Are you over 18 and want to see adult content?
More Annotations
A complete backup of gulfnews.com/entertainment/photos-priyanka-chopra-nick-jonas-sizzle-at-grammys-in-bold-plunging-gown-and-go
Are you over 18 and want to see adult content?

A complete backup of www.bbc.com/news/world-us-canada-51263718
Are you over 18 and want to see adult content?

A complete backup of www.forbes.com/sites/blakeoestriecher/2020/01/26/wwe-royal-rumble-2020-results-drew-mcintyre-upsets-roman-r
Are you over 18 and want to see adult content?
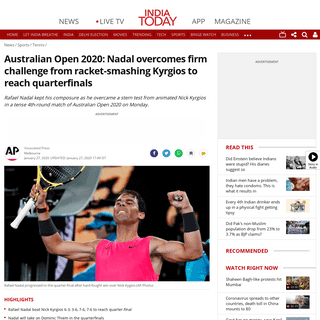
A complete backup of www.indiatoday.in/sports/tennis/story/australian-open-2020-rafael-nadal-beats-nick-kyrgios-books-quarterfin
Are you over 18 and want to see adult content?
Favourite Annotations
A complete backup of strongstrongsupply.com
Are you over 18 and want to see adult content?
A complete backup of landcruiserclub.net
Are you over 18 and want to see adult content?
Text
2021.
IN 2020, FDA AUTHORIZED 132 NOVEL MEDICAL DEVICES In 2020, FDA Authorized 132 Novel Medical Devices. The record-high number of authorizations last year was a significant jump over 2019. In 2009, FDA authorized just 29 novel devices; in 2015 it authorized 86; and five years later the number hit 132 novel devices. 2020 was a year for innovation, but the COVID-19 pandemic was only partially SECURING MEDICAL DEVICES: WHAT IS REALLY NEEDED? A security solution for medical devices must protect firmware from tampering, secure the data stored by the device, secure communication, and protect the device from cyberattacks. This can only be achieved by building in security from the early stages of design. There is no “one-size-fits-all security solution” for medical devices. PEDIATRIC MEDICAL DEVICES ADDRESS COVID-19, 16 COMPANIES In addition to its annual “Make Your Medical Device Pitch for Kids!” competition, which focuses on innovation in the pediatric space, this year the National Capital Consortium for Pediatric Device Innovation (NCC-PDI) launched a COVID-19 edition for 2020. Although COVID-19 has not been affecting the pediatric population as much as other age groups, it is remains a serious threat to children. FIVE STEPS TO MEDICAL DEVICE COMMERCIALIZATION EU MDR COST OF COMPLIANCE: THE RESULTS ARE IN 32% of respondents expect compliance with EU MDR will take up more than 8% of their company’s EU revenue. An estimate of the cost of compliance connected to global revenue was much smaller. Continue to the rest of the survey results on page 2. Pages: 1 2. RECENT FDA ACTION: THREE RECALLS AND TEN MEDICAL DEVICE Recent FDA Action: Three Recalls and Ten Medical Device Approvals. The device approvals address critical patient need. FDA recently released a list of medical devices it has approved and recalled over the past few months. All of the recalls were Class I recalls. MEDICAL DEVICE REGULATION With the application date fast approaching, gain insights into the new MDR requirements. Following the webinar, you will be better prepared to meet the increased requirements that have been placed on medical device manufactures. You will gain an understanding of your next steps, whether it is additional MDR transition training, or what to discuss with your notified body. SOURCING IN ASIA: PARTNER WITH THE RIGHT SUPPLIER FROM THE Most international medical device companies work with Asian distributors to sell their products in the local marketplace. Finding, qualifying, and signing up the right distributors is an important job since these distributors will sell your products locally. WHEN COMPLAINT INVESTIGATIONS MEET SERVICING AND REPAIRING When Complaint Investigations Meet Servicing and Repairing Devices. Many companies are unsure of how to complete a complaint investigation when the service and repair process is involved. Manufacturers of medical devices must often service and repair products once they are out in the field. If these device are involved in a future complaint HOME | MEDTECH INTELLIGENCEBUSINESS ANALYSISCOMBINATION PRODUCTSMARKET ACCESSOPERATIONSPRODUCT DEVELOPMENT The Transformation to True Digital Healthcare. By Stephen Bernstein, Dana Safran, Jody Tropeano, Lisa Schmitz Mazur, Sara Vaezy, Tom Kiesau. Digital health tools and technology have the potential to dramatically improve the patient care experience, but the shift requires purposeful coordination and planned architecture. April 28,2021.
IN 2020, FDA AUTHORIZED 132 NOVEL MEDICAL DEVICES In 2020, FDA Authorized 132 Novel Medical Devices. The record-high number of authorizations last year was a significant jump over 2019. In 2009, FDA authorized just 29 novel devices; in 2015 it authorized 86; and five years later the number hit 132 novel devices. 2020 was a year for innovation, but the COVID-19 pandemic was only partially SECURING MEDICAL DEVICES: WHAT IS REALLY NEEDED? A security solution for medical devices must protect firmware from tampering, secure the data stored by the device, secure communication, and protect the device from cyberattacks. This can only be achieved by building in security from the early stages of design. There is no “one-size-fits-all security solution” for medical devices. PEDIATRIC MEDICAL DEVICES ADDRESS COVID-19, 16 COMPANIES In addition to its annual “Make Your Medical Device Pitch for Kids!” competition, which focuses on innovation in the pediatric space, this year the National Capital Consortium for Pediatric Device Innovation (NCC-PDI) launched a COVID-19 edition for 2020. Although COVID-19 has not been affecting the pediatric population as much as other age groups, it is remains a serious threat to children. FIVE STEPS TO MEDICAL DEVICE COMMERCIALIZATION EU MDR COST OF COMPLIANCE: THE RESULTS ARE IN 32% of respondents expect compliance with EU MDR will take up more than 8% of their company’s EU revenue. An estimate of the cost of compliance connected to global revenue was much smaller. Continue to the rest of the survey results on page 2. Pages: 1 2. RECENT FDA ACTION: THREE RECALLS AND TEN MEDICAL DEVICE Recent FDA Action: Three Recalls and Ten Medical Device Approvals. The device approvals address critical patient need. FDA recently released a list of medical devices it has approved and recalled over the past few months. All of the recalls were Class I recalls. MEDICAL DEVICE REGULATION With the application date fast approaching, gain insights into the new MDR requirements. Following the webinar, you will be better prepared to meet the increased requirements that have been placed on medical device manufactures. You will gain an understanding of your next steps, whether it is additional MDR transition training, or what to discuss with your notified body. SOURCING IN ASIA: PARTNER WITH THE RIGHT SUPPLIER FROM THE Most international medical device companies work with Asian distributors to sell their products in the local marketplace. Finding, qualifying, and signing up the right distributors is an important job since these distributors will sell your products locally. WHEN COMPLAINT INVESTIGATIONS MEET SERVICING AND REPAIRING When Complaint Investigations Meet Servicing and Repairing Devices. Many companies are unsure of how to complete a complaint investigation when the service and repair process is involved. Manufacturers of medical devices must often service and repair products once they are out in the field. If these device are involved in a future complaint COLUMN | REMOTE AUDITS AND OTHER NEW WAYS OF WORKING IN A Audits for medical device companies and other organizations in the medtech space used to be primarily in person. However, the pandemic magnified flaws in the global regulatory process. A shift to remote audits may be the key to interorganizational and REDUCE YOUR MEDICAL DEVICE TIME-TO-MARKET WITH CPQ Specifically designed for the Medical and Mobility industry’s complex and custom products, discover how CPQ (configure, price, quote) software can help your company save money, increase customer satisfaction, and generate real ROI. EMERGING TRENDS IN CARDIOVASCULAR DEVICES Cardiac ablation devices accounted for almost 72% of the cardiovascular devices market share in 2020 and are expected to gain massive momentum and, as per GMI estimates. The rising prevalence of atrial fibrillation is a major contributor to the popularity of these devices. The products are addressing an ever-increasing demand forminimally
BIOLOGICAL EVALUATION OF MEDICAL DEVICES Biological Evaluation of Medical Devices. The biological safety of a device is established for both short-term (acute) and longtime (chronic) safety risks to patients and device users. Medical device manufacturers must investigate their devices thoroughly to meet these expectations to receive approval to market their devices. MEDTECH ECONOMICS ARCHIVES A strategy that pulls in regulatory, quality, clinical, reimbursement, marketing, compliance, legal and finance is required. Continue reading→
BUILD AN EFFECTIVE GLOBAL REGULATORY STRATEGY Global Medical Device Markets. According to the U.S. Department of Commerce, the medical device marketplace worldwide is expected to experience a 5% annual growth rate over the next few years. By the year 2020, the total market is expected to reach approximately $440 billion. The U.S. accounts for 45% of this market share. RECENT FDA ACTION: THREE RECALLS AND TEN MEDICAL DEVICE Recent FDA Action: Three Recalls and Ten Medical Device Approvals. The device approvals address critical patient need. FDA recently released a list of medical devices it has approved and recalled over the past few months. All of the recalls were Class I recalls. EIGHT STEPS TO POST-MARKET SURVEILLANCE COMPLIANCE Follow these eight key steps to stay on top of post-market surveillance and navigate the changing regulatory climate requirements. 1. Stay Ahead of the Game. Manufacturers should not rely solely on waiting to be informed that significant regulatory changes have been made. Manufacturers should take a proactive approach when itcomes to post
WHAT INDUSTRY IS SAYING ABOUT PROPOSED ETHYLENE OXIDE More than 20 billion medical devices sold in the United States each year are sterilized using ethylene oxide (EtO), according to an agency statement from then-FDA Acting Commissioner Ned Sharpless, M.D. on October 25. EtO is the most common method of sterilization in the country, and as such, the closures of EtO facilities across the country are raising strong concerns among stakeholders. WHEN COMPLAINT INVESTIGATIONS MEET SERVICING AND REPAIRING When Complaint Investigations Meet Servicing and Repairing Devices. Many companies are unsure of how to complete a complaint investigation when the service and repair process is involved. Manufacturers of medical devices must often service and repair products once they are out in the field. If these device are involved in a future complaint HOME | MEDTECH INTELLIGENCEBUSINESS ANALYSISCOMBINATION PRODUCTSMARKET ACCESSOPERATIONSPRODUCT DEVELOPMENT The Transformation to True Digital Healthcare. By Stephen Bernstein, Dana Safran, Jody Tropeano, Lisa Schmitz Mazur, Sara Vaezy, Tom Kiesau. Digital health tools and technology have the potential to dramatically improve the patient care experience, but the shift requires purposeful coordination and planned architecture. April 28,2021.
