Are you over 18 and want to see adult content?
More Annotations
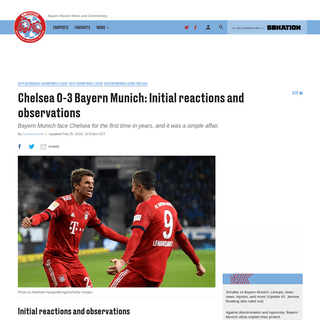
A complete backup of www.bavarianfootballworks.com/2020/2/25/21152214/chelsea-vs-bayern-munich-lineups-live-stream-how-to-watch-
Are you over 18 and want to see adult content?
A complete backup of news.mt.co.kr/mtview.php?no=2020030207010037546
Are you over 18 and want to see adult content?
Favourite Annotations

A complete backup of cartierwomensinitiative.com
Are you over 18 and want to see adult content?
A complete backup of calciodangolo.com
Are you over 18 and want to see adult content?

A complete backup of pkingcompu.wordpress.com
Are you over 18 and want to see adult content?

A complete backup of institutomanhattanschool.com
Are you over 18 and want to see adult content?
A complete backup of healthmanagement.org
Are you over 18 and want to see adult content?
Text
points to consider.
RISK MANAGEMENT IN MEDICAL DEVICE DESIGN The key to successful risk management in medical device design is to start early. As soon as conceptual designs are available, the risk management process can begin. A preliminary hazard analysis can be useful in selecting the concept with the highest level of inherentsafety.
GET READY FOR THE HOSPITAL OF THE FUTURE DETERMINING SAMPLE SIZE FOR TESTING An MD&DI May 1997 Column HELP DESK Director, corporate statistical resources, at Medtronic, Inc. (Minneapolis), explains the statistical strategies used during clinical trials for determining equivalence and offers ways to calculate sample sizes. TOP 10 MOLECULAR DIAGNOSTICS COMPANIES: A CLOSER LOOK The list was compiled by analysis firm Kalorama Information. The names on the list are not surprising, but it's worth looking at them, and the molecular diagnostics industry in general, a little more closely to help determine what kind of factors translate to success in this exciting field. In 2012 MEDICAL DESIGN & MANUFACTURING (MD&M) EAST 2021 Medical Design & Manufacturing (MD&M) East 2021. Dec 07, 2021 to Dec 09, 2021. Jacob K. Javits Convention Center, New York City. See medtech innovation on display in New York City. Medtech moves fast. Keep the pace with the latest innovations and immerse yourself in a community of visionaries and industry leaders. 2021 MEDICAL DESIGN EXCELLENCE AWARDS FINALISTS The Medical Design Excellence Awards competition is the medical technology industry's premier awards program. Since 1998, the MDEAs have recognized achievements in medical product design and engineering that improve the quality of healthcare delivery and accessibility. Each year, a panel of jurors REMOTE MEDICAL DEVICE SERVICING IS THE FUTURE Remote Medical Device Servicing Is the Future. Image courtesy of Dedicated Computing. Remote strategies add value even if the OEM does need to go out and do a physical repair. Remote diagnosis ensure the field tech arrives with the correct part in hand – no multi-step process or multiple visits are necessary. ROOT CAUSE ANALYSIS: ADVENTURES IN MEDICAL DEVICE Identifying the causes is the key to eliminating the errors. This is what Root Cause Analysis is all about--finding the causes of errors made in usability testing. The term "root cause" is used to emphasize that the goal is to find the "deep" or "underlying" causes, as opposed to the superficial causes. The root causes lead to things that can WHAT TYPES OF BIOCOMPATIBILITY TESTING DO YOU NEED TO On Thursday, December 4 at BIOMEDevice San Jose, Thor Rollins, in vivo biocompatibility section leader at Nelson Laboratories Inc. (Salt Lake City, UT), will speak on "Materials Selection and Sampling Techniques for Biocompatibility (ISO 10993)." Some of the themes that he will present at the conference are addressed in the following Q&A. MPMN: Please go into ISO 10993-1 and why 7 CONSIDERATIONS FOR CHOOSING A TESTING PARTNER Expectations for medical device testing are changing, thanks to evolving regulations and standards as well as other factors. Finding the right testing partner is critical for success, so here are a fewpoints to consider.
