Are you over 18 and want to see adult content?
More Annotations
A complete backup of atheistalliance.org
Are you over 18 and want to see adult content?
A complete backup of independentwestand.org
Are you over 18 and want to see adult content?
A complete backup of leninimports.com
Are you over 18 and want to see adult content?

A complete backup of duurzaam-beleggen.nl
Are you over 18 and want to see adult content?
Favourite Annotations
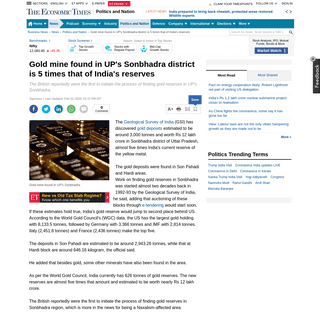
A complete backup of economictimes.indiatimes.com/news/politics-and-nation/gold-deposits-found-in-uttar-pradeshs-sonbhadra/artic
Are you over 18 and want to see adult content?
A complete backup of www.libertatea.ro/stiri/dana-chera-fosta-grecu-nu-se-mai-intoarce-la-antena-3-televiziunea-m-a-stors-de-ene
Are you over 18 and want to see adult content?
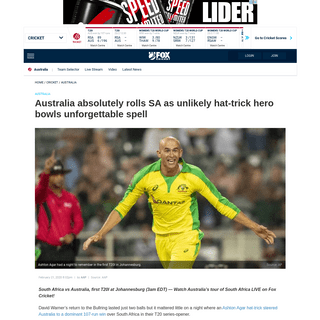
A complete backup of www.foxsports.com.au/cricket/australia/cricket-australia-vs-south-africa-first-t20-live-scores-start-time-h
Are you over 18 and want to see adult content?
Text
BRILIFE VACCINE
WHICH VACCINE FIRST: HPV OR COVID-19 The American Cancer Society (ACS) announced today's efforts highlighting Mission: HPV Cancer Free, a campaign to eliminate human papillomavirus (HPV) cancers. The goal is to reach an HPV vaccination rate of 80% of 13-year-olds in the United States by 2026. COVID-19 VACCINE BREAKTHROUGH FATALITIES AND As of May 22, 2021, the US CDC reported 127,769,851 people over 12 years of age had been fully vaccinated against COVID-19. This data represents about 45.6% of the US population. The CDC also reported today the current 7-day moving average of daily new cases (27,789) decreased 19.5% compared with the previous 7-day moving average. CERTAIN ADULTS SHOULD AVOID ASTRAZENECA'S COVID-19 VACCINE (Precision Vaccinations) The UK's Joint Committee on Vaccination and Immunisation issued advice to the UK government on using the COVID-19 Oxford - AstraZeneca (Vaxzevria) vaccine for people aged under 40 on May 7, 2021.The JCVI issued advice for adults aged 30 to 39 without underlying health conditions to receive an alternative to the Oxford - AstraZeneca COVID-19 vaccine. UB-612 COVID-19 VACCINE UB-612 COVID-19 Vaccine Description. The UB-612 vaccine candidate is specifically designed to allow for the inclusion and presentation of multiple epitopes to elicit both B-cell (antibody) and T-cell (cellular) responses without enhancing COVID-19 disease.. The UB-612 COVAXX vaccine is designed to target a critical antigen from the S protein (Receptor Binding Domain, RBD) thought to be CORAVAX COVID-19 VACCINE CoraVax COVID-19 Description. CoraVax vaccine candidate is made from the spike protein component of the SARS-CoV-2 virus. CoraVax, similar to other vaccines, uses a 'carrier' vaccine. VAXART COVID-19 ORAL VACCINE Vaxart COVID-19 Oral Vaccine (VXA-CoV2-1) Description. Vaxart announced the initiation of a program to develop an oral vaccine VXA-CoV2-1 targeting the novel coronavirus SARS-CoV-2 that causes COVID-19.. VXA-CoV2-1 triggers mucosal immune responses in humans. Mucosal immunity is believed to be the first defense against airborne viruses, such as coronavirus and flu.BRILIFE VACCINE