IN 2020, FDA AUTHORIZED 132 NOVEL MEDICAL DEVICES In 2020, FDA Authorized 132 Novel Medical Devices. The record-high number of authorizations last year was a significant jump over 2019. In 2009, FDA authorized just 29 novel devices; in 2015 it authorized 86; and five years later the number hit 132 novel devices. 2020 was a year for innovation, but the COVID-19 pandemic was only partially SECURING MEDICAL DEVICES: WHAT IS REALLY NEEDED? A security solution for medical devices must protect firmware from tampering, secure the data stored by the device, secure communication, and protect the device from cyberattacks. This can only be achieved by building in security from the early stages of design. There is no “one-size-fits-all security solution” for medical devices. PEDIATRIC MEDICAL DEVICES ADDRESS COVID-19, 16 COMPANIES In addition to its annual “Make Your Medical Device Pitch for Kids!” competition, which focuses on innovation in the pediatric space, this year the National Capital Consortium for Pediatric Device Innovation (NCC-PDI) launched a COVID-19 edition for 2020. Although COVID-19 has not been affecting the pediatric population as much as other age groups, it is remains a serious threat to children. FIVE STEPS TO MEDICAL DEVICE COMMERCIALIZATION EU MDR COST OF COMPLIANCE: THE RESULTS ARE IN 32% of respondents expect compliance with EU MDR will take up more than 8% of their company’s EU revenue. An estimate of the cost of compliance connected to global revenue was much smaller. Continue to the rest of the survey results on page 2. Pages: 1 2. RECENT FDA ACTION: THREE RECALLS AND TEN MEDICAL DEVICE Recent FDA Action: Three Recalls and Ten Medical Device Approvals. The device approvals address critical patient need. FDA recently released a list of medical devices it has approved and recalled over the past few months. All of the recalls were Class I recalls. MEDICAL DEVICE REGULATION With the application date fast approaching, gain insights into the new MDR requirements. Following the webinar, you will be better prepared to meet the increased requirements that have been placed on medical device manufactures. You will gain an understanding of your next steps, whether it is additional MDR transition training, or what to discuss with your notified body. SOURCING IN ASIA: PARTNER WITH THE RIGHT SUPPLIER FROM THE Most international medical device companies work with Asian distributors to sell their products in the local marketplace. Finding, qualifying, and signing up the right distributors is an important job since these distributors will sell your products locally. WHEN COMPLAINT INVESTIGATIONS MEET SERVICING AND REPAIRING When Complaint Investigations Meet Servicing and Repairing Devices. Many companies are unsure of how to complete a complaint investigation when the service and repair process is involved. Manufacturers of medical devices must often service and repair products once they are out in the field. If these device are involved in a future complaint HOME | MEDTECH INTELLIGENCEBUSINESS ANALYSISCOMBINATION PRODUCTSMARKET ACCESSOPERATIONSPRODUCT DEVELOPMENT The Transformation to True Digital Healthcare. By Stephen Bernstein, Dana Safran, Jody Tropeano, Lisa Schmitz Mazur, Sara Vaezy, Tom Kiesau. Digital health tools and technology have the potential to dramatically improve the patient care experience, but the shift requires purposeful coordination and planned architecture. April 28,2021.
IN 2020, FDA AUTHORIZED 132 NOVEL MEDICAL DEVICES In 2020, FDA Authorized 132 Novel Medical Devices. The record-high number of authorizations last year was a significant jump over 2019. In 2009, FDA authorized just 29 novel devices; in 2015 it authorized 86; and five years later the number hit 132 novel devices. 2020 was a year for innovation, but the COVID-19 pandemic was only partially SECURING MEDICAL DEVICES: WHAT IS REALLY NEEDED? A security solution for medical devices must protect firmware from tampering, secure the data stored by the device, secure communication, and protect the device from cyberattacks. This can only be achieved by building in security from the early stages of design. There is no “one-size-fits-all security solution” for medical devices. PEDIATRIC MEDICAL DEVICES ADDRESS COVID-19, 16 COMPANIES In addition to its annual “Make Your Medical Device Pitch for Kids!” competition, which focuses on innovation in the pediatric space, this year the National Capital Consortium for Pediatric Device Innovation (NCC-PDI) launched a COVID-19 edition for 2020. Although COVID-19 has not been affecting the pediatric population as much as other age groups, it is remains a serious threat to children. FIVE STEPS TO MEDICAL DEVICE COMMERCIALIZATION EU MDR COST OF COMPLIANCE: THE RESULTS ARE IN 32% of respondents expect compliance with EU MDR will take up more than 8% of their company’s EU revenue. An estimate of the cost of compliance connected to global revenue was much smaller. Continue to the rest of the survey results on page 2. Pages: 1 2. RECENT FDA ACTION: THREE RECALLS AND TEN MEDICAL DEVICE Recent FDA Action: Three Recalls and Ten Medical Device Approvals. The device approvals address critical patient need. FDA recently released a list of medical devices it has approved and recalled over the past few months. All of the recalls were Class I recalls. MEDICAL DEVICE REGULATION With the application date fast approaching, gain insights into the new MDR requirements. Following the webinar, you will be better prepared to meet the increased requirements that have been placed on medical device manufactures. You will gain an understanding of your next steps, whether it is additional MDR transition training, or what to discuss with your notified body. SOURCING IN ASIA: PARTNER WITH THE RIGHT SUPPLIER FROM THE Most international medical device companies work with Asian distributors to sell their products in the local marketplace. Finding, qualifying, and signing up the right distributors is an important job since these distributors will sell your products locally. WHEN COMPLAINT INVESTIGATIONS MEET SERVICING AND REPAIRING When Complaint Investigations Meet Servicing and Repairing Devices. Many companies are unsure of how to complete a complaint investigation when the service and repair process is involved. Manufacturers of medical devices must often service and repair products once they are out in the field. If these device are involved in a future complaint COLUMN | REMOTE AUDITS AND OTHER NEW WAYS OF WORKING IN A Audits for medical device companies and other organizations in the medtech space used to be primarily in person. However, the pandemic magnified flaws in the global regulatory process. A shift to remote audits may be the key to interorganizational and REDUCE YOUR MEDICAL DEVICE TIME-TO-MARKET WITH CPQ Specifically designed for the Medical and Mobility industry’s complex and custom products, discover how CPQ (configure, price, quote) software can help your company save money, increase customer satisfaction, and generate real ROI. EMERGING TRENDS IN CARDIOVASCULAR DEVICES Cardiac ablation devices accounted for almost 72% of the cardiovascular devices market share in 2020 and are expected to gain massive momentum and, as per GMI estimates. The rising prevalence of atrial fibrillation is a major contributor to the popularity of these devices. The products are addressing an ever-increasing demand forminimally
BIOLOGICAL EVALUATION OF MEDICAL DEVICES Biological Evaluation of Medical Devices. The biological safety of a device is established for both short-term (acute) and longtime (chronic) safety risks to patients and device users. Medical device manufacturers must investigate their devices thoroughly to meet these expectations to receive approval to market their devices. MEDTECH ECONOMICS ARCHIVES A strategy that pulls in regulatory, quality, clinical, reimbursement, marketing, compliance, legal and finance is required. Continue reading→
BUILD AN EFFECTIVE GLOBAL REGULATORY STRATEGY Global Medical Device Markets. According to the U.S. Department of Commerce, the medical device marketplace worldwide is expected to experience a 5% annual growth rate over the next few years. By the year 2020, the total market is expected to reach approximately $440 billion. The U.S. accounts for 45% of this market share. RECENT FDA ACTION: THREE RECALLS AND TEN MEDICAL DEVICE Recent FDA Action: Three Recalls and Ten Medical Device Approvals. The device approvals address critical patient need. FDA recently released a list of medical devices it has approved and recalled over the past few months. All of the recalls were Class I recalls. EIGHT STEPS TO POST-MARKET SURVEILLANCE COMPLIANCE Follow these eight key steps to stay on top of post-market surveillance and navigate the changing regulatory climate requirements. 1. Stay Ahead of the Game. Manufacturers should not rely solely on waiting to be informed that significant regulatory changes have been made. Manufacturers should take a proactive approach when itcomes to post
WHAT INDUSTRY IS SAYING ABOUT PROPOSED ETHYLENE OXIDE More than 20 billion medical devices sold in the United States each year are sterilized using ethylene oxide (EtO), according to an agency statement from then-FDA Acting Commissioner Ned Sharpless, M.D. on October 25. EtO is the most common method of sterilization in the country, and as such, the closures of EtO facilities across the country are raising strong concerns among stakeholders. WHEN COMPLAINT INVESTIGATIONS MEET SERVICING AND REPAIRING When Complaint Investigations Meet Servicing and Repairing Devices. Many companies are unsure of how to complete a complaint investigation when the service and repair process is involved. Manufacturers of medical devices must often service and repair products once they are out in the field. If these device are involved in a future complaint HOME | MEDTECH INTELLIGENCEBUSINESS ANALYSISCOMBINATION PRODUCTSMARKET ACCESSOPERATIONSPRODUCT DEVELOPMENT The Transformation to True Digital Healthcare. By Stephen Bernstein, Dana Safran, Jody Tropeano, Lisa Schmitz Mazur, Sara Vaezy, Tom Kiesau. Digital health tools and technology have the potential to dramatically improve the patient care experience, but the shift requires purposeful coordination and planned architecture. April 28,2021.