RISK MANAGEMENT IN MEDICAL DEVICE DESIGN The key to successful risk management in medical device design is to start early. As soon as conceptual designs are available, the risk management process can begin. A preliminary hazard analysis can be useful in selecting the concept with the highest level of inherentsafety.
GET READY FOR THE HOSPITAL OF THE FUTURE DETERMINING SAMPLE SIZE FOR TESTING An MD&DI May 1997 Column HELP DESK Director, corporate statistical resources, at Medtronic, Inc. (Minneapolis), explains the statistical strategies used during clinical trials for determining equivalence and offers ways to calculate sample sizes. TOP 10 MOLECULAR DIAGNOSTICS COMPANIES: A CLOSER LOOK The list was compiled by analysis firm Kalorama Information. The names on the list are not surprising, but it's worth looking at them, and the molecular diagnostics industry in general, a little more closely to help determine what kind of factors translate to success in this exciting field. In 2012 MEDICAL DEVICE SUPPLY-CHAIN OPERATIONS AND PANDEMIC The COVID-19 pandemic tested the resilience of supply chains. The medical device industry was no exception. The pandemic has disrupted everything from supply and demand to shipping and workplace capacity. Medical device companies have experienced delays within theirfacilities, and
ABBOTT SEES COVID-19 TESTING DEMAND DROOP AS RESTRICTIONS Abbott's outlook for rapid COVID-19 testing has changed significantly as the company has seen a sudden drop in COVID-19 testing demand due to shifting in guidelines related to testing for people who are fully vaccinated. In April, the company reported READY FOR A TURNKEY AUTOMATION TRANSFORMATION It’s not a new trend. But automation today is poised to reach new heights in medical device manufacturing. With skilled labor scarce and demand surging for products related to COVID-19, many companies are looking to incorporate automation to accelerate WHAT DOES TELEHEALTH’S POST-PANDEMIC FUTURE LOOK LIKE Before the global health crisis, seeking a consultation too often meant going in-person to a hospital or doctor’s office and waiting for a one-to-one appointment. The effectiveness, efficiency, and cost associated with telehealth were regarded with skepticism by providersand patients alike
REMOTE MEDICAL DEVICE SERVICING IS THE FUTURE Remote Medical Device Servicing Is the Future. Image courtesy of Dedicated Computing. Remote strategies add value even if the OEM does need to go out and do a physical repair. Remote diagnosis ensure the field tech arrives with the correct part in hand – no multi-step process or multiple visits are necessary. THE MEDICAL DEVICE INDUSTRY NEEDS RISK MANAGEMENT IN THE Amid the current COVID-19 pandemic, many medical device manufacturers and suppliers are moving at unprecedented speeds to develop and enact updated risk management policies and procedures. Much of what they are developing are plans to help manage manufacturing and supply-chain challenges in times of ABIOMED STRENGTHENS HF OFFERINGS WITH PRECARDIA Abiomed is augmenting its heart failure treatment offerings with the acquisition of preCARDIA for an undisclosed sum. St. Paul, MN-based preCARDIA has developed a catheter-based system designed to rapidly treat ADHF-related volume overload by reducing cardiac filling pressures and promoting decongestion to improve overall cardiac andrenal function.
