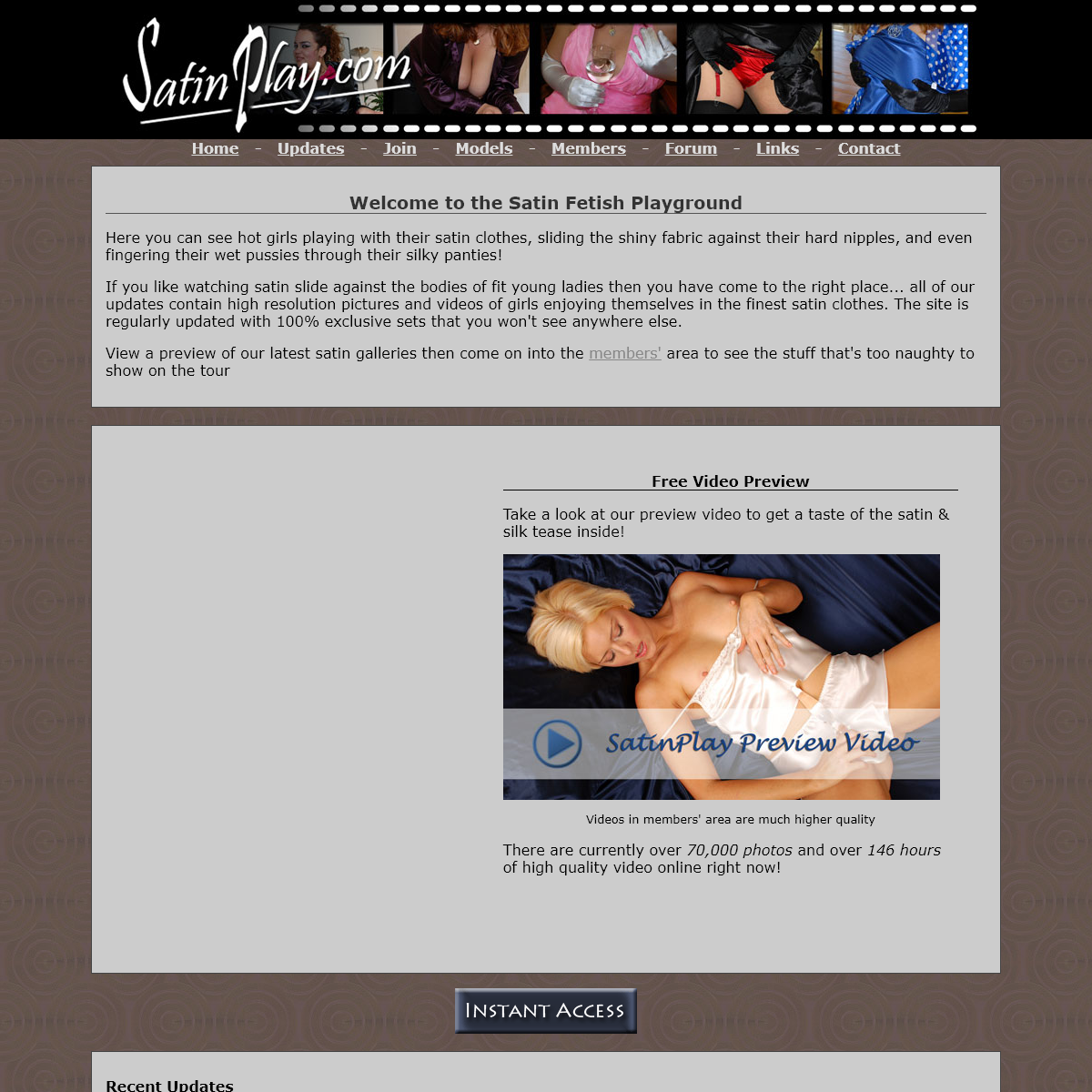
Are you over 18 and want to see adult content?
More Annotations

A complete backup of amateurvoyeurforum.com
Are you over 18 and want to see adult content?

A complete backup of badgirlsblog.com
Are you over 18 and want to see adult content?