IN 2020, FDA AUTHORIZED 132 NOVEL MEDICAL DEVICES In 2020, FDA Authorized 132 Novel Medical Devices. The record-high number of authorizations last year was a significant jump over 2019. In 2009, FDA authorized just 29 novel devices; in 2015 it authorized 86; and five years later the number hit 132 novel devices. 2020 was a year for innovation, but the COVID-19 pandemic was only partially EMERGING TRENDS IN CARDIOVASCULAR DEVICES Cardiac ablation devices accounted for almost 72% of the cardiovascular devices market share in 2020 and are expected to gain massive momentum and, as per GMI estimates. The rising prevalence of atrial fibrillation is a major contributor to the popularity of these devices. The products are addressing an ever-increasing demand forminimally
AI IN MEDTECH: RISKS AND OPPORTUNITIES OF INNOVATIVE Dr. Abtin Rad has 13 years of professional experience as a Biomedical and electrical Engineer with focus on Software, Cybersecurity and Artificial Intelligence, as well as medical Imaging and Therapy using Ultrasound and optical laser systems. SECURING MEDICAL DEVICES: WHAT IS REALLY NEEDED? A security solution for medical devices must protect firmware from tampering, secure the data stored by the device, secure communication, and protect the device from cyberattacks. This can only be achieved by building in security from the early stages of design. There is no “one-size-fits-all security solution” for medical devices. FIVE STEPS TO MEDICAL DEVICE COMMERCIALIZATION PRESIDENT BIDEN NOMINATES CHIQUITA BROOKS-LASURE TO CMS Last week President Biden nominated Chiquita Brooks-LaSure to the position of CMS Administrator.Currently a managing director at Manatt Health, Brooks-LaSure has more than 20 years of health policy experience, and has directly worked in CMS as deputy director for policy at the Center for Consumer Information and Insurance Oversight, and as director of coverage policy at HHS. HOW DO PROVIDERS HANDLE DEVICE RECALLS? Solution: Include the following information in every recall notice: Affected Product. Item and lot number. The problem. Date range for when the product was manufactured and distributed. Whether a replacement product is available. Issue #3: Clearly stating whether an alert is a broad notification or it directly applies to the hospital. HOT TOPICS IN RISK MANAGEMENT & MEDICAL DEVICES Industry stakeholders, including FDA officials, will discuss recalls and other key topics at MedTech Intelligence’s HHE and Recalls/Global Recalls conference, May 10–12, 2016 in Washington, DC Roberta Goode, president & CEO of Goode Compliance International, weighs in on the latest challenges medical device manufacturers are facing in risk management at the 2015 MedTech Intelligence HHE MOST SERIOUS MEDICAL DEVICE RECALLS OF 2015 FDA initiates Class I recalls when use of a device can lead to severe or life-threatening injuries, or death. HeartWare topped the list with five Class I recalls, while Teleflex Medical had four, and Boston Scientific, Covidien, and Insulet Corp. tied with two recalls each. One of the most serious recalls in 2015 involved HeartWare’s Heart HOME | MEDTECH INTELLIGENCEBUSINESS ANALYSISCOMBINATION PRODUCTSMARKET ACCESSOPERATIONSPRODUCT DEVELOPMENT The Transformation to True Digital Healthcare. By Stephen Bernstein, Dana Safran, Jody Tropeano, Lisa Schmitz Mazur, Sara Vaezy, Tom Kiesau. Digital health tools and technology have the potential to dramatically improve the patient care experience, but the shift requires purposeful coordination and planned architecture. April 28,2021.
IN 2020, FDA AUTHORIZED 132 NOVEL MEDICAL DEVICES In 2020, FDA Authorized 132 Novel Medical Devices. The record-high number of authorizations last year was a significant jump over 2019. In 2009, FDA authorized just 29 novel devices; in 2015 it authorized 86; and five years later the number hit 132 novel devices. 2020 was a year for innovation, but the COVID-19 pandemic was only partially EMERGING TRENDS IN CARDIOVASCULAR DEVICES Cardiac ablation devices accounted for almost 72% of the cardiovascular devices market share in 2020 and are expected to gain massive momentum and, as per GMI estimates. The rising prevalence of atrial fibrillation is a major contributor to the popularity of these devices. The products are addressing an ever-increasing demand forminimally
AI IN MEDTECH: RISKS AND OPPORTUNITIES OF INNOVATIVE Dr. Abtin Rad has 13 years of professional experience as a Biomedical and electrical Engineer with focus on Software, Cybersecurity and Artificial Intelligence, as well as medical Imaging and Therapy using Ultrasound and optical laser systems. SECURING MEDICAL DEVICES: WHAT IS REALLY NEEDED? A security solution for medical devices must protect firmware from tampering, secure the data stored by the device, secure communication, and protect the device from cyberattacks. This can only be achieved by building in security from the early stages of design. There is no “one-size-fits-all security solution” for medical devices. FIVE STEPS TO MEDICAL DEVICE COMMERCIALIZATION PRESIDENT BIDEN NOMINATES CHIQUITA BROOKS-LASURE TO CMS Last week President Biden nominated Chiquita Brooks-LaSure to the position of CMS Administrator.Currently a managing director at Manatt Health, Brooks-LaSure has more than 20 years of health policy experience, and has directly worked in CMS as deputy director for policy at the Center for Consumer Information and Insurance Oversight, and as director of coverage policy at HHS. HOW DO PROVIDERS HANDLE DEVICE RECALLS? Solution: Include the following information in every recall notice: Affected Product. Item and lot number. The problem. Date range for when the product was manufactured and distributed. Whether a replacement product is available. Issue #3: Clearly stating whether an alert is a broad notification or it directly applies to the hospital. HOT TOPICS IN RISK MANAGEMENT & MEDICAL DEVICES Industry stakeholders, including FDA officials, will discuss recalls and other key topics at MedTech Intelligence’s HHE and Recalls/Global Recalls conference, May 10–12, 2016 in Washington, DC Roberta Goode, president & CEO of Goode Compliance International, weighs in on the latest challenges medical device manufacturers are facing in risk management at the 2015 MedTech Intelligence HHE MOST SERIOUS MEDICAL DEVICE RECALLS OF 2015 FDA initiates Class I recalls when use of a device can lead to severe or life-threatening injuries, or death. HeartWare topped the list with five Class I recalls, while Teleflex Medical had four, and Boston Scientific, Covidien, and Insulet Corp. tied with two recalls each. One of the most serious recalls in 2015 involved HeartWare’s Heart COLUMN | REMOTE AUDITS AND OTHER NEW WAYS OF WORKING IN A Audits for medical device companies and other organizations in the medtech space used to be primarily in person. However, the pandemic magnified flaws in the global regulatory process. A shift to remote audits may be the key to interorganizational and REPROCESSED MEDICAL DEVICES: EASING THE COST OF HEALTHCARE The utilization of reprocessed medical devices can help cut down the increasing cost of healthcare for consumers while also tackling device shortages. Reprocessing of medical devices and equipment has gained a lot of attention, not just across hospitals saving AI IN MEDTECH: RISKS AND OPPORTUNITIES OF INNOVATIVE Dr. Abtin Rad has 13 years of professional experience as a Biomedical and electrical Engineer with focus on Software, Cybersecurity and Artificial Intelligence, as well as medical Imaging and Therapy using Ultrasound and optical laser systems. TRENDS IN MEDICAL DEVICE RECALLS Since the FDA last issued its Medical Device Recall Report covering FY 2003 to FY 2012, changes in the medical device industry have shifted the nature of medical device recalls. 1 Our team’s goal was to analyze U.S. medical device recalls from 2013 to 2018 to provide an update on notable trends and lessons that medical device firms can incorporate into their quality and risk management systems. BUILD AN EFFECTIVE GLOBAL REGULATORY STRATEGY Global Medical Device Markets. According to the U.S. Department of Commerce, the medical device marketplace worldwide is expected to experience a 5% annual growth rate over the next few years. By the year 2020, the total market is expected to reach approximately $440 billion. The U.S. accounts for 45% of this market share. THE ERA OF THE ROBOT NURSE The Era of the Robot Nurse. It’s no longer science fiction. Healthcare and technology are two industries that have always gone hand in hand. Healthcare professionals are able to do their jobs more efficiently, effectively and safety thanks to innovations in medical technology. These days, one of the biggest emerging technology sectorsin
QUALITY/REGULATORY ARCHIVES Welcome back to this sixth installment of Devine Guidance . I am not one to assume a Panglossian view (time to pull out the dictionary again) when dealing with problem suppliers and the need to pursuecorrective action.