ALIVECOR MOVES TOWARD AMBULATORY MONITORING SYSTEMS WITH AliveCor, an ECG technology specialist, is going in a bold direction with its newest acquisition. The Mountain View, CA-based company is purchasing CardioLabs, a monitoring and cardiac diagnostic service provider. AliveCor said the acquisition would set THE NEW AND IMPROVED ISO 10993-12:2021 Every time there is an update to a standard, especially an overarching one like the ISO 10993-12 standard that covers sample preparation recommendations, it comes with a slight dose of anxiety. This is because it might mean there is now a gap between what is VERILY STRIKES OUT ON ADDING PARKINSON’S ASSESSMENT TO 1 day ago · Verily Life Sciences has hit a snag in bringing a Parkinson’s disease exam tool to its research smartwatch. The Alphabet Company’s bid to add the virtual motor exam (VME III) was recently rejected by FDA. Data gathered as a result of the assessment could be used in drug development studies MEDICAL DESIGN & MANUFACTURING (MD&M) EAST 2021 Medical Design & Manufacturing (MD&M) East 2021. Dec 07, 2021 to Dec 09, 2021. Jacob K. Javits Convention Center, New York City. See medtech innovation on display in New York City. Medtech moves fast. Keep the pace with the latest innovations and immerse yourself in a community of visionaries and industry leaders. 2021 MEDICAL DESIGN EXCELLENCE AWARDS FINALISTS The Medical Design Excellence Awards competition is the medical technology industry's premier awards program. Since 1998, the MDEAs have recognized achievements in medical product design and engineering that improve the quality of healthcare delivery and accessibility. Each year, a panel of jurors REMOTE MEDICAL DEVICE SERVICING IS THE FUTURE Remote Medical Device Servicing Is the Future. Image courtesy of Dedicated Computing. Remote strategies add value even if the OEM does need to go out and do a physical repair. Remote diagnosis ensure the field tech arrives with the correct part in hand – no multi-step process or multiple visits are necessary. ROOT CAUSE ANALYSIS: ADVENTURES IN MEDICAL DEVICE Identifying the causes is the key to eliminating the errors. This is what Root Cause Analysis is all about--finding the causes of errors made in usability testing. The term "root cause" is used to emphasize that the goal is to find the "deep" or "underlying" causes, as opposed to the superficial causes. The root causes lead to things that can WHAT TYPES OF BIOCOMPATIBILITY TESTING DO YOU NEED TO On Thursday, December 4 at BIOMEDevice San Jose, Thor Rollins, in vivo biocompatibility section leader at Nelson Laboratories Inc. (Salt Lake City, UT), will speak on "Materials Selection and Sampling Techniques for Biocompatibility (ISO 10993)." Some of the themes that he will present at the conference are addressed in the following Q&A. MPMN: Please go into ISO 10993-1 and why 7 CONSIDERATIONS FOR CHOOSING A TESTING PARTNER Expectations for medical device testing are changing, thanks to evolving regulations and standards as well as other factors. Finding the right testing partner is critical for success, so here are a fewpoints to consider.
RISK MANAGEMENT IN MEDICAL DEVICE DESIGN The key to successful risk management in medical device design is to start early. As soon as conceptual designs are available, the risk management process can begin. A preliminary hazard analysis can be useful in selecting the concept with the highest level of inherentsafety.
GET READY FOR THE HOSPITAL OF THE FUTURE DETERMINING SAMPLE SIZE FOR TESTING An MD&DI May 1997 Column HELP DESK Director, corporate statistical resources, at Medtronic, Inc. (Minneapolis), explains the statistical strategies used during clinical trials for determining equivalence and offers ways to calculate sample sizes. TOP 10 MOLECULAR DIAGNOSTICS COMPANIES: A CLOSER LOOK The list was compiled by analysis firm Kalorama Information. The names on the list are not surprising, but it's worth looking at them, and the molecular diagnostics industry in general, a little more closely to help determine what kind of factors translate to success in this exciting field. In 2012 MEDICAL DESIGN & MANUFACTURING (MD&M) EAST 2021 Medical Design & Manufacturing (MD&M) East 2021. Dec 07, 2021 to Dec 09, 2021. Jacob K. Javits Convention Center, New York City. See medtech innovation on display in New York City. Medtech moves fast. Keep the pace with the latest innovations and immerse yourself in a community of visionaries and industry leaders. 