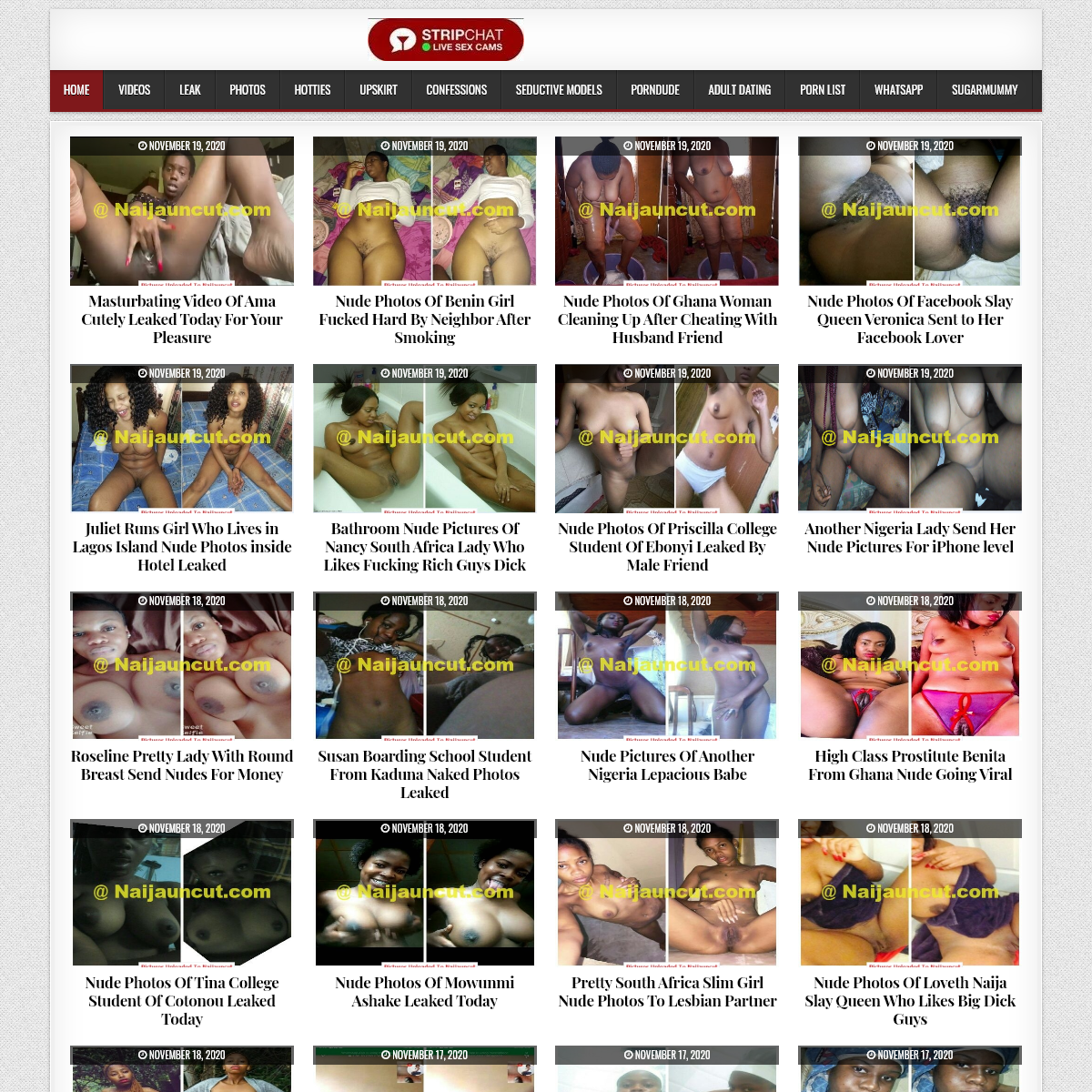
A complete backup of www.naijauncut.com
Are you over 18 and want to see adult content?

A complete backup of grometsplaza.net
Are you over 18 and want to see adult content?

A complete backup of www.asiaerotica.com
Are you over 18 and want to see adult content?

A complete backup of brutal-facesitting.com
Are you over 18 and want to see adult content?
Favourite Annotations
A complete backup of grizzlycustom.com
Are you over 18 and want to see adult content?
A complete backup of easycashin24.com
Are you over 18 and want to see adult content?

A complete backup of sanpellegrino.com
Are you over 18 and want to see adult content?
Text
Fax: +353 1 276
IMMCO REFERENCE LAB SERVICES Autoimmune and Immunogenetics Testing Services. Immco Diagnostics Reference Laboratory, located in Buffalo, NY, USA is licensed throughout the United States for testing serology, biopsy and genetic specimens.Our lab specializes in diagnostics for autoimmune disease such as Sjögren’s syndrome, Thyroiditis, Rheumatoid Arthritis, Type 1 Diabetes, Celiac Disease, Ulcerative Colitis, Crohn’sELISA ARCHIVES
Trinity Biotech offers a range of Captia™ EIAs for autoimmune disorders such as: Antiphospholipid syndrome Coeliac disease Systemic Rheumatoid diseases Vasculitis Thyroid disease Liver disease Please see below for products available in your location.OXALATE ARCHIVES
Trinity Biotech’s Oxalate kit is intended for the quantitative, enzymatic determination of oxalate in urine. Trinity Biotech can provide complete kits and individual components for assaying oxalate concentration. A decreased excretion of oxalate in the urine is associated with hyperglycinemia or hyperglycinuria while an increased excretion of oxalate in the urine can be attributed to an UNI-GOLD™ ARCHIVES The Uni-Gold™ range of rapid point-of-care test devices consists of high-quality, single use immunoassays to aid in the diagnosis of HIV, STDs, respiratory and gastrointestinal diseases. The ease of use and robustness of the products ensures they can be reliably used in diverse settings including clinical laboratories, STD clinics and VCT centres. All products in the range offer: Short timeFINANCIAL REPORTS
Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location.BOARD OF DIRECTORS
James D. Merselis Non-executive director . James joined the board of Trinity Biotech in February 2009. He is currently a Co-Founder and Managing Director of Synchrony Bio LLC, a newly formed healthcare-focused venture investment fund based in St. Louis, MO.G-6-PDH ARCHIVES
Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Trinity Biotech provides a range of tests and components for the quantitative and qualitative determination of Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Deficiency: Quantitative Kinetic tests Qualitative screening test Qualitative Dye Reduction tests Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Deficiency is an X-linked disorder. Males who inherit G-6-PDH DEFICIENCY SCREEN BY SPOT TEST KIT Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location. TRINITY BIOTECH PLC IS A PUBLIC COMPANY, SPECIALISING INOUR COMPANYOUR PRODUCTSIMMCO REFERENCE LAB SERVICESINVESTOR RELATIONSNEWS & EVENTS Company Overview. Trinity Biotech plc is a public company, specialising in the development, manufacture and marketing of clinical diagnostic products. CONTACT US - TRINITY BIOTECH PLC IS A PUBLIC COMPANY Headquarters and ROW enquiries: Trinity Biotech Plc, IDA Business Park, Bray, Co Wicklow, Ireland. A98 H5C8 Telephone: +353 1 276 9800Fax: +353 1 276
IMMCO REFERENCE LAB SERVICES Autoimmune and Immunogenetics Testing Services. Immco Diagnostics Reference Laboratory, located in Buffalo, NY, USA is licensed throughout the United States for testing serology, biopsy and genetic specimens.Our lab specializes in diagnostics for autoimmune disease such as Sjögren’s syndrome, Thyroiditis, Rheumatoid Arthritis, Type 1 Diabetes, Celiac Disease, Ulcerative Colitis, Crohn’sELISA ARCHIVES
Trinity Biotech offers a range of Captia™ EIAs for autoimmune disorders such as: Antiphospholipid syndrome Coeliac disease Systemic Rheumatoid diseases Vasculitis Thyroid disease Liver disease Please see below for products available in your location.OXALATE ARCHIVES
Trinity Biotech’s Oxalate kit is intended for the quantitative, enzymatic determination of oxalate in urine. Trinity Biotech can provide complete kits and individual components for assaying oxalate concentration. A decreased excretion of oxalate in the urine is associated with hyperglycinemia or hyperglycinuria while an increased excretion of oxalate in the urine can be attributed to an UNI-GOLD™ ARCHIVES The Uni-Gold™ range of rapid point-of-care test devices consists of high-quality, single use immunoassays to aid in the diagnosis of HIV, STDs, respiratory and gastrointestinal diseases. The ease of use and robustness of the products ensures they can be reliably used in diverse settings including clinical laboratories, STD clinics and VCT centres. All products in the range offer: Short timeFINANCIAL REPORTS
Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location.BOARD OF DIRECTORS