INNOVATIONS IN SELF-MONITORING BLOOD GLUCOSE DEVICES WILL A meteoric rise in the diabetic population has made blood glucose monitoring technology a global research topic. It has subsequently sent self-monitoring technologies mainstream, as they proved to be pragmatic and convenient. THE INTEGRATION OF COMPLAINT HANDLING AND RISK MANAGEMENT The Integration of Complaint Handling and Risk Management. A review of the important aspects of risk management and post-market surveillance processes, and how to resolve common concerns. In another first for warning letters, complaint handling supplanted CAPA as the violation found most in letters. 1 Medical device manufacturers often struggle PREPARING FOR MDR: DON’T FORGET ABOUT CLASS I REUSABLE Under MDR, there’s a new subclass for Class I reusable devices (Class Ir), such as surgical instruments and endoscopes, putting such devices under a higher level of scrutiny and introducing new regulatory oversight. Extensions won’t be granted for Class Ir, and grandfathering in legacy devices will be strictly prohibited. HOME | MEDTECH INTELLIGENCEBUSINESS ANALYSISCOMBINATION PRODUCTSMARKET ACCESSOPERATIONSPRODUCT DEVELOPMENT The Transformation to True Digital Healthcare. By Stephen Bernstein, Dana Safran, Jody Tropeano, Lisa Schmitz Mazur, Sara Vaezy, Tom Kiesau. Digital health tools and technology have the potential to dramatically improve the patient care experience, but the shift requires purposeful coordination and planned architecture. April 28,2021.
EMERGING TRENDS IN CARDIOVASCULAR DEVICES Cardiac ablation devices accounted for almost 72% of the cardiovascular devices market share in 2020 and are expected to gain massive momentum and, as per GMI estimates. The rising prevalence of atrial fibrillation is a major contributor to the popularity of these devices. The products are addressing an ever-increasing demand forminimally
IN 2020, FDA AUTHORIZED 132 NOVEL MEDICAL DEVICES In 2020, FDA Authorized 132 Novel Medical Devices. The record-high number of authorizations last year was a significant jump over 2019. In 2009, FDA authorized just 29 novel devices; in 2015 it authorized 86; and five years later the number hit 132 novel devices. 2020 was a year for innovation, but the COVID-19 pandemic was only partially AI IN MEDTECH: RISKS AND OPPORTUNITIES OF INNOVATIVE Dr. Abtin Rad has 13 years of professional experience as a Biomedical and electrical Engineer with focus on Software, Cybersecurity and Artificial Intelligence, as well as medical Imaging and Therapy using Ultrasound and optical laser systems. FIVE STEPS TO MEDICAL DEVICE COMMERCIALIZATION SECURING MEDICAL DEVICES: WHAT IS REALLY NEEDED? A security solution for medical devices must protect firmware from tampering, secure the data stored by the device, secure communication, and protect the device from cyberattacks. This can only be achieved by building in security from the early stages of design. There is no “one-size-fits-all security solution” for medical devices. THE ERA OF THE ROBOT NURSE The Era of the Robot Nurse. It’s no longer science fiction. Healthcare and technology are two industries that have always gone hand in hand. Healthcare professionals are able to do their jobs more efficiently, effectively and safety thanks to innovations in medical technology. These days, one of the biggest emerging technology sectorsin
PRESIDENT BIDEN NOMINATES CHIQUITA BROOKS-LASURE TO CMS Last week President Biden nominated Chiquita Brooks-LaSure to the position of CMS Administrator.Currently a managing director at Manatt Health, Brooks-LaSure has more than 20 years of health policy experience, and has directly worked in CMS as deputy director for policy at the Center for Consumer Information and Insurance Oversight, and as director of coverage policy at HHS. HOW DO PROVIDERS HANDLE DEVICE RECALLS? Solution: Include the following information in every recall notice: Affected Product. Item and lot number. The problem. Date range for when the product was manufactured and distributed. Whether a replacement product is available. Issue #3: Clearly stating whether an alert is a broad notification or it directly applies to the hospital. SOURCING IN ASIA: PARTNER WITH THE RIGHT SUPPLIER FROM THE Most international medical device companies work with Asian distributors to sell their products in the local marketplace. Finding, qualifying, and signing up the right distributors is an important job since these distributors will sell your products locally. HOME | MEDTECH INTELLIGENCEBUSINESS ANALYSISCOMBINATION PRODUCTSMARKET ACCESSOPERATIONSPRODUCT DEVELOPMENT The Transformation to True Digital Healthcare. By Stephen Bernstein, Dana Safran, Jody Tropeano, Lisa Schmitz Mazur, Sara Vaezy, Tom Kiesau. Digital health tools and technology have the potential to dramatically improve the patient care experience, but the shift requires purposeful coordination and planned architecture. April 28,2021.