2021 MEDICAL DESIGN EXCELLENCE AWARDS FINALISTS The Medical Design Excellence Awards competition is the medical technology industry's premier awards program. Since 1998, the MDEAs have recognized achievements in medical product design and engineering that improve the quality of healthcare delivery and accessibility. Each year, a panel of jurors REMOTE MEDICAL DEVICE SERVICING IS THE FUTURE Remote Medical Device Servicing Is the Future. Image courtesy of Dedicated Computing. Remote strategies add value even if the OEM does need to go out and do a physical repair. Remote diagnosis ensure the field tech arrives with the correct part in hand – no multi-step process or multiple visits are necessary. ROOT CAUSE ANALYSIS: ADVENTURES IN MEDICAL DEVICE Identifying the causes is the key to eliminating the errors. This is what Root Cause Analysis is all about--finding the causes of errors made in usability testing. The term "root cause" is used to emphasize that the goal is to find the "deep" or "underlying" causes, as opposed to the superficial causes. The root causes lead to things that can WHAT TYPES OF BIOCOMPATIBILITY TESTING DO YOU NEED TO On Thursday, December 4 at BIOMEDevice San Jose, Thor Rollins, in vivo biocompatibility section leader at Nelson Laboratories Inc. (Salt Lake City, UT), will speak on "Materials Selection and Sampling Techniques for Biocompatibility (ISO 10993)." Some of the themes that he will present at the conference are addressed in the following Q&A. MPMN: Please go into ISO 10993-1 and why 7 CONSIDERATIONS FOR CHOOSING A TESTING PARTNER Expectations for medical device testing are changing, thanks to evolving regulations and standards as well as other factors. Finding the right testing partner is critical for success, so here are a fewpoints to consider.
RISK MANAGEMENT IN MEDICAL DEVICE DESIGN The key to successful risk management in medical device design is to start early. As soon as conceptual designs are available, the risk management process can begin. A preliminary hazard analysis can be useful in selecting the concept with the highest level of inherentsafety.
GET READY FOR THE HOSPITAL OF THE FUTURE DETERMINING SAMPLE SIZE FOR TESTING An MD&DI May 1997 Column HELP DESK Director, corporate statistical resources, at Medtronic, Inc. (Minneapolis), explains the statistical strategies used during clinical trials for determining equivalence and offers ways to calculate sample sizes. TOP 10 MOLECULAR DIAGNOSTICS COMPANIES: A CLOSER LOOK The list was compiled by analysis firm Kalorama Information. The names on the list are not surprising, but it's worth looking at them, and the molecular diagnostics industry in general, a little more closely to help determine what kind of factors translate to success in this exciting field. In 2012 MEDICAL DEVICE SUPPLY-CHAIN OPERATIONS AND PANDEMIC The COVID-19 pandemic tested the resilience of supply chains. The medical device industry was no exception. The pandemic has disrupted everything from supply and demand to shipping and workplace capacity. Medical device companies have experienced delays within theirfacilities, and
ABBOTT SEES COVID-19 TESTING DEMAND DROOP AS RESTRICTIONS 1 day ago · Abbott's outlook for rapid COVID-19 testing has changed significantly as the company has seen a sudden drop in COVID-19 testing demand due to shifting in guidelines related to testing for people who are fully vaccinated. In April, the company reported READY FOR A TURNKEY AUTOMATION TRANSFORMATION 1 day ago · It’s not a new trend. But automation today is poised to reach new heights in medical device manufacturing. With skilled labor scarce and demand surging for products related to COVID-19, many companies are looking to incorporate automation to accelerate WHAT DOES TELEHEALTH’S POST-PANDEMIC FUTURE LOOK LIKE Before the global health crisis, seeking a consultation too often meant going in-person to a hospital or doctor’s office and waiting for a one-to-one appointment. The effectiveness, efficiency, and cost associated with telehealth were regarded with skepticism by providersand patients alike
REMOTE MEDICAL DEVICE SERVICING IS THE FUTURE Remote Medical Device Servicing Is the Future. Image courtesy of Dedicated Computing. Remote strategies add value even if the OEM does need to go out and do a physical repair. Remote diagnosis ensure the field tech arrives with the correct part in hand – no multi-step process or multiple visits are necessary. THE MEDICAL DEVICE INDUSTRY NEEDS RISK MANAGEMENT IN THE Amid the current COVID-19 pandemic, many medical device manufacturers and suppliers are moving at unprecedented speeds to develop and enact updated risk management policies and procedures. Much of what they are developing are plans to help manage manufacturing and supply-chain challenges in times of ABIOMED STRENGTHENS HF OFFERINGS WITH PRECARDIA Abiomed is augmenting its heart failure treatment offerings with the acquisition of preCARDIA for an undisclosed sum. St. Paul, MN-based preCARDIA has developed a catheter-based system designed to rapidly treat ADHF-related volume overload by reducing cardiac filling pressures and promoting decongestion to improve overall cardiac andrenal function.