James D. Merselis Non-executive director . James joined the board of Trinity Biotech in February 2009. He is currently a Co-Founder and Managing Director of Synchrony Bio LLC, a newly formed healthcare-focused venture investment fund based in St. Louis, MO.G-6-PDH ARCHIVES
Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Trinity Biotech provides a range of tests and components for the quantitative and qualitative determination of Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Deficiency: Quantitative Kinetic tests Qualitative screening test Qualitative Dye Reduction tests Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Deficiency is an X-linked disorder. Males who inherit G-6-PDH DEFICIENCY SCREEN BY SPOT TEST KIT Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location. UNI-GOLD™ ARCHIVES The Uni-Gold™ range of rapid point-of-care test devices consists of high-quality, single use immunoassays to aid in the diagnosis of HIV, STDs, respiratory and gastrointestinal diseases. The ease of use and robustness of the products ensures they can be reliably used in diverse settings including clinical laboratories, STD clinics and VCT centres. All products in the range offer: Short time CAREERS - TRINITY BIOTECH PLC IS A PUBLIC COMPANY Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location. BORONATE AFFINITY CHROMATOGRAPHY Boronate Affinity HPLC. Trinity Biotech’s Premier Hb9210 instruments employ the principles of boronate affinity and high-performance liquid chromatography (HPLC). The analytical column contains aminophenylboronic acid bonded to a porous polymer support (gel) and pumps transfer reagents & patient samples through the analyticalcolumn.
TRINITY BIOTECH SUBSIDIARY, PRIMUS CORPORATION RECEIVES Press Release dated August 17, 2020 Contact: Trinity Biotech plc Kevin Tansley (353)-1-2769800 E-mail: kevin.tansley@trinitybiotech.com Trinity Biotech subsidiary, Primus CorporationSYPHILIS ARCHIVES
Trinity Biotech provides one of the most comprehensive ranges of diagnostic tests to aid in the diagnosis of Treponema pallidum (T. pallidum) at all stages of infection and levels of diagnostic testing: Treponemal tests Non–treponemal tests Screening tests Confirmatory tests Blood donor screening Please see below for products available inyour location.
IMMCO REFERENCE LABORATORY Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location. REQUEST SPECIMEN COLLECTION KITS Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location. EARLY SJOGREN'S SYNDROME PROFILE INCLUDES 094, 095, 096 Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location. CLIA WAIVER OF RAPID SYPHILIS TEST TRINITY BIOTECH PLC IS December 16, 2014 | News. Trinity Biotech plc. (Nasdaq: TRIB) today announced that a CLIA waiver has been received for Syphilis Health Check –a point-of-care rapid test for the detection of Syphilis which is distributed by Trinity Biotech in the USA. JOB DESCRIPTION: HEAD OF QUALITY Job Description: Head of QA and RA The Management of non-conformance and corrective and preventative action systems including the control of further processing and delivery of non-conforming TRINITY BIOTECH PLC IS A PUBLIC COMPANY, SPECIALISING INOUR COMPANYOUR PRODUCTSIMMCO REFERENCE LAB SERVICESINVESTOR RELATIONSNEWS & EVENTS Company Overview. Trinity Biotech plc is a public company, specialising in the development, manufacture and marketing of clinical diagnostic products. CONTACT US - TRINITY BIOTECH PLC IS A PUBLIC COMPANY Headquarters and ROW enquiries: Trinity Biotech Plc, IDA Business Park, Bray, Co Wicklow, Ireland. A98 H5C8 Telephone: +353 1 276 9800Fax: +353 1 276
IMMCO REFERENCE LAB SERVICES Autoimmune and Immunogenetics Testing Services. Immco Diagnostics Reference Laboratory, located in Buffalo, NY, USA is licensed throughout the United States for testing serology, biopsy and genetic specimens.Our lab specializes in diagnostics for autoimmune disease such as Sjögren’s syndrome, Thyroiditis, Rheumatoid Arthritis, Type 1 Diabetes, Celiac Disease, Ulcerative Colitis, Crohn’sOXALATE ARCHIVES
Trinity Biotech’s Oxalate kit is intended for the quantitative, enzymatic determination of oxalate in urine. Trinity Biotech can provide complete kits and individual components for assaying oxalate concentration. A decreased excretion of oxalate in the urine is associated with hyperglycinemia or hyperglycinuria while an increased excretion of oxalate in the urine can be attributed to anFINANCIAL REPORTS
Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location.ELISA ARCHIVES
Trinity Biotech offers a range of Captia™ EIAs for autoimmune disorders such as: Antiphospholipid syndrome Coeliac disease Systemic Rheumatoid diseases Vasculitis Thyroid disease Liver disease Please see below for products available in your location.BOARD OF DIRECTORS
James D. Merselis Non-executive director . James joined the board of Trinity Biotech in February 2009. He is currently a Co-Founder and Managing Director of Synchrony Bio LLC, a newly formed healthcare-focused venture investment fund based in St. Louis, MO.G-6-PDH ARCHIVES
Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Trinity Biotech provides a range of tests and components for the quantitative and qualitative determination of Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Deficiency: Quantitative Kinetic tests Qualitative screening test Qualitative Dye Reduction tests Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Deficiency is an X-linked disorder. Males who inherit G-6-PDH DEFICIENCY SCREEN BY SPOT TEST KIT Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location. CLIA WAIVER OF RAPID SYPHILIS TEST TRINITY BIOTECH PLC IS December 16, 2014 | News. Trinity Biotech plc. (Nasdaq: TRIB) today announced that a CLIA waiver has been received for Syphilis Health Check –a point-of-care rapid test for the detection of Syphilis which is distributed by Trinity Biotech in the USA. TRINITY BIOTECH PLC IS A PUBLIC COMPANY, SPECIALISING INOUR COMPANYOUR PRODUCTSIMMCO REFERENCE LAB SERVICESINVESTOR RELATIONSNEWS & EVENTS Company Overview. Trinity Biotech plc is a public company, specialising in the development, manufacture and marketing of clinical diagnostic products. CONTACT US - TRINITY BIOTECH PLC IS A PUBLIC COMPANY Headquarters and ROW enquiries: Trinity Biotech Plc, IDA Business Park, Bray, Co Wicklow, Ireland. A98 H5C8 Telephone: +353 1 276 9800Fax: +353 1 276
IMMCO REFERENCE LAB SERVICES Autoimmune and Immunogenetics Testing Services. Immco Diagnostics Reference Laboratory, located in Buffalo, NY, USA is licensed throughout the United States for testing serology, biopsy and genetic specimens.Our lab specializes in diagnostics for autoimmune disease such as Sjögren’s syndrome, Thyroiditis, Rheumatoid Arthritis, Type 1 Diabetes, Celiac Disease, Ulcerative Colitis, Crohn’sOXALATE ARCHIVES
Trinity Biotech’s Oxalate kit is intended for the quantitative, enzymatic determination of oxalate in urine. Trinity Biotech can provide complete kits and individual components for assaying oxalate concentration. A decreased excretion of oxalate in the urine is associated with hyperglycinemia or hyperglycinuria while an increased excretion of oxalate in the urine can be attributed to anFINANCIAL REPORTS
Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location.ELISA ARCHIVES
Trinity Biotech offers a range of Captia™ EIAs for autoimmune disorders such as: Antiphospholipid syndrome Coeliac disease Systemic Rheumatoid diseases Vasculitis Thyroid disease Liver disease Please see below for products available in your location.BOARD OF DIRECTORS
James D. Merselis Non-executive director . James joined the board of Trinity Biotech in February 2009. He is currently a Co-Founder and Managing Director of Synchrony Bio LLC, a newly formed healthcare-focused venture investment fund based in St. Louis, MO.G-6-PDH ARCHIVES
Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Trinity Biotech provides a range of tests and components for the quantitative and qualitative determination of Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Deficiency: Quantitative Kinetic tests Qualitative screening test Qualitative Dye Reduction tests Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Deficiency is an X-linked disorder. Males who inherit G-6-PDH DEFICIENCY SCREEN BY SPOT TEST KIT Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location. CLIA WAIVER OF RAPID SYPHILIS TEST TRINITY BIOTECH PLC IS December 16, 2014 | News. Trinity Biotech plc. (Nasdaq: TRIB) today announced that a CLIA waiver has been received for Syphilis Health Check –a point-of-care rapid test for the detection of Syphilis which is distributed by Trinity Biotech in the USA. UNI-GOLD™ ARCHIVES The Uni-Gold™ range of rapid point-of-care test devices consists of high-quality, single use immunoassays to aid in the diagnosis of HIV, STDs, respiratory and gastrointestinal diseases. The ease of use and robustness of the products ensures they can be reliably used in diverse settings including clinical laboratories, STD clinics and VCT centres. All products in the range offer: Short time CAREERS - TRINITY BIOTECH PLC IS A PUBLIC COMPANY Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location. BORONATE AFFINITY CHROMATOGRAPHY Boronate Affinity HPLC. Trinity Biotech’s Premier Hb9210 instruments employ the principles of boronate affinity and high-performance liquid chromatography (HPLC). The analytical column contains aminophenylboronic acid bonded to a porous polymer support (gel) and pumps transfer reagents & patient samples through the analyticalcolumn.
TRINITY BIOTECH SUBSIDIARY, PRIMUS CORPORATION RECEIVES Press Release dated August 17, 2020 Contact: Trinity Biotech plc Kevin Tansley (353)-1-2769800 E-mail: kevin.tansley@trinitybiotech.com Trinity Biotech subsidiary, Primus Corporation IMMUGLO™ IFA ARCHIVES Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location.SYPHILIS ARCHIVES
Trinity Biotech provides one of the most comprehensive ranges of diagnostic tests to aid in the diagnosis of Treponema pallidum (T. pallidum) at all stages of infection and levels of diagnostic testing: Treponemal tests Non–treponemal tests Screening tests Confirmatory tests Blood donor screening Please see below for products available inyour location.