EMERGING TRENDS IN CARDIOVASCULAR DEVICES Cardiac ablation devices accounted for almost 72% of the cardiovascular devices market share in 2020 and are expected to gain massive momentum and, as per GMI estimates. The rising prevalence of atrial fibrillation is a major contributor to the popularity of these devices. The products are addressing an ever-increasing demand forminimally
IN 2020, FDA AUTHORIZED 132 NOVEL MEDICAL DEVICES In 2020, FDA Authorized 132 Novel Medical Devices. The record-high number of authorizations last year was a significant jump over 2019. In 2009, FDA authorized just 29 novel devices; in 2015 it authorized 86; and five years later the number hit 132 novel devices. 2020 was a year for innovation, but the COVID-19 pandemic was only partially AI IN MEDTECH: RISKS AND OPPORTUNITIES OF INNOVATIVE Dr. Abtin Rad has 13 years of professional experience as a Biomedical and electrical Engineer with focus on Software, Cybersecurity and Artificial Intelligence, as well as medical Imaging and Therapy using Ultrasound and optical laser systems. FIVE STEPS TO MEDICAL DEVICE COMMERCIALIZATION SECURING MEDICAL DEVICES: WHAT IS REALLY NEEDED? A security solution for medical devices must protect firmware from tampering, secure the data stored by the device, secure communication, and protect the device from cyberattacks. This can only be achieved by building in security from the early stages of design. There is no “one-size-fits-all security solution” for medical devices. THE ERA OF THE ROBOT NURSE The Era of the Robot Nurse. It’s no longer science fiction. Healthcare and technology are two industries that have always gone hand in hand. Healthcare professionals are able to do their jobs more efficiently, effectively and safety thanks to innovations in medical technology. These days, one of the biggest emerging technology sectorsin
PRESIDENT BIDEN NOMINATES CHIQUITA BROOKS-LASURE TO CMS Last week President Biden nominated Chiquita Brooks-LaSure to the position of CMS Administrator.Currently a managing director at Manatt Health, Brooks-LaSure has more than 20 years of health policy experience, and has directly worked in CMS as deputy director for policy at the Center for Consumer Information and Insurance Oversight, and as director of coverage policy at HHS. HOW DO PROVIDERS HANDLE DEVICE RECALLS? Solution: Include the following information in every recall notice: Affected Product. Item and lot number. The problem. Date range for when the product was manufactured and distributed. Whether a replacement product is available. Issue #3: Clearly stating whether an alert is a broad notification or it directly applies to the hospital. SOURCING IN ASIA: PARTNER WITH THE RIGHT SUPPLIER FROM THE Most international medical device companies work with Asian distributors to sell their products in the local marketplace. Finding, qualifying, and signing up the right distributors is an important job since these distributors will sell your products locally. COLUMN | REMOTE AUDITS AND OTHER NEW WAYS OF WORKING IN A Audits for medical device companies and other organizations in the medtech space used to be primarily in person. However, the pandemic magnified flaws in the global regulatory process. A shift to remote audits may be the key to interorganizational and REPROCESSED MEDICAL DEVICES: EASING THE COST OF HEALTHCARE The utilization of reprocessed medical devices can help cut down the increasing cost of healthcare for consumers while also tackling device shortages. Reprocessing of medical devices and equipment has gained a lot of attention, not just across hospitals saving BUILD AN EFFECTIVE GLOBAL REGULATORY STRATEGY Global Medical Device Markets. According to the U.S. Department of Commerce, the medical device marketplace worldwide is expected to experience a 5% annual growth rate over the next few years. By the year 2020, the total market is expected to reach approximately $440 billion. The U.S. accounts for 45% of this market share. TRENDS IN MEDICAL DEVICE RECALLS Since the FDA last issued its Medical Device Recall Report covering FY 2003 to FY 2012, changes in the medical device industry have shifted the nature of medical device recalls. 1 Our team’s goal was to analyze U.S. medical device recalls from 2013 to 2018 to provide an update on notable trends and lessons that medical device firms can incorporate into their quality and risk management systems. TECH Q&A: ARTIFICIAL INTELLIGENCE HAS PROMISE OF Maria Fontanazza has more than 15 years of experience in journalism, marketing and communications. She was previously marketing communications manager and market research manager at Secant Medical, Inc., a manufacturer of biomedical textiles and advanced biomaterials. THE ERA OF THE ROBOT NURSE The Era of the Robot Nurse. It’s no longer science fiction. Healthcare and technology are two industries that have always gone hand in hand. Healthcare professionals are able to do their jobs more efficiently, effectively and safety thanks to innovations in medical technology. These days, one of the biggest emerging technology sectorsin
QUALITY/REGULATORY ARCHIVES Welcome back to this sixth installment of Devine Guidance . I am not one to assume a Panglossian view (time to pull out the dictionary again) when dealing with problem suppliers and the need to pursuecorrective action.
THE INTEGRATION OF COMPLAINT HANDLING AND RISK MANAGEMENT The Integration of Complaint Handling and Risk Management. A review of the important aspects of risk management and post-market surveillance processes, and how to resolve common concerns. In another first for warning letters, complaint handling supplanted CAPA as the violation found most in letters. 1 Medical device manufacturers often struggle INTEGRATING DATA SCIENCE INTO MEDICAL DEVICE DEVELOPMENT The FDA’s approval of an electrocardiogram (EKG) that enables atrial fibrillation detection right from a patient’s watch band is just one example of how the digitization of medical devices, a part of the Internet of Things movement, is leading product development and innovation in medicine. However, while medical devices built on a connected services platform include components for data PREPARING FOR MDR: DON’T FORGET ABOUT CLASS I REUSABLE Under MDR, there’s a new subclass for Class I reusable devices (Class Ir), such as surgical instruments and endoscopes, putting such devices under a higher level of scrutiny and introducing new regulatory oversight. Extensions won’t be granted for Class Ir, and grandfathering in legacy devices will be strictly prohibited. HOME | MEDTECH INTELLIGENCEBUSINESS ANALYSISCOMBINATION PRODUCTSMARKET ACCESSOPERATIONSPRODUCT DEVELOPMENT The Transformation to True Digital Healthcare. By Stephen Bernstein, Dana Safran, Jody Tropeano, Lisa Schmitz Mazur, Sara Vaezy, Tom Kiesau. Digital health tools and technology have the potential to dramatically improve the patient care experience, but the shift requires purposeful coordination and planned architecture. April 28,2021.
EMERGING TRENDS IN CARDIOVASCULAR DEVICES Cardiac ablation devices accounted for almost 72% of the cardiovascular devices market share in 2020 and are expected to gain massive momentum and, as per GMI estimates. The rising prevalence of atrial fibrillation is a major contributor to the popularity of these devices. The products are addressing an ever-increasing demand forminimally
IN 2020, FDA AUTHORIZED 132 NOVEL MEDICAL DEVICES In 2020, FDA Authorized 132 Novel Medical Devices. The record-high number of authorizations last year was a significant jump over 2019. In 2009, FDA authorized just 29 novel devices; in 2015 it authorized 86; and five years later the number hit 132 novel devices. 2020 was a year for innovation, but the COVID-19 pandemic was only partially FIVE STEPS TO MEDICAL DEVICE COMMERCIALIZATION PEDIATRIC MEDICAL DEVICES ADDRESS COVID-19, 16 COMPANIES In addition to its annual “Make Your Medical Device Pitch for Kids!” competition, which focuses on innovation in the pediatric space, this year the National Capital Consortium for Pediatric Device Innovation (NCC-PDI) launched a COVID-19 edition for 2020. Although COVID-19 has not been affecting the pediatric population as much as other age groups, it is remains a serious threat to children. SECURING MEDICAL DEVICES: WHAT IS REALLY NEEDED? A security solution for medical devices must protect firmware from tampering, secure the data stored by the device, secure communication, and protect the device from cyberattacks. This can only be achieved by building in security from the early stages of design. There is no “one-size-fits-all security solution” for medical devices. HOT TOPICS IN RISK MANAGEMENT & MEDICAL DEVICES Industry stakeholders, including FDA officials, will discuss recalls and other key topics at MedTech Intelligence’s HHE and Recalls/Global Recalls conference, May 10–12, 2016 in Washington, DC Roberta Goode, president & CEO of Goode Compliance International, weighs in on the latest challenges medical device manufacturers are facing in risk management at the 2015 MedTech Intelligence HHE INTEGRATING DATA SCIENCE INTO MEDICAL DEVICE DEVELOPMENT The FDA’s approval of an electrocardiogram (EKG) that enables atrial fibrillation detection right from a patient’s watch band is just one example of how the digitization of medical devices, a part of the Internet of Things movement, is leading product development and innovation in medicine. However, while medical devices built on a connected services platform include components for data RECENT FDA ACTION: THREE RECALLS AND TEN MEDICAL DEVICE Recent FDA Action: Three Recalls and Ten Medical Device Approvals. The device approvals address critical patient need. FDA recently released a list of medical devices it has approved and recalled over the past few months. All of the recalls were Class I recalls. EU MDR COST OF COMPLIANCE: THE RESULTS ARE IN 32% of respondents expect compliance with EU MDR will take up more than 8% of their company’s EU revenue. An estimate of the cost of compliance connected to global revenue was much smaller. Continue to the rest of the survey results on page 2. Pages: 1 2. HOME | MEDTECH INTELLIGENCEBUSINESS ANALYSISCOMBINATION PRODUCTSMARKET ACCESSOPERATIONSPRODUCT DEVELOPMENT The Transformation to True Digital Healthcare. By Stephen Bernstein, Dana Safran, Jody Tropeano, Lisa Schmitz Mazur, Sara Vaezy, Tom Kiesau. Digital health tools and technology have the potential to dramatically improve the patient care experience, but the shift requires purposeful coordination and planned architecture. April 28,2021.