WHICH VACCINE FIRST: HPV OR COVID-19 The American Cancer Society (ACS) announced today's efforts highlighting Mission: HPV Cancer Free, a campaign to eliminate human papillomavirus (HPV) cancers. The goal is to reach an HPV vaccination rate of 80% of 13-year-olds in the United States by 2026. COVID-19 VACCINE BREAKTHROUGH FATALITIES AND As of May 22, 2021, the US CDC reported 127,769,851 people over 12 years of age had been fully vaccinated against COVID-19. This data represents about 45.6% of the US population. The CDC also reported today the current 7-day moving average of daily new cases (27,789) decreased 19.5% compared with the previous 7-day moving average. CERTAIN ADULTS SHOULD AVOID ASTRAZENECA'S COVID-19 VACCINE (Precision Vaccinations) The UK's Joint Committee on Vaccination and Immunisation issued advice to the UK government on using the COVID-19 Oxford - AstraZeneca (Vaxzevria) vaccine for people aged under 40 on May 7, 2021.The JCVI issued advice for adults aged 30 to 39 without underlying health conditions to receive an alternative to the Oxford - AstraZeneca COVID-19 vaccine. WELCOME TO PRECISION VACCINATIONS Precision Vaccinations publishes vaccine news patients can trust to make informed immunization decisions in partnership with doctors, nurses, and pharmacists. VAXART COVID-19 ORAL VACCINE Vaxart COVID-19 Oral Vaccine (VXA-CoV2-1) Description. Vaxart announced the initiation of a program to develop an oral vaccine VXA-CoV2-1 targeting the novel coronavirus SARS-CoV-2 that causes COVID-19.. VXA-CoV2-1 triggers mucosal immune responses in humans. Mucosal immunity is believed to be the first defense against airborne viruses, such as coronavirus and flu. CORAVAX COVID-19 VACCINE CoraVax COVID-19 Description. CoraVax vaccine candidate is made from the spike protein component of the SARS-CoV-2 virus. CoraVax, similar to other vaccines, uses a 'carrier' vaccine. ZYCOV-D COVID-19 VACCINE ZyCoV-D is a DNA vaccine candidate to fight COVID-19 disease. ZyCoV-D COVID-19 Vaccine Description. ZyCoV-D is India's first indigenously developed DNA vaccine candidate against COVID-19. The ZyCoV-D vaccine candidate is being developed by Zydus Cadila and approved by the Drugs Controller General of India (DCGI) to conduct Phase III clinical trial to the Department of BioteBiotechnology.VACCINES LIST
VLA15 is a multivalent Lyme Disease vaccine candidate targeting the outer surface protein A (OspA) of Borrelia. It is designed for prophylactic, active immunization against Lyme disease aiming for protection against the majority of human pathogenic Borrelia species.VACCINES LIST
Vyrologix (Leronlimab, PRO 140) is a drug candidate that is a CCR5 antagonist with the potential for multiple therapeutic indications including COVID-19Read moreVACCINES LIST
Menveo consists of two portions: 10 µg of lyophilized meningococcal serogroup A capsular polysaccharide conjugated to CRM197 and 5 μg each of capsular polysaccharide of serogroup C, W, and Y conjugated to CRM197 in 0.5 mL of phosphate-buffered saline, which is used to reconstitute the lyophilized MenA component.BRILIFE VACCINE
BriLife (IIBR-100) Vaccine Description. The Brilife (IIBR-100) vaccine candidate contains a recombinant vesicular stomatitis virus (VSV), an animal virus that does not cause disease in humans. The spike protein was replaced with that of SARS-CoV-2 that “results in rapid and potent induction of neutralizing antibodies against the SARS-CoV-2coronavirus.”
VACCINES LIST
RH5.1/AS01 is a vaccine candidate that make many different types of antibodies against the same RH5 protein target. The malaria parasite's protein, RH5, must bind to a human protein onVACCINES LIST
Actemra (Tocilizumab) is an Interleukin-6 (IL-6) rheumatoid arthritis that could help to save the lives of one in 12 intensive care patientswith severe Covid.