REQUEST SPECIMEN COLLECTION KITS Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location. EARLY SJOGREN'S SYNDROME PROFILE INCLUDES 094, 095, 096 Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location. CLIA WAIVER OF RAPID SYPHILIS TEST TRINITY BIOTECH PLC IS December 16, 2014 | News. Trinity Biotech plc. (Nasdaq: TRIB) today announced that a CLIA waiver has been received for Syphilis Health Check –a point-of-care rapid test for the detection of Syphilis which is distributed by Trinity Biotech in the USA. JOB DESCRIPTION: HEAD OF QUALITY Job Description: Head of QA and RA The Management of non-conformance and corrective and preventative action systems including the control of further processing and delivery of non-conforming TRINITY BIOTECH PLC IS A PUBLIC COMPANY, SPECIALISING INOUR COMPANYOUR PRODUCTSIMMCO REFERENCE LAB SERVICESINVESTOR RELATIONSNEWS & EVENTS Company Overview. Trinity Biotech plc is a public company, specialising in the development, manufacture and marketing of clinical diagnostic products. UNI-GOLD™ ARCHIVES The Uni-Gold™ range of rapid point-of-care test devices consists of high-quality, single use immunoassays to aid in the diagnosis of HIV, STDs, respiratory and gastrointestinal diseases. The ease of use and robustness of the products ensures they can be reliably used in diverse settings including clinical laboratories, STD clinics and VCT centres. All products in the range offer: Short timeTECHNICAL SUPPORT
Rest of World Telephone: +353 1 276 9800 Fax: +353 1 276 9888 Clinical Chemistry Clinicalchemistry.techsupport@trinitybiotech.com Infectious Disease: Infectiousdisease.techsupport@trinitybiotech.com Autoimmune: autoimmunetechsupport@trinitybiotech.com Haemoglobins: Haemoglobins.techsupport@trinitybiotech.comFINANCIAL REPORTS
Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location.OXALATE ARCHIVES
Trinity Biotech’s Oxalate kit is intended for the quantitative, enzymatic determination of oxalate in urine. Trinity Biotech can provide complete kits and individual components for assaying oxalate concentration. A decreased excretion of oxalate in the urine is associated with hyperglycinemia or hyperglycinuria while an increased excretion of oxalate in the urine can be attributed to an SYPHILIS HEALTH CHECK™ Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location.G-6-PDH ARCHIVES
Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Trinity Biotech provides a range of tests and components for the quantitative and qualitative determination of Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Deficiency: Quantitative Kinetic tests Qualitative screening test Qualitative Dye Reduction tests Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Deficiency is an X-linked disorder. Males who inherit IMMCO SJÖGREN’S DIAGNOSTIC TEST TOOL Immco Sjögren’s Diagnostic Test Interactive Tool. The Immco Sjögren’s Diagnostic Test Panel analyzes and identifies the most comprehensive set of biomarkers for advanced detection of autoimmune dry eye and early Sjögren’s syndrome. SECURITIES AND EXCHANGE COMMISSION FORM 20-F securities and exchange commission washington d.c. 20549 form 20-f ☐ registration statement pursuant to section 12(b) or (g) of the securities exchange act of 1934 or ☒ annual report pursuant to section 13 or 15(d) of the securities exchange act of 1934 for the fiscal year ended december 31, 2019 or ☐ transition report pursuant to section 13 or 15(d) of the securities exchange act of 1934 CLIA WAIVER OF RAPID SYPHILIS TEST TRINITY BIOTECH PLC IS December 16, 2014 | News. Trinity Biotech plc. (Nasdaq: TRIB) today announced that a CLIA waiver has been received for Syphilis Health Check –a point-of-care rapid test for the detection of Syphilis which is distributed by Trinity Biotech in the USA. TRINITY BIOTECH PLC IS A PUBLIC COMPANY, SPECIALISING INOUR COMPANYOUR PRODUCTSIMMCO REFERENCE LAB SERVICESINVESTOR RELATIONSNEWS & EVENTS Company Overview. Trinity Biotech plc is a public company, specialising in the development, manufacture and marketing of clinical diagnostic products. UNI-GOLD™ ARCHIVES The Uni-Gold™ range of rapid point-of-care test devices consists of high-quality, single use immunoassays to aid in the diagnosis of HIV, STDs, respiratory and gastrointestinal diseases. The ease of use and robustness of the products ensures they can be reliably used in diverse settings including clinical laboratories, STD clinics and VCT centres. All products in the range offer: Short timeTECHNICAL SUPPORT
Rest of World Telephone: +353 1 276 9800 Fax: +353 1 276 9888 Clinical Chemistry Clinicalchemistry.techsupport@trinitybiotech.com Infectious Disease: Infectiousdisease.techsupport@trinitybiotech.com Autoimmune: autoimmunetechsupport@trinitybiotech.com Haemoglobins: Haemoglobins.techsupport@trinitybiotech.comFINANCIAL REPORTS
Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location.OXALATE ARCHIVES
Trinity Biotech’s Oxalate kit is intended for the quantitative, enzymatic determination of oxalate in urine. Trinity Biotech can provide complete kits and individual components for assaying oxalate concentration. A decreased excretion of oxalate in the urine is associated with hyperglycinemia or hyperglycinuria while an increased excretion of oxalate in the urine can be attributed to an SYPHILIS HEALTH CHECK™ Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location.G-6-PDH ARCHIVES
Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Trinity Biotech provides a range of tests and components for the quantitative and qualitative determination of Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Deficiency: Quantitative Kinetic tests Qualitative screening test Qualitative Dye Reduction tests Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Deficiency is an X-linked disorder. Males who inherit IMMCO SJÖGREN’S DIAGNOSTIC TEST TOOL Immco Sjögren’s Diagnostic Test Interactive Tool. The Immco Sjögren’s Diagnostic Test Panel analyzes and identifies the most comprehensive set of biomarkers for advanced detection of autoimmune dry eye and early Sjögren’s syndrome. SECURITIES AND EXCHANGE COMMISSION FORM 20-F securities and exchange commission washington d.c. 20549 form 20-f ☐ registration statement pursuant to section 12(b) or (g) of the securities exchange act of 1934 or ☒ annual report pursuant to section 13 or 15(d) of the securities exchange act of 1934 for the fiscal year ended december 31, 2019 or ☐ transition report pursuant to section 13 or 15(d) of the securities exchange act of 1934 CLIA WAIVER OF RAPID SYPHILIS TEST TRINITY BIOTECH PLC IS December 16, 2014 | News. Trinity Biotech plc. (Nasdaq: TRIB) today announced that a CLIA waiver has been received for Syphilis Health Check –a point-of-care rapid test for the detection of Syphilis which is distributed by Trinity Biotech in the USA.TECHNICAL SUPPORT
Rest of World Telephone: +353 1 276 9800 Fax: +353 1 276 9888 Clinical Chemistry Clinicalchemistry.techsupport@trinitybiotech.com Infectious Disease: Infectiousdisease.techsupport@trinitybiotech.com Autoimmune: autoimmunetechsupport@trinitybiotech.com Haemoglobins: Haemoglobins.techsupport@trinitybiotech.comQUALITY STANDARDS
Trinity Biotech is committed to providing quality products for the diagnostics market. Our US and Irish facilities are each FDA and ISO registered facilities. Trinity BiotechCONTACT INFORMATION
Immco Diagnostics, Inc. 10 Earhart Drive Buffalo, New York 14221 USA Phone: +1-716-691-0091 Phone: +1-800-537-TEST Fax: +1-716-691-6955 Email: laboratoryservices@immco.comBOARD OF DIRECTORS
James D. Merselis Non-executive director . James joined the board of Trinity Biotech in February 2009. He is currently a Co-Founder and Managing Director of Synchrony Bio LLC, a newly formed healthcare-focused venture investment fund based in St. Louis, MO. BORONATE AFFINITY CHROMATOGRAPHY Boronate Affinity HPLC. Trinity Biotech’s Premier Hb9210 instruments employ the principles of boronate affinity and high-performance liquid chromatography (HPLC). The analytical column contains aminophenylboronic acid bonded to a porous polymer support (gel) and pumps transfer reagents & patient samples through the analyticalcolumn.
RESPIRATORY ARCHIVES Trinity Biotech provides a broad range of products to aid in the diagnosis of bacterial and viral respiratory infections across a range of technologies. Our core areas of focus are: Legionella S. pneumoniae Mycoplasma pneumoniae Viral Respiratory Diseases Influenza A & B Parinfluenzae Adenovirus Respiratory Syncytial Virus (RSV) Please see below for products available in your location.EIA ARCHIVES
Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location. TRINITY BIOTECH SUBSIDIARY, PRIMUS CORPORATION RECEIVES Press Release dated August 17, 2020 Contact: Trinity Biotech plc Kevin Tansley (353)-1-2769800 E-mail: kevin.tansley@trinitybiotech.com Trinity Biotech subsidiary, Primus Corporation CLIA WAIVER OF RAPID SYPHILIS TEST TRINITY BIOTECH PLC IS December 16, 2014 | News. Trinity Biotech plc. (Nasdaq: TRIB) today announced that a CLIA waiver has been received for Syphilis Health Check –a point-of-care rapid test for the detection of Syphilis which is distributed by Trinity Biotech in the USA. RAPID SYPHILIS TEST RESULTS IN 10 MINUTES. Rapid Syphilis Test Results in 10 Minutes. Rapid qualitative sc reening for human TP antibodies in whole blood, serum or plasma. • First and only rapid syphilis test, with FDA clearance TRINITY BIOTECH PLC IS A PUBLIC COMPANY, SPECIALISING INOUR COMPANYOUR PRODUCTSIMMCO REFERENCE LAB SERVICESINVESTOR RELATIONSNEWS & EVENTS Company Overview. Trinity Biotech plc is a public company, specialising in the development, manufacture and marketing of clinical diagnostic products. CONTACT US - TRINITY BIOTECH PLC IS A PUBLIC COMPANY Headquarters and ROW enquiries: Trinity Biotech Plc, IDA Business Park, Bray, Co Wicklow, Ireland. A98 H5C8 Telephone: +353 1 276 9800Fax: +353 1 276
UNI-GOLD™ ARCHIVES The Uni-Gold™ range of rapid point-of-care test devices consists of high-quality, single use immunoassays to aid in the diagnosis of HIV, STDs, respiratory and gastrointestinal diseases. The ease of use and robustness of the products ensures they can be reliably used in diverse settings including clinical laboratories, STD clinics and VCT centres. All products in the range offer: Short timeTECHNICAL SUPPORT
Rest of World Telephone: +353 1 276 9800 Fax: +353 1 276 9888 Clinical Chemistry Clinicalchemistry.techsupport@trinitybiotech.com Infectious Disease: Infectiousdisease.techsupport@trinitybiotech.com Autoimmune: autoimmunetechsupport@trinitybiotech.com Haemoglobins: Haemoglobins.techsupport@trinitybiotech.comFINANCIAL REPORTS
Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location.OXALATE ARCHIVES
Trinity Biotech’s Oxalate kit is intended for the quantitative, enzymatic determination of oxalate in urine. Trinity Biotech can provide complete kits and individual components for assaying oxalate concentration. A decreased excretion of oxalate in the urine is associated with hyperglycinemia or hyperglycinuria while an increased excretion of oxalate in the urine can be attributed to an SYPHILIS HEALTH CHECK™ Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location.G-6-PDH ARCHIVES
Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Trinity Biotech provides a range of tests and components for the quantitative and qualitative determination of Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Deficiency: Quantitative Kinetic tests Qualitative screening test Qualitative Dye Reduction tests Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Deficiency is an X-linked disorder. Males who inherit IMMCO SJÖGREN’S DIAGNOSTIC TEST TOOL Immco Sjögren’s Diagnostic Test Interactive Tool. The Immco Sjögren’s Diagnostic Test Panel analyzes and identifies the most comprehensive set of biomarkers for advanced detection of autoimmune dry eye and early Sjögren’s syndrome. How to use this tool. SECURITIES AND EXCHANGE COMMISSION FORM 20-F securities and exchange commission washington d.c. 20549 form 20-f ☐ registration statement pursuant to section 12(b) or (g) of the securities exchange act of 1934 or ☒ annual report pursuant to section 13 or 15(d) of the securities exchange act of 1934 for the fiscal year ended december 31, 2019 or ☐ transition report pursuant to section 13 or 15(d) of the securities exchange act of 1934 TRINITY BIOTECH PLC IS A PUBLIC COMPANY, SPECIALISING INOUR COMPANYOUR PRODUCTSIMMCO REFERENCE LAB SERVICESINVESTOR RELATIONSNEWS & EVENTS Company Overview. Trinity Biotech plc is a public company, specialising in the development, manufacture and marketing of clinical diagnostic products. CONTACT US - TRINITY BIOTECH PLC IS A PUBLIC COMPANY Headquarters and ROW enquiries: Trinity Biotech Plc, IDA Business Park, Bray, Co Wicklow, Ireland. A98 H5C8 Telephone: +353 1 276 9800Fax: +353 1 276
UNI-GOLD™ ARCHIVES The Uni-Gold™ range of rapid point-of-care test devices consists of high-quality, single use immunoassays to aid in the diagnosis of HIV, STDs, respiratory and gastrointestinal diseases. The ease of use and robustness of the products ensures they can be reliably used in diverse settings including clinical laboratories, STD clinics and VCT centres. All products in the range offer: Short timeTECHNICAL SUPPORT
Rest of World Telephone: +353 1 276 9800 Fax: +353 1 276 9888 Clinical Chemistry Clinicalchemistry.techsupport@trinitybiotech.com Infectious Disease: Infectiousdisease.techsupport@trinitybiotech.com Autoimmune: autoimmunetechsupport@trinitybiotech.com Haemoglobins: Haemoglobins.techsupport@trinitybiotech.comFINANCIAL REPORTS
Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location.OXALATE ARCHIVES
Trinity Biotech’s Oxalate kit is intended for the quantitative, enzymatic determination of oxalate in urine. Trinity Biotech can provide complete kits and individual components for assaying oxalate concentration. A decreased excretion of oxalate in the urine is associated with hyperglycinemia or hyperglycinuria while an increased excretion of oxalate in the urine can be attributed to an SYPHILIS HEALTH CHECK™ Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location.G-6-PDH ARCHIVES
Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Trinity Biotech provides a range of tests and components for the quantitative and qualitative determination of Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Deficiency: Quantitative Kinetic tests Qualitative screening test Qualitative Dye Reduction tests Glucose-6-Phosphate Dehydrogenase (G-6-PDH) Deficiency is an X-linked disorder. Males who inherit IMMCO SJÖGREN’S DIAGNOSTIC TEST TOOL Immco Sjögren’s Diagnostic Test Interactive Tool. The Immco Sjögren’s Diagnostic Test Panel analyzes and identifies the most comprehensive set of biomarkers for advanced detection of autoimmune dry eye and early Sjögren’s syndrome. How to use this tool. SECURITIES AND EXCHANGE COMMISSION FORM 20-F securities and exchange commission washington d.c. 20549 form 20-f ☐ registration statement pursuant to section 12(b) or (g) of the securities exchange act of 1934 or ☒ annual report pursuant to section 13 or 15(d) of the securities exchange act of 1934 for the fiscal year ended december 31, 2019 or ☐ transition report pursuant to section 13 or 15(d) of the securities exchange act of 1934 IMMCO REFERENCE LAB SERVICES Autoimmune and Immunogenetics Testing Services. Immco Diagnostics Reference Laboratory, located in Buffalo, NY, USA is licensed throughout the United States for testing serology, biopsy and genetic specimens.Our lab specializes in diagnostics for autoimmune disease such as Sjögren’s syndrome, Thyroiditis, Rheumatoid Arthritis, Type 1 Diabetes, Celiac Disease, Ulcerative Colitis, Crohn’sTECHNICAL SUPPORT
Cookie Type Duration Description; fpestid: 0: 1 year: Fpestid is a ShareThis cookie ID set in the domain of the website operator. locale: session: 1 month: Used by the site to show content to users based on their selected location.QUALITY STANDARDS
Trinity Biotech is committed to providing quality products for the diagnostics market. Our US and Irish facilities are each FDA and ISO registered facilities. Trinity BiotechBOARD OF DIRECTORS
James D. Merselis Non-executive director . James joined the board of Trinity Biotech in February 2009. He is currently a Co-Founder and Managing Director of Synchrony Bio LLC, a newly formed healthcare-focused venture investment fund based in St. Louis, MO.CONTACT INFORMATION
Immco Diagnostics, Inc. 10 Earhart Drive Buffalo, New York 14221 USA Phone: +1-716-691-0091 Phone: +1-800-537-TEST Fax: +1-716-691-6955 Email: laboratoryservices@immco.com RESPIRATORY ARCHIVES Trinity Biotech provides a broad range of products to aid in the diagnosis of bacterial and viral respiratory infections across a range of technologies. Our core areas of focus are: Legionella S. pneumoniae Mycoplasma pneumoniae Viral Respiratory Diseases Influenza A & B Parinfluenzae Adenovirus Respiratory Syncytial Virus (RSV) Please see below for products available in your location. BORONATE AFFINITY CHROMATOGRAPHY Boronate Affinity HPLC. Trinity Biotech’s Premier Hb9210 instruments employ the principles of boronate affinity and high-performance liquid chromatography (HPLC). The analytical column contains aminophenylboronic acid bonded to a porous polymer support (gel) and pumps transfer reagents & patient samples through the analyticalcolumn.