EMERGING TRENDS IN CARDIOVASCULAR DEVICES Cardiac ablation devices accounted for almost 72% of the cardiovascular devices market share in 2020 and are expected to gain massive momentum and, as per GMI estimates. The rising prevalence of atrial fibrillation is a major contributor to the popularity of these devices. The products are addressing an ever-increasing demand forminimally
IN 2020, FDA AUTHORIZED 132 NOVEL MEDICAL DEVICES In 2020, FDA Authorized 132 Novel Medical Devices. The record-high number of authorizations last year was a significant jump over 2019. In 2009, FDA authorized just 29 novel devices; in 2015 it authorized 86; and five years later the number hit 132 novel devices. 2020 was a year for innovation, but the COVID-19 pandemic was only partially FIVE STEPS TO MEDICAL DEVICE COMMERCIALIZATION PEDIATRIC MEDICAL DEVICES ADDRESS COVID-19, 16 COMPANIES In addition to its annual “Make Your Medical Device Pitch for Kids!” competition, which focuses on innovation in the pediatric space, this year the National Capital Consortium for Pediatric Device Innovation (NCC-PDI) launched a COVID-19 edition for 2020. Although COVID-19 has not been affecting the pediatric population as much as other age groups, it is remains a serious threat to children. SECURING MEDICAL DEVICES: WHAT IS REALLY NEEDED? A security solution for medical devices must protect firmware from tampering, secure the data stored by the device, secure communication, and protect the device from cyberattacks. This can only be achieved by building in security from the early stages of design. There is no “one-size-fits-all security solution” for medical devices. HOT TOPICS IN RISK MANAGEMENT & MEDICAL DEVICES Industry stakeholders, including FDA officials, will discuss recalls and other key topics at MedTech Intelligence’s HHE and Recalls/Global Recalls conference, May 10–12, 2016 in Washington, DC Roberta Goode, president & CEO of Goode Compliance International, weighs in on the latest challenges medical device manufacturers are facing in risk management at the 2015 MedTech Intelligence HHE INTEGRATING DATA SCIENCE INTO MEDICAL DEVICE DEVELOPMENT The FDA’s approval of an electrocardiogram (EKG) that enables atrial fibrillation detection right from a patient’s watch band is just one example of how the digitization of medical devices, a part of the Internet of Things movement, is leading product development and innovation in medicine. However, while medical devices built on a connected services platform include components for data RECENT FDA ACTION: THREE RECALLS AND TEN MEDICAL DEVICE Recent FDA Action: Three Recalls and Ten Medical Device Approvals. The device approvals address critical patient need. FDA recently released a list of medical devices it has approved and recalled over the past few months. All of the recalls were Class I recalls. EU MDR COST OF COMPLIANCE: THE RESULTS ARE IN 32% of respondents expect compliance with EU MDR will take up more than 8% of their company’s EU revenue. An estimate of the cost of compliance connected to global revenue was much smaller. Continue to the rest of the survey results on page 2. Pages: 1 2. COLUMN | REMOTE AUDITS AND OTHER NEW WAYS OF WORKING IN A 18 hours ago · Audits for medical device companies and other organizations in the medtech space used to be primarily in person. However, the pandemic magnified flaws in the global regulatory process. A shift to remote audits may be the key tointerorganizational
IN 2020, FDA AUTHORIZED 132 NOVEL MEDICAL DEVICES In 2020, FDA Authorized 132 Novel Medical Devices. The record-high number of authorizations last year was a significant jump over 2019. In 2009, FDA authorized just 29 novel devices; in 2015 it authorized 86; and five years later the number hit 132 novel devices. 2020 was a year for innovation, but the COVID-19 pandemic was only partially REPROCESSED MEDICAL DEVICES: EASING THE COST OF HEALTHCARE The utilization of reprocessed medical devices can help cut down the increasing cost of healthcare for consumers while also tackling device shortages. Reprocessing of medical devices and equipment has gained a lot of attention, not just across hospitals saving TRENDS IN MEDICAL DEVICE RECALLS Since the FDA last issued its Medical Device Recall Report covering FY 2003 to FY 2012, changes in the medical device industry have shifted the nature of medical device recalls. 1 Our team’s goal was to analyze U.S. medical device recalls from 2013 to 2018 to provide an update on notable trends and lessons that medical device firms can incorporate into their quality and risk management systems. SECURING MEDICAL DEVICES: WHAT IS REALLY NEEDED? A security solution for medical devices must protect firmware from tampering, secure the data stored by the device, secure communication, and protect the device from cyberattacks. This can only be achieved by building in security from the early stages of design. There is no “one-size-fits-all security solution” for medical devices. BUILD AN EFFECTIVE GLOBAL REGULATORY STRATEGY Global Medical Device Markets. According to the U.S. Department of Commerce, the medical device marketplace worldwide is expected to experience a 5% annual growth rate over the next few years. By the year 2020, the total market is expected to reach approximately $440 billion. The U.S. accounts for 45% of this market share. QUALITY CONSIDERATIONS FOR DRUG-DEVICE COMBINATION Quality Considerations for Drug-Device Combination Products. Understanding the GMP requirements of each component is critical to product and compliance success. The rapid development of combination products—biologics, pharmaceuticals and their devices—provides great opportunity for technological advancement, opening new doors forpatient
EMPOWERING EMERGING MEDICAL DEVICE MANUFACTURERS This eBook reviews the industry trends in MDD and the unique challenges facing SMB. The requirements for a digital infrastructure that addresses those challenges are detailed, and the Siemens solution developed according to those requirements is explained. MEDTECH ECONOMICS ARCHIVES With an established base of medtech companies and a strong workforce, Ireland remains a key player in the global supply chain. Continuereading →
QUALITY/REGULATORY ARCHIVES Welcome back to this sixth installment of Devine Guidance . I am not one to assume a Panglossian view (time to pull out the dictionary again) when dealing with problem suppliers and the need to pursuecorrective action.
HOME | MEDTECH INTELLIGENCEBUSINESS ANALYSISCOMBINATION PRODUCTSMARKET ACCESSOPERATIONSPRODUCT DEVELOPMENT The Transformation to True Digital Healthcare. By Stephen Bernstein, Dana Safran, Jody Tropeano, Lisa Schmitz Mazur, Sara Vaezy, Tom Kiesau. Digital health tools and technology have the potential to dramatically improve the patient care experience, but the shift requires purposeful coordination and planned architecture. April 28,2021.
QUALITY CONSIDERATIONS FOR DRUG-DEVICE COMBINATION Quality Considerations for Drug-Device Combination Products. Understanding the GMP requirements of each component is critical to product and compliance success. The rapid development of combination products—biologics, pharmaceuticals and their devices—provides great opportunity for technological advancement, opening new doors forpatient
FIVE STEPS TO MEDICAL DEVICE COMMERCIALIZATION TRENDS IN MEDICAL DEVICE RECALLS TRENDS IN MEDICAL DEVICE PURCHASING, EVALUATION OF VALUE This website uses cookies so that we can provide you with the best user experience possible. Cookie information is stored in your browser and performs functions such as recognising you when you return to our website and helping our team to understand which DEVELOPING MEDICAL DEVICES FOR DRUGS AND ALCOHOL The Bridge is a small device, around two inches wide. It has electrodes attached inside the ear. The electrodes target cranial nerves that cause withdrawal effects. It is a disposable unit and is only used once. Most patients use it for the first five days of detox, or for the entire detox treatment duration. WHAT INDUSTRY IS SAYING ABOUT PROPOSED ETHYLENE OXIDE More than 20 billion medical devices sold in the United States each year are sterilized using ethylene oxide (EtO), according to an agency statement from then-FDA Acting Commissioner Ned Sharpless, M.D. on October 25. EtO is the most common method of sterilization in the country, and as such, the closures of EtO facilities across the country are raising strong concerns among stakeholders. INTEGRATING DATA SCIENCE INTO MEDICAL DEVICE DEVELOPMENT The FDA’s approval of an electrocardiogram (EKG) that enables atrial fibrillation detection right from a patient’s watch band is just one example of how the digitization of medical devices, a part of the Internet of Things movement, is leading product development and innovation in medicine. However, while medical devices built on a connected services platform include components for data APPLE WATCH 4 GETS FDA CLEARANCE Apple Watch 4 Gets FDA Clearance. The product can conduct electrocardiograms and measure the heart’s rhythms. In addition to all the bells and whistles that come with an Apple Watch, the Series 4 product has received FDA clearance—specifically De Novo classification. Unveiled at an Apple event last week, the Series 4 watch is the first over TWO KEY PROCESSES YOU MUST KNOW AS A COMBINATION PRODUCT Comments. David Meller October 5, 2016 at 5:32 pm. Thank you for this useful and informative article. I work for a small medical device company managing a project to develop a drug/device combination product in which the PMOA is a well-known disinfecting API and there are well-known and comparable competitor products already on themarket.
HOME | MEDTECH INTELLIGENCEBUSINESS ANALYSISCOMBINATION PRODUCTSMARKET ACCESSOPERATIONSPRODUCT DEVELOPMENT The Transformation to True Digital Healthcare. By Stephen Bernstein, Dana Safran, Jody Tropeano, Lisa Schmitz Mazur, Sara Vaezy, Tom Kiesau. Digital health tools and technology have the potential to dramatically improve the patient care experience, but the shift requires purposeful coordination and planned architecture. April 28,2021.