WELCOME TO PRECISION VACCINATIONSVACCINESFLUHEPATITISHERPESMEASLESSHINGLES Precision Vaccinations publishes vaccine news patients can trust to make informed immunization decisions in partnership with doctors, nurses, and pharmacists. UB-612 COVID-19 VACCINE UB-612 COVID-19 Vaccine Description. The UB-612 vaccine candidate is specifically designed to allow for the inclusion and presentation of multiple epitopes to elicit both B-cell (antibody) and T-cell (cellular) responses without enhancing COVID-19 disease.. The UB-612 COVAXX vaccine is designed to target a critical antigen from the S protein (Receptor Binding Domain, RBD) thought to be CORAVAX COVID-19 VACCINE CoraVax COVID-19 News. October 16, 2020 - Rabies virus-based COVID-19 vaccine CORAVAX™ induces high levels of neutralizing antibodies against SARS-CoV-2. The results presented in this preclinical study indicate that CORAVAX is an excellent candidate vaccine against COVID-19. August 28, 2020 - An award of $508,000 was granted to Thomas VAXART COVID-19 ORAL VACCINE Vaxart COVID-19 Oral Vaccine (VXA-CoV2-1) Description. Vaxart announced the initiation of a program to develop an oral vaccine VXA-CoV2-1 targeting the novel coronavirus SARS-CoV-2 that causes COVID-19.. VXA-CoV2-1 triggers mucosal immune responses in humans. Mucosal immunity is believed to be the first defense against airborne viruses, such as coronavirus and flu. WHICH VACCINE FIRST: HPV OR COVID-19 Recently, the US Centers for Disease Control and Prevention (CDC) estimated that HPV vaccinations are down by more than 20% compared to 2019. Adding to the challenge is the expected availability of the COVID-19 vaccine for adolescents. It is predicted that COVID-19 vaccines will be available for children as young as 12 years old earlythis summer.
CUBA SOBERANA 02 COVID-19 VACCINE Cuba Soberana 02 COVID-19 Vaccine Description. Soberana 02 (FINLAY-FR-2) COVID-19 vaccine candidate is a conjugate vaccine in which the virus antigen, the receptor-binding domain (RBD), is chemically bound to the tetanus toxoid.On August 13, 2020, Cuba's national regulatory agency, the Center for Quality Control of Medicines, Equipment, and Medical Devices authorized clinical trialsfor
CERTAIN ADULTS SHOULD AVOID ASTRAZENECA'S COVID-19 VACCINE (Precision Vaccinations) The UK's Joint Committee on Vaccination and Immunisation issued advice to the UK government on using the COVID-19 Oxford - AstraZeneca (Vaxzevria) vaccine for people aged under 40 on May 7, 2021.The JCVI issued advice for adults aged 30 to 39 without underlying health conditions to receive an alternative to the Oxford - AstraZeneca COVID-19 vaccine. CAN MRNA VACCINES DEFEAT HERPES? UPenn HSV2 vaccine candidate embraces mRNA technology. A research team at the University of Pennsylvania (UPenn) has been working on preventing and treating genital herpes (HSV-2) for years and recently found success with an mRNA vaccine approach, even before the COVID-19 pandemic. The goals of a HSV-2 vaccine are to keep the herpes virusdormant.
R21 / MATRIX-M MALARIA VACCINE R21/Matrix-M Malaria Vaccine is indicated to prevent Plasmodium falciparum malaria, which is a mosquito-borne disease. There were an estimated 229 million cases of malaria worldwide in 2019, with an estimated 409,000 deaths. According to the WHO, children under the age of five are the most vulnerable, accounting for 67 percent of deaths HSV529 HERPES VACCINE HSV529 Herpes Vaccine Description. Sanofi Pasteur's HSV529 is a herpes vaccine candidate classified as a replication-defective virus. The virus possesses all the wild-type HSV virus components except two proteins UL5 and UL29, involved in viral DNA replication. WELCOME TO PRECISION VACCINATIONSVACCINESFLUHEPATITISHERPESMEASLESSHINGLES Precision Vaccinations publishes vaccine news patients can trust to make informed immunization decisions in partnership with doctors, nurses, and pharmacists. UB-612 COVID-19 VACCINE UB-612 COVID-19 Vaccine Description. The UB-612 vaccine candidate is specifically designed to allow for the inclusion and presentation of multiple epitopes to elicit both B-cell (antibody) and T-cell (cellular) responses without enhancing COVID-19 disease.. The UB-612 COVAXX vaccine is designed to target a critical antigen from the S protein (Receptor Binding Domain, RBD) thought to be CORAVAX COVID-19 VACCINE CoraVax COVID-19 News. October 16, 2020 - Rabies virus-based COVID-19 vaccine CORAVAX™ induces high levels of neutralizing antibodies against SARS-CoV-2. The results presented in this preclinical study indicate that CORAVAX is an excellent candidate vaccine against COVID-19. August 28, 2020 - An award of $508,000 was granted to Thomas VAXART COVID-19 ORAL VACCINE Vaxart COVID-19 Oral Vaccine (VXA-CoV2-1) Description. Vaxart announced the initiation of a program to develop an oral vaccine VXA-CoV2-1 targeting the novel coronavirus SARS-CoV-2 that causes COVID-19.. VXA-CoV2-1 triggers mucosal immune responses in humans. Mucosal immunity is believed to be the first defense against airborne viruses, such as coronavirus and flu. WHICH VACCINE FIRST: HPV OR COVID-19 Recently, the US Centers for Disease Control and Prevention (CDC) estimated that HPV vaccinations are down by more than 20% compared to 2019. Adding to the challenge is the expected availability of the COVID-19 vaccine for adolescents. It is predicted that COVID-19 vaccines will be available for children as young as 12 years old earlythis summer.
