优”潜力的创新生物药。我们的使命是研制具有突破性的创新生物药,填补临床治疗需求的空白,并且切实提高全球患者的生存质量。I-MAB BIOPHARMA
I-Mab Biopharma (“I-Mab”), a China-based clinical stage biopharmaceutical company exclusively focused on the development of innovative biologics in immuno-oncology and autoimmune diseases, announces on Jan. 25, 2019 that its IND application for TJC4, has been approved by the US Food & Drug Administration (FDA).I-MAB BIOPHARMA
A first-in-patient study of lemzoparlimab, a differentiated anti-CD47 antibody, in subjects with relapsed/refractory malignancy: initialmonotherapy results
I-MAB BIOPHARMA
This is a strong testament to our discovery capability in target biology, antibody engineering, translational medicine and antibody CMC. Bi-specific antibody panel. Despite the recent success of checkpoint inhibitors, it is estimated that more than 60% of cancerpatients
I-MAB BIOPHARMA
These in-licensing deals enable us to acquire multiple novel or highly differentiated clinical-stage assets with favorable clinical safety and early efficacy data. We have quickly built our China Portfolio through in-licensing deals with global biotech partners, including MorphoSys, Genexine, MacroGenics and Ferring, and is the reason we areI-MAB BIOPHARMA
I-Mab (Nasdaq: IMAB) is an innovation-driven global biotech company focusing on discovery, development and soon commercialization of novel and highly differentiated biologics in immuno-oncology therapeutic area. The Company's mission is to bring transformational medicines to patients around the world through drug innovation.I-MAB BIOPHARMA
I-Mab (Nasdaq: IMAB) is a dynamic, global biotech company exclusively focused on discovery, development and soon commercialization of novel or highly differentiated biologics in the therapeutic areas of immuno-oncology and autoimmune diseases. The Company’s mission is to bring transformational medicines to patients around the world throughI-MAB BIOPHARMA
raven.lin@i-mabbiopharma.com. About ABL Bio: ABL Bio is a privately held South Korean biotechnology company developing antibody therapeutics for immuno-oncology and neurodegenerative disease. ABL Bio was founded in 2016 and recently completed series C fund raising of USD 65 million in 2018 after successful series A and B financing in2016 and 2017.
I-MAB | SEC FILING
I-Mab Appoints Ruyi He and Rong Shao to Board of Directors . SHANGHAI, China, and GAITHERSBURG, MD. – June 1, 2021 – I-Mab (the “Company”) (Nasdaq: IMAB), a clinical stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, today announced that Dr. Ruyi He and Professor Rong Shao have been elected as members of the I-MAB | I-MAB TO HOST 2021 R&D DAY Mandarin session on April 7, 2021 and English session on April 26, 2021 for investors and research analysts. SHANGHAI, China and GAITHERSBURG, Md., March 24, 2021 (GLOBE NEWSWIRE) -- I-Mab (the “Company”) (Nasdaq: IMAB), a clinical stage biopharmaceutical company committed to the discovery, development and commercialization of novel biologics, today announced that it will host itsI-MAB BIOPHARMA
TJD5, also known as TJ004309, is a CD73 antibody from I-Mab’s proprietary discovery pipeline and is now co-developed with TRACON via an agreement signed on November 28, 2018, as part of a broad strategic partnership to develop multiple immuno-oncology programs with first-in-class and best-in-class potential from I-Mab’s immuno-oncology portfolio. I-MAB | ABBVIE AND I-MAB ENTER INTO GLOBAL STRATEGIC NORTH CHICAGO, Illinois and SHANGHAI, Sept. 4, 2020 /PRNewswire/ -- AbbVie (NYSE: ABBV) and I-Mab (Nasdaq: IMAB) announced today that AbbVie and I-Mab have signed a broad, global collaboration agreement for the development and commercialization of lemzoparlimab (also known as TJC4), an innovative anti-CD47 monoclonal antibody internally discovered and developed by I-Mab for I-MAB | I-MAB PRESENTS PHASE 1 DATA ON HIGHLY Uliledlimab is a highly differentiated CD73 antibody with a unique epitope that confers pharmacological advantages; Data from U.S. phase 1 dose escalation trial demonstrates favorable safety and tolerability with no dose-limiting toxicities; preliminary clinical activity of uliledlimab has been observed in patients with advanced cancers; ASCO abstract among Top 12 selected for poster* I-Mab
About Us
Our Vision
Important Events
Organization and Management Team Scientific Advisory BoardInvestors
Contact
* Science
Discovery Research
Technology Platforms Publications and Presentations Intellectual Property* R&D Capability
CMC Development
Non-clinical Development Translational Medicine Clinical DevelopmentR&D Governance
* Pipeline
Pipeline Overview
China Portfolio
Global Portfolio
* Partnerships
Overview
Partners
Partnering Opportunities * Facilities and Operations R&D Site in ShanghaiBeijing Site
US Site
Hong Kong Office
* Media and Investors Corporate GovernancePress Release
Media Reports
Partner News
Awards and RecognitionInvestor Relations
* Careers
Working at I-Mab
Join Us
*
*
*
*
*
Previous
DEVELOPING TRANSFORMATIONAL MEDICINES TO IMPROVE THE LIVES OF PATIENTS*
I-MAB ANNOUNCES UPCOMING PARTICIPATION AT JUNE CONFERENCES I-Mab today announced its participation in the following conferencesin June.
IMMUNOLOGY AND INNOVATION FOR TRANSFORMATIONAL MEDICINES*
I-MAB TO HOLD INVESTOR CALL TO PRESENT IN-DEPTH PHASE 1 CLINICAL DATA ON HIGHLY DIFFERENTIATED CD73 ANTIBODY ULILEDLIMAB I-Mab today announced that it will hold a call with investors on Monday, June 7 at 8:00 a.m. ET to provide an in-depth clinical data analysis of its U.S. phase 1 study of uliledlimab in combination with atezolizumab in patients with advanced cancers. BUILDING A PIPELINE OF POTENTIAL NOVEL AND HIGHLY DIFFERENTIATEDMEDICINES
*
I-MAB APPOINTS RUYI HE AND RONG SHAO TO BOARD OF DIRECTORS I-Mab today announced that Dr. Ruyi He and Professor Rong Shao have been elected as members of the Company’s board of directors. DEVELOPING TRANSFORMATIONAL MEDICINES TO IMPROVE THE LIVES OF PATIENTS*
I-MAB ANNOUNCES UPCOMING PARTICIPATION AT JUNE CONFERENCES I-Mab today announced its participation in the following conferencesin June.
IMMUNOLOGY AND INNOVATION FOR TRANSFORMATIONAL MEDICINES*
I-MAB TO HOLD INVESTOR CALL TO PRESENT IN-DEPTH PHASE 1 CLINICAL DATA ON HIGHLY DIFFERENTIATED CD73 ANTIBODY ULILEDLIMAB I-Mab today announced that it will hold a call with investors on Monday, June 7 at 8:00 a.m. ET to provide an in-depth clinical data analysis of its U.S. phase 1 study of uliledlimab in combination with atezolizumab in patients with advanced cancers.Next
* 1
* 2
* 3
*
COMPANY OVERVIEW
*
SCIENCE
*
R&D CAPABILITY
*
PRODUCT PIPELINE
*
GLOBAL PARTNERSHIPS
Copyright I-Mab Biopharma Co., Ltd. all rights reserved 2019 沪ICP备17007960号-2Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0