ALIVECOR MOVES TOWARD AMBULATORY MONITORING SYSTEMS WITH AliveCor, an ECG technology specialist, is going in a bold direction with its newest acquisition. The Mountain View, CA-based company is purchasing CardioLabs, a monitoring and cardiac diagnostic service provider. AliveCor said the acquisition would set THE NEW AND IMPROVED ISO 10993-12:2021 Every time there is an update to a standard, especially an overarching one like the ISO 10993-12 standard that covers sample preparation recommendations, it comes with a slight dose of anxiety. This is because it might mean there is now a gap between what is VERILY STRIKES OUT ON ADDING PARKINSON’S ASSESSMENT TO 14 hours ago · Verily Life Sciences has hit a snag in bringing a Parkinson’s disease exam tool to its research smartwatch. The Alphabet Company’s bid to add the virtual motor exam (VME III) was recently rejected by FDA. Data gathered as a result of the assessment could be used in drug development studies We use cookies to improve your website experience. To learn about our use of cookies and how you can manage your cookie settings, please see our Cookie Policy . By continuing to use the website, you consent to our use of cookies.×
https://www.mddionline.com/sites/all/themes/penton_subtheme_mddionline/images/logos/footer.pngInforma
MD+DI Online is part of the Informa Markets Division of Informa PLC* Informa PLC
* About us
* Investor relations* Talent
This site is operated by a business or businesses owned by Informa PLC and all copyright resides with them. Informa PLC's registered office is 5 Howick Place, London SW1P 1WG. Registered in England and Wales.Number 8860726.
__ __
__ __
* Supplier Directory* Events
* Subscribe
* Advertise
* About
* More__
*
Search __ __
* Register
* Log In
* __
* Register
* Log In
* Markets
* Cardiovascular
* Diabetes
* Drug-Delivery
* General Hospital
* IVD
* Neurological
* Orthopedic
* Radiological
* Surgical
See All Markets »
* Product Development* Components
* Design & Engineering* Human Factors
* Materials
* R&D
See All Product Development »* Manufacturing
* Automation
* Contract Manufacturing* Equipment
* Inspection
* Manufacturing Processes* Packaging
* Sterilization
See All Manufacturing » * Regulatory & Quality* Regulations
* Legal
* QA/QC
* Standards
* Testing
See All Regulatory & Quality »* Technologies
* 3D Printing
* Artificial Intelligence * Augmented/Virtual Reality* Digital Health
* Implants
* Robotics
See All Technologies »* Qmed
* " style="">Resources* Brochures
* Videos
* Webinars
* White Papers
* Supplier News
* Industry Reports
Featured
Medtronic Pulls the Plug on the HVAD SystemJun 03, 2021
Abbott said it has the capacity to fill in the gap left by Medtronic stopping the sale and distribution of the HVAD system. Medical Device Sterilization: 4 Alternatives to Using Ethylene OxideMay 29, 2021
Why ethylene oxide (EO) is used to sterilize medical devices, plus four alternative sterilization methods to consider. iRhythm’s President and CEO ResignsJun 02, 2021
Mike Coyle, who took over the leadership position of iRhythm Technologies in January, said he was leaving the company for “personal reasons.” The New and Improved ISO 10993-12:2021—What Is Really ChangingJun
02, 2021
While the updated standard features no major revisions, there is more discussion on what to consider for sample preparation. Regulatory & Quality FDA Hopes to Curb Infection Risks with New Endoscope Clearances.May 31, 2021
The federal agency gave out nods to EndoFresh and UroViu forendoscopes.