CUBA SOBERANA 02 COVID-19 VACCINE Cuba Soberana 02 COVID-19 Vaccine Description. Soberana 02 (FINLAY-FR-2) COVID-19 vaccine candidate is a conjugate vaccine in which the virus antigen, the receptor-binding domain (RBD), is chemically bound to the tetanus toxoid.On August 13, 2020, Cuba's national regulatory agency, the Center for Quality Control of Medicines, Equipment, and Medical Devices authorized clinical trialsfor
CERTAIN ADULTS SHOULD AVOID ASTRAZENECA'S COVID-19 VACCINE (Precision Vaccinations) The UK's Joint Committee on Vaccination and Immunisation issued advice to the UK government on using the COVID-19 Oxford - AstraZeneca (Vaxzevria) vaccine for people aged under 40 on May 7, 2021.The JCVI issued advice for adults aged 30 to 39 without underlying health conditions to receive an alternative to the Oxford - AstraZeneca COVID-19 vaccine. CAN MRNA VACCINES DEFEAT HERPES? UPenn HSV2 vaccine candidate embraces mRNA technology. A research team at the University of Pennsylvania (UPenn) has been working on preventing and treating genital herpes (HSV-2) for years and recently found success with an mRNA vaccine approach, even before the COVID-19 pandemic. The goals of a HSV-2 vaccine are to keep the herpes virusdormant.
R21 / MATRIX-M MALARIA VACCINE R21/Matrix-M Malaria Vaccine is indicated to prevent Plasmodium falciparum malaria, which is a mosquito-borne disease. There were an estimated 229 million cases of malaria worldwide in 2019, with an estimated 409,000 deaths. According to the WHO, children under the age of five are the most vulnerable, accounting for 67 percent of deaths HSV529 HERPES VACCINE HSV529 Herpes Vaccine Description. Sanofi Pasteur's HSV529 is a herpes vaccine candidate classified as a replication-defective virus. The virus possesses all the wild-type HSV virus components except two proteins UL5 and UL29, involved in viral DNA replication. WELCOME TO PRECISION VACCINATIONS Precision Vaccinations publishes vaccine news patients can trust to make informed immunization decisions in partnership with doctors, nurses, and pharmacists. CORAVAX COVID-19 VACCINE CoraVax COVID-19 News. October 16, 2020 - Rabies virus-based COVID-19 vaccine CORAVAX™ induces high levels of neutralizing antibodies against SARS-CoV-2. The results presented in this preclinical study indicate that CORAVAX is an excellent candidate vaccine against COVID-19. August 28, 2020 - An award of $508,000 was granted to ThomasVACCINES LIST
Vyrologix (Leronlimab, PRO 140) is a drug candidate that is a CCR5 antagonist with the potential for multiple therapeutic indications including COVID-19Read moreVACCINES LIST
NanoFlu is an influenza vaccine candidate which is a recombinant hemagglutinin protein nanoparticle influenza vaccine produced in an Sf9 insect cell-baculovirus system.VACCINES LIST
Actemra (Tocilizumab) is an Interleukin-6 (IL-6) rheumatoid arthritis that could help to save the lives of one in 12 intensive care patientswith severe Covid.