QUALITY CONSIDERATIONS FOR DRUG-DEVICE COMBINATION Quality Considerations for Drug-Device Combination Products. Understanding the GMP requirements of each component is critical to product and compliance success. The rapid development of combination products—biologics, pharmaceuticals and their devices—provides great opportunity for technological advancement, opening new doors forpatient
FIVE STEPS TO MEDICAL DEVICE COMMERCIALIZATION TRENDS IN MEDICAL DEVICE RECALLS TRENDS IN MEDICAL DEVICE PURCHASING, EVALUATION OF VALUE This website uses cookies so that we can provide you with the best user experience possible. Cookie information is stored in your browser and performs functions such as recognising you when you return to our website and helping our team to understand which DEVELOPING MEDICAL DEVICES FOR DRUGS AND ALCOHOL The Bridge is a small device, around two inches wide. It has electrodes attached inside the ear. The electrodes target cranial nerves that cause withdrawal effects. It is a disposable unit and is only used once. Most patients use it for the first five days of detox, or for the entire detox treatment duration. WHAT INDUSTRY IS SAYING ABOUT PROPOSED ETHYLENE OXIDE More than 20 billion medical devices sold in the United States each year are sterilized using ethylene oxide (EtO), according to an agency statement from then-FDA Acting Commissioner Ned Sharpless, M.D. on October 25. EtO is the most common method of sterilization in the country, and as such, the closures of EtO facilities across the country are raising strong concerns among stakeholders. INTEGRATING DATA SCIENCE INTO MEDICAL DEVICE DEVELOPMENT The FDA’s approval of an electrocardiogram (EKG) that enables atrial fibrillation detection right from a patient’s watch band is just one example of how the digitization of medical devices, a part of the Internet of Things movement, is leading product development and innovation in medicine. However, while medical devices built on a connected services platform include components for data APPLE WATCH 4 GETS FDA CLEARANCE Apple Watch 4 Gets FDA Clearance. The product can conduct electrocardiograms and measure the heart’s rhythms. In addition to all the bells and whistles that come with an Apple Watch, the Series 4 product has received FDA clearance—specifically De Novo classification. Unveiled at an Apple event last week, the Series 4 watch is the first over TWO KEY PROCESSES YOU MUST KNOW AS A COMBINATION PRODUCT Comments. David Meller October 5, 2016 at 5:32 pm. Thank you for this useful and informative article. I work for a small medical device company managing a project to develop a drug/device combination product in which the PMOA is a well-known disinfecting API and there are well-known and comparable competitor products already on themarket.
COLUMN | REMOTE AUDITS AND OTHER NEW WAYS OF WORKING IN A 12 hours ago · Audits for medical device companies and other organizations in the medtech space used to be primarily in person. However, the pandemic magnified flaws in the global regulatory process. A shift to remote audits may be the key tointerorganizational
REPROCESSED MEDICAL DEVICES: EASING THE COST OF HEALTHCARE The utilization of reprocessed medical devices can help cut down the increasing cost of healthcare for consumers while also tackling device shortages. Reprocessing of medical devices and equipment has gained a lot of attention, not just across hospitals saving EMERGING TRENDS IN CARDIOVASCULAR DEVICES Cardiac ablation devices accounted for almost 72% of the cardiovascular devices market share in 2020 and are expected to gain massive momentum and, as per GMI estimates. The rising prevalence of atrial fibrillation is a major contributor to the popularity of these devices. The products are addressing an ever-increasing demand forminimally
PEDIATRIC MEDICAL DEVICES ADDRESS COVID-19, 16 COMPANIES In addition to its annual “Make Your Medical Device Pitch for Kids!” competition, which focuses on innovation in the pediatric space, this year the National Capital Consortium for Pediatric Device Innovation (NCC-PDI) launched a COVID-19 edition for 2020. Although COVID-19 has not been affecting the pediatric population as much as other age groups, it is remains a serious threat to children. COMPLIANCE MANAGEMENT FOR MEDICAL DEVICES Flexible and easy to use, Teamcenter combines a robust set of compliance management, traceability and reporting capabilities to deliver the industry’s best solution for medical devices manufacturers facing strict regulatory requirements, increasing competition and demanding time-to-market pressures. MEDTECH ECONOMICS ARCHIVES With an established base of medtech companies and a strong workforce, Ireland remains a key player in the global supply chain. Continuereading →
WILL DIGITAL HEALTH TECHNOLOGY PLAY A ROLE IN TRAVELER That being said, digital health monitoring tools will play a role in assisting workers as they assess passenger health. During a discussion with MedTech Intelligence, David Maman, CEO and co-founder of Tel Aviv-based Binah.ai, explains how digital health technologies will be leveraged for prevention and monitoring now and in the future. TRENDS IN MEDICAL DEVICE RECALLS Since the FDA last issued its Medical Device Recall Report covering FY 2003 to FY 2012, changes in the medical device industry have shifted the nature of medical device recalls. 1 Our team’s goal was to analyze U.S. medical device recalls from 2013 to 2018 to provide an update on notable trends and lessons that medical device firms can incorporate into their quality and risk management systems. BUILD AN EFFECTIVE GLOBAL REGULATORY STRATEGY Global Medical Device Markets. According to the U.S. Department of Commerce, the medical device marketplace worldwide is expected to experience a 5% annual growth rate over the next few years. By the year 2020, the total market is expected to reach approximately $440 billion. The U.S. accounts for 45% of this market share. MEDTECH ECONOMICS ARCHIVES Industry consolidation, consumerized medical devices and cybersecurity are the top issues in the year ahead. Continue reading → Skip to main content* Home
* About Us
* Advisory Board
* Advertise
* Editorial Submissions* Contact Us
* Get The Newsletter! * View The Latest Newsletter * ____Follow me on Twitter * ____Friend me on Facebook * ____Connect with me on LinkedIn * ____Subscribe to me on YouTube * ____Subscribe to my RSS FeedMENU
Skip to content
* Digital Health
* MedTech Economics * Manufacturing Execution * Product Design & Development * Quality/Regulatory* Resource Centers
* Medical Device Audits, Certification & Verification * Medical Device Component * Medical Recall Resource Center * Quality & Regulatory Market Access Solutions Resource Center * Events & Webinars* Library
MEDTRONIC HALTS SALE OF HVAD SYSTEM, URGES HEALTHCARE PROVIDERS TOSTOP IMPLANTATION
By MedTech Intelligence Staff FDA also supports the removal of the HeartWare HVAD System from themarket.
FEATURED ARTICLES
June 4, 2021
MEDTRONIC HALTS SALE OF HVAD SYSTEM, URGES HEALTHCARE PROVIDERS TOSTOP IMPLANTATION
By MedTech Intelligence Staff FDA also supports the removal of the HeartWare HVAD System from themarket.
June 4, 2021
REPROCESSED MEDICAL DEVICES: EASING THE COST OF HEALTHCARE By Hrishikesh Kadam The utilization of reprocessed medical devices can help cut down the increasing cost of healthcare for consumers while also tackling deviceshortages.
May 24, 2021
BOSTON SCIENTIFIC RECALLS VENOUS STENT SYSTEM DUE DEVICE MIGRATION By MedTech Intelligence Staff Nearly 32,000 stent systems have been recalled in the United States.May 19, 2021
HOW TO ACHIEVE QUALITY THROUGH PROCESS VALIDATIONBy David Kachoui
The key is to keep quality high and risk low.May 12, 2021
INCIDENCE OF SEVERE CARDIOVASCULAR CONDITIONS FUELS CARDIAC ARRHYTHMIA MONITORING DEVICES INDUSTRYBy Saloni Walimbe
The pandemic has ushered in a novel era of cardiac health management, with remote patient monitoring at its forefront.May 7, 2021
AI IN MEDTECH:
RISKS AND OPPORTUNITIES OF INNOVATIVE TECHNOLOGIES IN MEDICAL APPLICATIONSBy Dr. Abtin Rad
A review of common risks and pitfalls of incorporating artificial intelligence in medical devices and an overview of the regulatoryframework.
May 6, 2021
ASK THE EXPERT:
HOW CAN MEDICAL DEVICE MANUFACTURERS BENEFIT FROM CONFIGURE, PRICE, QUOTE (CPQ) SOFTWAREBy Experlogix, LLC
A discussion with Peter Willemse about how configure, price, quote (CPQ) software can help medical device manufacturers deal with the nuances of everyday challenges and continue to drive revenue and increase profit margins.May 5, 2021
CGM DEVICES REPRESENT THE ERA OF DIGITAL DIABETES MANAGEMENT By Hrishikesh Kadam Robust trends are expected to power the global digital diabetesmanagement market.