Medtech Design Summit 2021: Design Inspiration for Your Next MedicalInnovation
Learn best practices in medtech design and hear the winners of the 2021 Medical Design Excellence Awards in this free webinar series May13. Register today!
Recent
Medtech in a Minute: Big News from Cardio Players Medtronic, LivaNova, iRhythm, and AbiomedJun 04, 2021
Senseonics Touts Solid Data for 180 Day CGMJun 04, 2021
AliveCor Moves Toward Ambulatory Monitoring Systems with AcquisitionJun 04, 2021
Surgical Navigation: Breakthrough Innovations in 6DOF Expand Potential for Minimally Invasive Diagnostic and Therapeutic DevicesJun 03, 2021
What Does Telehealth’s Post-Pandemic Future Look Like?Jun
03, 2021
Abiomed Strengthens HF Offerings with PreCardia AcquisitionJun 03, 2021
LivaNova Completes Initial Close of Heart Valve Biz DivestitureJun 01, 2021
How to Manage Massive Medical Device Packaging ChangesJun
01, 2021
directory
Is Your Company in Medtech's Top 100?directory
Find Qualified Medtech Industry Suppliers in the Qmed DirectoryPopular
30 Minutes of Healthy Sweat Will Easily Power a Small Medical DeviceJun 01, 2021
Like 3D Chess These Hospital Robots Ride the Elevators Floor to FloorJun 01, 2021
Exoskeleton Has Positive Impact on Patients with MSJun 01,
2021
Verily’s Onduo Branches into Mental and Behavioral HealthJun 01, 2021
Medtech in a Minute: AliveCor's Spat with Apple, Boston Scientific's Stent Problem, and MoreMay 28, 2021
IVD
Veracyte to Expand Cancer-Testing Reach with HalioDx AcquisitionJun
01, 2021
Marc Stapley starts his role as Veracyte CEO with this acquisition. Stapley takes over the role from Bonnie Anderson, who led the companyfor 14...
Guardant’s Partnership with Janssen Bears FruitMay 24, 2021
Redwood City, CA-based Guardant won a nod from FDA to use the Guardant 360 as companion diagnostic for Janssen Pharmaceutical’sRybrevant...
Upcoming Events
Medical Design & Manufacturing (MD&M) West 2021 Aug 10, 2021 to Aug 12, 2021 BIOMEDevice Boston 2021 Sep 21, 2021 to Sep 22, 2021 Medical Design & Manufacturing (MD&M) East 2021 Dec 07, 2021 to Dec 09, 2021 Design & Engineering 20 Years of PillCam Capsule Endoscopy Innovation__May 28, 2021
A Medtronic engineer shares the greatest triumphs and challenges in the PillCam technology's 20-year evolution. The Big Shift in Healthcare—Is Medtech Ready to Help?May 28,
2021
‘Healthcare at home should be on every nonsurgical company’s mind,’ according to a healthcare executive speaking during MedExecWomen’s Spring event. Phillips-Medisize Launches Aria Smart Autoinjector Platform and MoreSupplier News__
May 28, 2021
Here’s what was new in the world of medical device suppliers duringthe week of May 23.
The Next Stage of Evolution for iRhythm’s Zio PatchMay
27, 2021
The San Francisco, CA-based company has won FDA clearance for a new design and upgraded artificial intelligence for the wearable cardiacmonitor.