VAXART COVID-19 ORAL VACCINE Vaxart COVID-19 Oral Vaccine (VXA-CoV2-1) Description. Vaxart announced the initiation of a program to develop an oral vaccine VXA-CoV2-1 targeting the novel coronavirus SARS-CoV-2 that causes COVID-19.. VXA-CoV2-1 triggers mucosal immune responses in humans. Mucosal immunity is believed to be the first defense against airborne viruses, such as coronavirus and flu.VACCINES LIST
VLA15 is a multivalent Lyme Disease vaccine candidate targeting the outer surface protein A (OspA) of Borrelia. It is designed for prophylactic, active immunization against Lyme disease aiming for protection against the majority of human pathogenic Borrelia species.BRILIFE VACCINE
Brilife is a vector-based vaccine that takes VSV and genetically engineers to express the spike protein of the SARS-CoV-2 beta coronavirus on its envelope. The human body recognizes the spike protein expressed on the envelope and begins to develop an immunological response.VACCINES LIST
Menveo consists of two portions: 10 µg of lyophilized meningococcal serogroup A capsular polysaccharide conjugated to CRM197 and 5 μg each of capsular polysaccharide of serogroup C, W, and Y conjugated to CRM197 in 0.5 mL of phosphate-buffered saline, which is used to reconstitute the lyophilized MenA component.VACCINES LIST
RH5.1/AS01 is a vaccine candidate that make many different types of antibodies against the same RH5 protein target. The malaria parasite's protein, RH5, must bind to a human protein on WELCOME TO PRECISION VACCINATIONSVACCINESFLUHEPATITISHERPESMEASLESSHINGLES Precision Vaccinations publishes vaccine news patients can trust to make informed immunization decisions in partnership with doctors, nurses, and pharmacists. UB-612 COVID-19 VACCINE UB-612 COVID-19 Vaccine Description. The UB-612 vaccine candidate is specifically designed to allow for the inclusion and presentation of multiple epitopes to elicit both B-cell (antibody) and T-cell (cellular) responses without enhancing COVID-19 disease.. The UB-612 COVAXX vaccine is designed to target a critical antigen from the S protein (Receptor Binding Domain, RBD) thought to be CORAVAX COVID-19 VACCINE CoraVax COVID-19 News. October 16, 2020 - Rabies virus-based COVID-19 vaccine CORAVAX™ induces high levels of neutralizing antibodies against SARS-CoV-2. The results presented in this preclinical study indicate that CORAVAX is an excellent candidate vaccine against COVID-19. August 28, 2020 - An award of $508,000 was granted to Thomas VAXART COVID-19 ORAL VACCINE Vaxart COVID-19 Oral Vaccine (VXA-CoV2-1) Description. Vaxart announced the initiation of a program to develop an oral vaccine VXA-CoV2-1 targeting the novel coronavirus SARS-CoV-2 that causes COVID-19.. VXA-CoV2-1 triggers mucosal immune responses in humans. Mucosal immunity is believed to be the first defense against airborne viruses, such as coronavirus and flu. WHICH VACCINE FIRST: HPV OR COVID-19 Recently, the US Centers for Disease Control and Prevention (CDC) estimated that HPV vaccinations are down by more than 20% compared to 2019. Adding to the challenge is the expected availability of the COVID-19 vaccine for adolescents. It is predicted that COVID-19 vaccines will be available for children as young as 12 years old earlythis summer.
CUBA SOBERANA 02 COVID-19 VACCINE Cuba Soberana 02 COVID-19 Vaccine Description. Soberana 02 (FINLAY-FR-2) COVID-19 vaccine candidate is a conjugate vaccine in which the virus antigen, the receptor-binding domain (RBD), is chemically bound to the tetanus toxoid.On August 13, 2020, Cuba's national regulatory agency, the Center for Quality Control of Medicines, Equipment, and Medical Devices authorized clinical trialsfor
CAN MRNA VACCINES DEFEAT HERPES? UPenn HSV2 vaccine candidate embraces mRNA technology. A research team at the University of Pennsylvania (UPenn) has been working on preventing and treating genital herpes (HSV-2) for years and recently found success with an mRNA vaccine approach, even before the COVID-19 pandemic. The goals of a HSV-2 vaccine are to keep the herpes virusdormant.