May 3, 2021
FDA REVOKES EUA FOR BATTELLE DECONTAMINATION SYSTEM By MedTech Intelligence Staff The CCDS system was used to decontaminate compatible N95 respirators for multiple re-use.April 27, 2021
AI IN HEALTHCARE TO REINFORCE DRUG DISCOVERY AND MEDTECH APPLICATIONSBy Saloni Walimbe
During the COVID-19 crisis, digital forces like artificial intelligence have assisted the healthcare industry to focus on patient care while achieving improved efficiency during treatments.COLUMNS
June 2, 2021
Soapbox
THE MDR AND OPPORTUNITIES FOR MEDTECH By Barry O’Sullivan The medtech regulatory environment in Europe has entered a new era. A Europe-wide medical device regulation has come into effect, presenting both challenges and opportunities for stakeholders in the sector.May 24, 2021
MEDdesign
BALANCING COMPLIANCE AND QUALITY IN MEDICAL DEVICE MANUFACTURINGBy Kim Wakeman
As the pace of innovation further accelerates in 2021, and the need to get new devices to market intensifies, maintaining adherence to regulatory controls is not enough.May 17, 2021
Soapbox
TOP 4 TECHNOLOGY CONSIDERATIONS USING RPM FOR EARLY INTERVENTIONBy Mark Denissen
Using the right strategy, remote patient monitoring turns episodic care into preventative care, potentially improving the patient experience and health outcomes.May 11, 2021
Soapbox
DOES THE LONG-TERM FUTURE OF TELEHEALTH MEAN LOWER HEALTHCARE COSTS?By Mary Jo Condon
To better understand the future of telehealth, it is important to understand what the costs look like over time and whether telehealth offers advantages in cutting healthcare costs overall.May 4, 2021
MEDdesign
THE TRANSFORMATION TO TRUE DIGITAL HEALTHCARE By Stephen Bernstein, Dana Safran, Jody Tropeano, Lisa Schmitz Mazur, Sara Vaezy, Tom Kiesau Digital health tools and technology have the potential to dramatically improve the patient care experience, but the shift requires purposeful coordination and planned architecture.April 28, 2021
MEDdesign
THE GREAT EQUALIZER: HOW DIGITAL HEALTH IS DEMOCRATIZING HEALTHCAREBy Busy Burr
We all need to play a role to drive the innovative changes necessary and not wait for someone else to do it. UPCOMING EVENTS & WEBINARS __ June 23, 2021 – 12:00 pm – 1:00 pm ACHIEVING OPERATIONAL EXCELLENCE THROUGH SMART MANUFACTURING INMEDICAL DEVICES
ON DEMAND EVENTS & WEBINARS __ April 27, 2021 – 12:00 pm – 2:30 pm STRATEGY AND PLANNING (EU IVDR INNOVATION IN PRACTICE VIRTUALEVENT SERIES)
__ April 20, 2021 – 12:00 pm – 2:30 pm SUPPLY CHAIN MANAGEMENT: ECONOMIC OPERATORS, UDI, AND LABELING (EU IVDR INNOVATION IN PRACTICE VIRTUAL EVENT SERIES) __ April 13, 2021 – 12:00 pm – 2:30 pm CLINICAL EVIDENCE & SURVEILLANCE (EU IVDR INNOVATION IN PRACTICE VIRTUAL EVENT SERIES) __ April 6, 2021 – 12:00 pm – 2:30 pm QUALITY PROCESSES AND DOCUMENTATION (EU IVDR INNOVATION IN PRACTICE VIRTUAL EVENT SERIES) __ March 30, 2021 – 12:00 pm – 2:30 pm EU IVDR INTRODUCTION (EU IVDR INNOVATION IN PRACTICE VIRTUALEVENT SERIES)
__ March 12, 2021 – 1:00 pm – 2:00 pm MEDICAL DEVICE REGULATION – MDR TRANSITION UPDATE __ February 10, 2021 – 1:00 pm – 2:00 pm BRINGING DIGITAL TRANSFORMATION TO YOUR SUPPLY CHAIN: CONNECTING OEMS & CONTRACT MANUFACTURERS FOR IMPROVED PERFORMANCE __ February 9, 2021 – 1:00 pm – 2:00 pm ISO 14971:2019 RISK MANAGEMENT FOR MEDICAL DEVICES __ January 28, 2021 – January 28, 2021 COMPUTER MODELING & SIMULATION IN MEDTECH PRODUCT DEVELOPMENT & SUBMISSIONS (DAY TWO) __ January 27, 2021 – January 27, 2021 COMPUTER MODELING & SIMULATION IN MEDTECH PRODUCT DEVELOPMENT & SUBMISSIONS (DAY ONE) JOIN THE MEDTECH INTELLIGENCE COMMUNITY Stay engaged the way you want to! * Subscribe to MTI's free weekly eNewsletter * Join the LinkedIn group * Follow MTI on Twitter * Follow MTI on Facebook * Subscribe to MTI's RSS feedsTOPIC ARCHIVES
* Digital Health
* MedTech Economics * Manufacturing Execution * Product Design & Development * Quality/RegulatoryCOLUMN ARCHIVES
* Ameing for Asia
* Ask the Engineer
* CAPA Corner
* Devine Guidance
* From The Editor’s Desk* MEDdesign
* Reimbursement Report* Soapbox
CONTACT US
* Advertising Opportunities * Editorial Submissions * General Inquiries* Privacy Policy
* Cookie Policy
* Sign Up for Newsletters INNOVATIVE PUBLISHING COMPANY, INC.* FoodSafetyTech
* Food Safety Consortium Conference & Expo * MedTech Intelligence * Cannabis Industry Journal � Copyright 2015 - 2021 Innovative Publishing Co. LLC, All RightsReserved
Other Innovative Publishing Co. LLC Sites: Food Safety Tech | Cannabis Industry Journal MedTech Intelligence is the leading online trade journal. Join the MedTech Intelligence community and stay engaged the way you want to! * Subscribe to MedTech Intelligence’s free weekly eNewsletter * Join the LinkedIn group * Follow MTI on Twitter * Follow MTI on Facebook * Subscribe to MedTech Intelligence’s RSS feedsNo Thanks
------------------------- WAIT! WAIT! DON’T GO YET! Sign up for our FREE newsletters and get the top stories from MTI right in your email inbox.No Thanks
We are using cookies to give you the best experience on our website. You can find out more about which cookies we are using or switch themoff in settings.
Accept
Close GDPR Cookie Settings* Privacy Overview
* Strictly Necessary Cookies* Cookie Policy
Powered by GDPR Cookie CompliancePrivacy Overview
This website uses cookies so that we can provide you with the best user experience possible. Cookie information is stored in your browser and performs functions such as recognising you when you return to our website and helping our team to understand which sections of the website you find most interesting and useful. Strictly Necessary Cookies Strictly Necessary Cookie should be enabled at all times so that we can save your preferences for cookie settings. We use tracking pixels that set your arrival time at our website, this is used as part of our anti-spam and security measures. Disabling this tracking pixel would disable some of our security measures, and is therefore considered necessary for the safe operation of the website. This tracking pixel is cleared from your system when you delete filesin your history.
We also use cookies to store your preferences regarding the setting of3rd Party Cookies.
Enable or Disable Cookies If you disable this cookie, we will not be able to save your preferences. This means that every time you visit this website you will need to enable or disable cookies again.Cookie Policy
A browser cookie is a small piece of data that is stored on your device to help websites and mobile apps remember things about you. Other technologies, including Web storage and identifiers associated with your device, may be used for similar purposes. In this policy, we say “cookies” to discuss all of these technologies. Our Privacy Policy explains how we collect and use information from and about you when you use This website and certain other Innovative Publishing Co LLC services. This policy explains more about how we use cookies and your related choices.HOW WE USE COOKIES
Data generated from cookies and other behavioral tracking technology is not made available to any outside parties, and is only used in the aggregate to make editorial decisions for the websites. Most browsers are initially set up to accept cookies, but you can reset your browser to refuse all cookies or to indicate when a cookie is being sent by visiting this Cookies Policy page. If your cookies are disabled in the browser, neither the tracking cookie nor the preference cookie is set, and you are in effect opted-out. In other cases, our advertisers request to use third-party tracking to verify our ad delivery, or to remarket their products and/or services to you on other websites. You may opt-out of these tracking pixels by adjusting the Do Not Track settings in your browser, or by visiting the Network Advertising Initiative Opt Outpage.
You have control over whether, how, and when cookies and other tracking technologies are installed on your devices. Although each browser is different, most browsers enable their users to access and edit their cookie preferences in their browser settings. The rejection or disabling of some cookies may impact certain features of the site or to cause some of the website’s services not to function properly. Individuals may opt-out of 3rd Party Cookies used on IPC websites by adjusting your cookie preferences through this Cookie Preferences tool, or by setting web browser settings to refuse cookies and similar tracking mechanisms. Please note that web browsers operate using different identifiers. As such, you must adjust your settings in each web browser and for each computer or device on which you would like to opt-out on. Further, if you simply delete your cookies, you will need to remove cookies from your device after every visit to the websites. You may download a browser plugin that will help you maintain your opt-out choices by visiting www.aboutads.info/pmc. You may block cookies entirely by disabling cookie use in your browser or by setting your browser to ask for your permission before setting a cookie. Blocking cookies entirely may cause some websites to work incorrectly or less effectively. The use of online tracking mechanisms by third parties is subject to those third parties’ own privacy policies, and not this Policy. If you prefer to prevent third parties from setting and accessing cookies on your computer, you may set your browser to block all cookies. Additionally, you may remove yourself from the targeted advertising of companies within the Network Advertising Initiative by optingout here , or of
companies participating in the Digital Advertising Alliance program by opting out here . Enable All Save ChangesDetails
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0