Let’s Talk Medtech Gaining 'Insight' on Cardiac Insight’s Recent SurgeMay
26, 2021
Let’s Talk Medtech spoke with Dave Hadley, Phd, Senior VP of Research & Development of Cardiac Insight, about the company and the exploding ECG market. 'Adversity Can Give Us the Hidden Gift of True Purpose'May 05, 2021
More Than Just A Surgical Robotics CompanyApr 21, 2021
ResMed’s Jim Hollingshead Speaks on Firm’s Growth and Role ofSoftware Engineers
Mar 29, 2021
Let's Talk Medtech: Hologic's Kevin Thornal Explores the Future ofDiagnostics__
Mar 15, 2021
MEDTECH CAREER PLANNING Medtech Salary Survey 2021__Apr 28, 2021
Video
Do We Need a Better Term for Digital Health?Gallery
Amcor Unveils New Heat Seal Coating for Tyvek and Paper and MoreSupplier News
BEHIND THE DESIGN AND ENGINEERING OF A BREAKTHROUGH DEVICE FOR STROKEREHAB
May 11, 2021
More
Want a Job in Mechanical Engineering? Here Are 20 Top Companies__May 26, 2021
Clinical Asset Management Can Do More Than Just Track Medical DevicesMay 26, 2021
Virtual Engineering Days Supported by Key Industry AssociationsMay 26, 2021
What Medtech CEOs Expect from the Second Half of the Year__May 26, 2021
* News
* Products
* Whitepapers
* Husky & Chem-Trend Create a Simple and Effective Color Change Feature for Altanium® Hot Runner Temperature Controllers * Join the ZEISS Innovation Rocks Spring Edition 2021 * NextPhase Medical Devices Acquires Proven Process Medical Devices * Flambeau Boosts Manufacturing Capability * FVD Variable Dispense Pump * Handheld Surface Analyst 5001 * Qosina Provides Cost-Effective Off-the-Shelf and Custom Tubing * LHQ Series Quiet Operation Solenoid Valve * Unlocking Medtech's Future Potential with Connected Devices andServices
* How Device Companies Can Create End-to-End Connectivity for In-hospital Equipment * Four Strategies for Mitigating Supply Chain Disruption * Porous Titianium Plasma: A New Generation of Porous Titanium Coating on Metallic Substrate for Medical Devices _Medical Device and Diagnostic Industry (MD+DI)_ is the primary resource for manufacturers of medical devices and in vitro diagnostic products. Its mission is to help medtech industry professionals develop, design, and manufacture products that comply with complex and demanding regulations and evolving market requirements. For more than 40 years, _MD+DI_'s editorial quality and reliability have won it an unparalleled degree of trust from the industry. Some of the best and brightest experts in medtech contribute to _MD+DI_, and many of their articles have become standards of reference for the medical deviceindustry.
Sign up for the QMED & MD+DI Daily newsletter.Select Country
Go
KEEP YOU INFORMED
*Please keep me up-to-date with the latest products and services from MDDIONLINE.COM, part of Informa Markets.SelectYesNo *I would like to hear from Informa Markets' partners about product and services relevant to me. Informa Markets will not share any of your details with our partners unless you agree to us doing so directly.SelectYesNo Receive Marketing Updates I don't want to receive updates from MDDIONLINE.COM, part of Informa Markets about other relevant products and services. *I would like to hear from Informa Markets' partners about product and services relevant to me. Informa Markets will not share any of your details with our partners unless you agree to us doing so directly.SelectYesNo Receive Marketing Updates I accept the PRIVACY TERMS.
By continuing, you accept that MDDIONLINE.COM may contact you with updates, relevant promotions and information about events provided by Informa Markets and its partners. Your details may be shared with carefully selected partners who may contact you about their productsand services.
Receive Marketing Updates Informa Markets may wish to contact you regarding other events and products including MDDIONLINE.COM. If you DO NOT wish to receive these communications, let us know by ticking the b Receive Marketing Updates MDDIONLINE.COM'S carefully selected partners may want to get in touch. If you DO NOT wish to receive these communications, let us know byticking the box.
You may withdraw your consent to receive any of our communications at any time. You understand that your information will be used in accordance with the Privacy PolicyAgree to Terms
Enter your email above to receive messages about offerings by Informa, its brands, affiliates and/or third-party partners, consistent with Informa's Privacy Statement.
List
Leave this field blank* About
* Advertise
* Subscribe
* Battery Technology* Design News
* PlasticsToday
* Packaging Digest
* Powder & Bulk Solids* MD&M West
* MD&M East
* MD&M Minneapolis
* BIOMEDevice Boston * BIOMEDevice San Jose* Sitemap
Follow us:
__ __
__ __
Copyright © 2021. All rights reserved. Informa Markets, a trading division of Informa PLC.* Accessibility
* Privacy Policy
* Cookie Policy
* Terms of Use
* Visitor Terms and ConditionsBack to top
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0