TRINITY BIOTECH SUBSIDIARY, PRIMUS CORPORATION RECEIVES Press Release dated August 17, 2020 Contact: Trinity Biotech plc Kevin Tansley (353)-1-2769800 E-mail: kevin.tansley@trinitybiotech.com Trinity Biotech subsidiary, Primus Corporation CLIA WAIVER OF RAPID SYPHILIS TEST TRINITY BIOTECH PLC IS December 16, 2014 | News. Trinity Biotech plc. (Nasdaq: TRIB) today announced that a CLIA waiver has been received for Syphilis Health Check –a point-of-care rapid test for the detection of Syphilis which is distributed by Trinity Biotech in the USA. RAPID SYPHILIS TEST RESULTS IN 10 MINUTES. Rapid Syphilis Test Results in 10 Minutes. Rapid qualitative sc reening for human TP antibodies in whole blood, serum or plasma. • First and only rapid syphilis test, with FDA clearance* Location: __
* USA
* Canada
* Global
* Careers
* Contact Us
* Technical Support
* News & Events
* Shop
*
* EUR, € CAD, $ USD, $WOOCS v.1.3.4
* Login
* Our Company
* Our Products __
* Point of Care
* Infectious Disease* Haemoglobins
* Autoimmune
* Clinical Chemistry * Antibodies & Proteins* Urinalysis
* Immco Reference Lab Services * Investor Relations* COVID-19 __
* Captia™ SARS-CoV-2 IgG ELISA * Bartels® FlexTrans™ Transport Medium*
Toggle navigation EUR, €CAD, $ USD, $
WOOCS v.1.3.4
OK
* Our Company
* Our Products __
* Point of Care
* Infectious Disease* Haemoglobins
* Autoimmune
* Clinical Chemistry * Antibodies & Proteins* Urinalysis
* Immco Reference Lab Services * Investor Relations* COVID-19 __
* Captia™ SARS-CoV-2 IgG ELISA * Bartels® FlexTrans™ Transport Medium* Location: __
* USA
* Canada
* Global
* Careers
* Contact Us
* Technical Support
* Shop
* Login
UNI-GOLD™ HIV COMPLETELearn more
PREMIER RESOLUTION
Haemoglobinopathies. See Everything.Learn more
FLEXTRANS™ TRANSPORT MEDIA The Trinity Biotech Offering for SARS-CoV-2Learn more
CAPTIA™ SARS-COV-2 IGG The Trinity Biotech Offering for SARS-CoV-2Learn more
TRI-STAT 2
Reliable HbA1c Results with a Small FootprintLearn more
__ __
COMPANY OVERVIEW
Trinity Biotech plc is a public company, specialising in the development, manufacture and marketing of clinical diagnostic products.FEATURED PRODUCTS
PREMIER RESOLUTION
A superior HPLC analyser for thalassemia and haemoglobin variantdetection.
TRI-STAT 2
An ideal solution for HbA1c testing in small laboratories with lowvolume throughput.
CAPTIA™ SARS-COV-2 IGG ELISA Qualitative, 96 well microplate ELISA assay, detecting IgG antibodies to antigens of the SARS-CoV-2 virus in serum & plasma. Provides maximum sensitivity and specificity. BARTELS® FLEXTRANS™ TRANSPORT MEDIUM Transport Medium intended to suppress microbial contamination during transport of specimens. *Now available for order and immediate shipment. Please contact us at Flextrans@trinitybiotech.com. UNI-GOLD™ HIV COMPLETE Single use immunoassay for the rapid detection of antibodies to HIV-1 and HIV-2 virus in serum, plasma or whole blood.__ __
OUR BRANDS
__ __
LATEST NEWS
TRINITY BIOTECH ANNOUNCES RESULTS FOR Q1, 2021 TRINITY BIOTECH PLC TO ANNOUNCE FIRST QUARTER FISCAL YEAR 2021FINANCIAL RESULTS
TRINITY BIOTECH ANNOUNCES THE SUBMISSION OF TRINSCREEN™ HIV TO THE WORLD HEALTH ORGANISATION FOR APPROVAL TRINITY BIOTECH ANNOUNCES QUARTER 4 AND FISCAL YEAR 2020 FINANCIALRESULTS
TRINITY BIOTECH PLC TO ANNOUNCE FOURTH QUARTER AND FISCAL YEAR 2020FINANCIAL RESULTS
March 23, 2021
__
* OUR PRODUCTS
* Point of Care
* Infectious Disease* Haemoglobins
* Autoimmune
* Clinical Chemistry * Antibodies & Proteins* Urinalysis
* Our Company
* Investor Relations * Immco Reference Lab Services* News & Events
* Legal
* Privacy Statement
* Careers
* Contact Us
* Login
Trinity Biotech Plc.IDA Business Park,
Bray,
Co.Wicklow,
Ireland.
A98 H5C8
info@trinitybiotech.com� 2021
Trinity Biotech
Ireland
All rights reserved
CHOOSE LOCATION
Global - Non USA/Canada USA CanadaDetails
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0