R21 / MATRIX-M MALARIA VACCINE R21/Matrix-M Malaria Vaccine is indicated to prevent Plasmodium falciparum malaria, which is a mosquito-borne disease. There were an estimated 229 million cases of malaria worldwide in 2019, with an estimated 409,000 deaths. According to the WHO, children under the age of five are the most vulnerable, accounting for 67 percent of deaths HSV529 HERPES VACCINE HSV529 Herpes Vaccine Description. Sanofi Pasteur's HSV529 is a herpes vaccine candidate classified as a replication-defective virus. The virus possesses all the wild-type HSV virus components except two proteins UL5 and UL29, involved in viral DNA replication. CAN MRNA VACCINES PROTECT IMMUNOCOMPROMISED INDIVIDUALS To better understand the immunogenicity of mRNA vaccines in immunocompromised individuals, researchers from Johns Hopkins University School of Medicine quantified the humoral response to the first vaccine dose in solid organ transplant recipients, reported a study published by the JAMA on March 15, 2021. After a median of 20 days post-vaccination, serologic tests showed that WELCOME TO PRECISION VACCINATIONSVACCINESFLUHEPATITISHERPESMEASLESSHINGLES Precision Vaccinations publishes vaccine news patients can trust to make informed immunization decisions in partnership with doctors, nurses, and pharmacists. UB-612 COVID-19 VACCINE UB-612 COVID-19 Vaccine Description. The UB-612 vaccine candidate is specifically designed to allow for the inclusion and presentation of multiple epitopes to elicit both B-cell (antibody) and T-cell (cellular) responses without enhancing COVID-19 disease.. The UB-612 COVAXX vaccine is designed to target a critical antigen from the S protein (Receptor Binding Domain, RBD) thought to be CORAVAX COVID-19 VACCINE CoraVax COVID-19 News. October 16, 2020 - Rabies virus-based COVID-19 vaccine CORAVAX™ induces high levels of neutralizing antibodies against SARS-CoV-2. The results presented in this preclinical study indicate that CORAVAX is an excellent candidate vaccine against COVID-19. August 28, 2020 - An award of $508,000 was granted to Thomas VAXART COVID-19 ORAL VACCINE Vaxart COVID-19 Oral Vaccine (VXA-CoV2-1) Description. Vaxart announced the initiation of a program to develop an oral vaccine VXA-CoV2-1 targeting the novel coronavirus SARS-CoV-2 that causes COVID-19.. VXA-CoV2-1 triggers mucosal immune responses in humans. Mucosal immunity is believed to be the first defense against airborne viruses, such as coronavirus and flu. WHICH VACCINE FIRST: HPV OR COVID-19 Recently, the US Centers for Disease Control and Prevention (CDC) estimated that HPV vaccinations are down by more than 20% compared to 2019. Adding to the challenge is the expected availability of the COVID-19 vaccine for adolescents. It is predicted that COVID-19 vaccines will be available for children as young as 12 years old earlythis summer.
CUBA SOBERANA 02 COVID-19 VACCINE Cuba Soberana 02 COVID-19 Vaccine Description. Soberana 02 (FINLAY-FR-2) COVID-19 vaccine candidate is a conjugate vaccine in which the virus antigen, the receptor-binding domain (RBD), is chemically bound to the tetanus toxoid.On August 13, 2020, Cuba's national regulatory agency, the Center for Quality Control of Medicines, Equipment, and Medical Devices authorized clinical trialsfor
CAN MRNA VACCINES DEFEAT HERPES? UPenn HSV2 vaccine candidate embraces mRNA technology. A research team at the University of Pennsylvania (UPenn) has been working on preventing and treating genital herpes (HSV-2) for years and recently found success with an mRNA vaccine approach, even before the COVID-19 pandemic. The goals of a HSV-2 vaccine are to keep the herpes virusdormant.
R21 / MATRIX-M MALARIA VACCINE R21/Matrix-M Malaria Vaccine is indicated to prevent Plasmodium falciparum malaria, which is a mosquito-borne disease. There were an estimated 229 million cases of malaria worldwide in 2019, with an estimated 409,000 deaths. According to the WHO, children under the age of five are the most vulnerable, accounting for 67 percent of deaths HSV529 HERPES VACCINE HSV529 Herpes Vaccine Description. Sanofi Pasteur's HSV529 is a herpes vaccine candidate classified as a replication-defective virus. The virus possesses all the wild-type HSV virus components except two proteins UL5 and UL29, involved in viral DNA replication. CAN MRNA VACCINES PROTECT IMMUNOCOMPROMISED INDIVIDUALS To better understand the immunogenicity of mRNA vaccines in immunocompromised individuals, researchers from Johns Hopkins University School of Medicine quantified the humoral response to the first vaccine dose in solid organ transplant recipients, reported a study published by the JAMA on March 15, 2021. After a median of 20 days post-vaccination, serologic tests showed that WELCOME TO PRECISION VACCINATIONS Precision Vaccinations publishes vaccine news patients can trust to make informed immunization decisions in partnership with doctors, nurses, and pharmacists. CORAVAX COVID-19 VACCINE CoraVax COVID-19 News. October 16, 2020 - Rabies virus-based COVID-19 vaccine CORAVAX™ induces high levels of neutralizing antibodies against SARS-CoV-2. The results presented in this preclinical study indicate that CORAVAX is an excellent candidate vaccine against COVID-19. August 28, 2020 - An award of $508,000 was granted to Thomas VAXART COVID-19 ORAL VACCINE Vaxart COVID-19 Oral Vaccine (VXA-CoV2-1) Description. Vaxart announced the initiation of a program to develop an oral vaccine VXA-CoV2-1 targeting the novel coronavirus SARS-CoV-2 that causes COVID-19.. VXA-CoV2-1 triggers mucosal immune responses in humans. Mucosal immunity is believed to be the first defense against airborne viruses, such as coronavirus and flu.VACCINES LIST
Vyrologix (Leronlimab, PRO 140) is a drug candidate that is a CCR5 antagonist with the potential for multiple therapeutic indications including COVID-19Read moreVACCINES LIST
Actemra (Tocilizumab) is an Interleukin-6 (IL-6) rheumatoid arthritis that could help to save the lives of one in 12 intensive care patientswith severe Covid.
ZYCOV-D COVID-19 VACCINE ZyCoV-D is a DNA vaccine candidate to fight COVID-19 disease. ZyCoV-D COVID-19 Vaccine Description. ZyCoV-D is India's first indigenously developed DNA vaccine candidate against COVID-19. The ZyCoV-D vaccine candidate is being developed by Zydus Cadila and approved by the Drugs Controller General of India (DCGI) to conduct Phase III clinical trial to the Department of BioteBiotechnology.BRILIFE VACCINE
Brilife is a vector-based vaccine that takes VSV and genetically engineers to express the spike protein of the SARS-CoV-2 beta coronavirus on its envelope. The human body recognizes the spike protein expressed on the envelope and begins to develop an immunological response.VACCINES LIST
VLA15 is a multivalent Lyme Disease vaccine candidate targeting the outer surface protein A (OspA) of Borrelia. It is designed for prophylactic, active immunization against Lyme disease aiming for protection against the majority of human pathogenic Borrelia species.VACCINES LIST
RH5.1/AS01 is a vaccine candidate that make many different types of antibodies against the same RH5 protein target. The malaria parasite's protein, RH5, must bind to a human protein onVACCINES LIST
Ebanga (mAb114) (Ansuvimab) is a human IgG1 MAb targeted to the Zaire ebolavirus glycoprotein (GP). Ebanga is a single monoclonal antibody that binds to the core receptor binding domain of the Zaire ebolavirus surface protein, preventing the virus from infecting human cells. VAXART COVID-19 ORAL VACCINE Vaxart COVID-19 Oral Vaccine (VXA-CoV2-1) Description. Vaxart announced the initiation of a program to develop an oral vaccine VXA-CoV2-1 targeting the novel coronavirus SARS-CoV-2 that causes COVID-19.. VXA-CoV2-1 triggers mucosal immune responses in humans. Mucosal immunity is believed to be the first defense against airborne viruses, such as coronavirus and flu.BRILIFE VACCINE
CAN MRNA VACCINES DEFEAT HERPES? UPenn HSV2 vaccine candidate embraces mRNA technology. A research team at the University of Pennsylvania (UPenn) has been working on preventing and treating genital herpes (HSV-2) for years and recently found success with an mRNA vaccine approach, even before the COVID-19 pandemic. The goals of a HSV-2 vaccine are to keep the herpes virusdormant.
WHICH VACCINE FIRST: HPV OR COVID-19 Recently, the US Centers for Disease Control and Prevention (CDC) estimated that HPV vaccinations are down by more than 20% compared to 2019. Adding to the challenge is the expected availability of the COVID-19 vaccine for adolescents. It is predicted that COVID-19 vaccines will be available for children as young as 12 years old earlythis summer.
COVID-19 VACCINE BREAKTHROUGH FATALITIES AND Furthermore, the CDC stated 'COVID-19 vaccines effectively prevent mild symptoms, severe disease, and hospitalizations due to COVID-19. But it is still possible for a fully vaccinated person to become infected if exposed to the SARS-CoV-2 beta coronavirus.'. As of May 17, 2021, hospitalized or fatal (353) vaccine Breakthrough Casesconfirmed by
COVID-19 ANTIBODIES REMAIN AT LEAST 10 MONTHS AFTER (Precision Vaccinations) Nearly 87% of naturally infected COVID-19 patients maintained antibodies to the SARS-CoV-2 coronavirus proteins for at least 10 months, according to an extensive Labcorp analysis of specimens announced on May 24, 2021.. The study was published in The Lancet - EClinicalMedicine offers real-world evidence of the body’s response to the virus and the possibility of HSV529 HERPES VACCINE HSV529 Herpes Vaccine Description. Sanofi Pasteur's HSV529 is a herpes vaccine candidate classified as a replication-defective virus. The virus possesses all the wild-type HSV virus components except two proteins UL5 and UL29, involved in viral DNA replication. VAXART COVID-19 ORAL VACCINE Vaxart COVID-19 Oral Vaccine (VXA-CoV2-1) Description. Vaxart announced the initiation of a program to develop an oral vaccine VXA-CoV2-1 targeting the novel coronavirus SARS-CoV-2 that causes COVID-19.. VXA-CoV2-1 triggers mucosal immune responses in humans. Mucosal immunity is believed to be the first defense against airborne viruses, such as coronavirus and flu.BRILIFE VACCINE
WHICH VACCINE FIRST: HPV OR COVID-19 Recently, the US Centers for Disease Control and Prevention (CDC) estimated that HPV vaccinations are down by more than 20% compared to 2019. Adding to the challenge is the expected availability of the COVID-19 vaccine for adolescents. It is predicted that COVID-19 vaccines will be available for children as young as 12 years old earlythis summer.
CAN MRNA VACCINES DEFEAT HERPES? UPenn HSV2 vaccine candidate embraces mRNA technology. A research team at the University of Pennsylvania (UPenn) has been working on preventing and treating genital herpes (HSV-2) for years and recently found success with an mRNA vaccine approach, even before the COVID-19 pandemic. The goals of a HSV-2 vaccine are to keep the herpes virusdormant.
COVID-19 VACCINE BREAKTHROUGH FATALITIES AND Furthermore, the CDC stated 'COVID-19 vaccines effectively prevent mild symptoms, severe disease, and hospitalizations due to COVID-19. But it is still possible for a fully vaccinated person to become infected if exposed to the SARS-CoV-2 beta coronavirus.'. As of May 17, 2021, hospitalized or fatal (353) vaccine Breakthrough Casesconfirmed by
COVID-19 ANTIBODIES REMAIN AT LEAST 10 MONTHS AFTER (Precision Vaccinations) Nearly 87% of naturally infected COVID-19 patients maintained antibodies to the SARS-CoV-2 coronavirus proteins for at least 10 months, according to an extensive Labcorp analysis of specimens announced on May 24, 2021.. The study was published in The Lancet - EClinicalMedicine offers real-world evidence of the body’s response to the virus and the possibility of HSV529 HERPES VACCINE HSV529 Herpes Vaccine Description. Sanofi Pasteur's HSV529 is a herpes vaccine candidate classified as a replication-defective virus. The virus possesses all the wild-type HSV virus components except two proteins UL5 and UL29, involved in viral DNA replication. WELCOME TO PRECISION VACCINATIONS Precision Vaccinations publishes vaccine news patients can trust to make informed immunization decisions in partnership with doctors, nurses, and pharmacists.VACCINES LIST
VLA15 is a multivalent Lyme Disease vaccine candidate targeting the outer surface protein A (OspA) of Borrelia. It is designed for prophylactic, active immunization against Lyme disease aiming for protection against the majority of human pathogenic Borrelia species.VACCINES LIST
RH5.1/AS01 is a vaccine candidate that make many different types of antibodies against the same RH5 protein target. The malaria parasite's protein, RH5, must bind to a human protein onBRILIFE VACCINE
Brilife is a vector-based vaccine that takes VSV and genetically engineers to express the spike protein of the SARS-CoV-2 beta coronavirus on its envelope. The human body recognizes the spike protein expressed on the envelope and begins to develop an immunological response. COVID-19 VACCINE UPDATE: BREAKTHROUGH INFECTIONS AND VAERS (Precision Vaccinations) As of May 12, 2021, three experimental COVID-19 vaccines have been Authorized for emergency use in the USA. Since December 11, 2020, the U.S. CDC had reported 116,576,359 people had been fully vaccinated against COVID-19. This data represents about 35.1% of the US population. As of April 26, 2021, the total number of COVID-19 vaccine Breakthrough InfectionsVACCINES LIST
Actemra (Tocilizumab) is an Interleukin-6 (IL-6) rheumatoid arthritis that could help to save the lives of one in 12 intensive care patientswith severe Covid.
VACCINES LIST
Menveo consists of two portions: 10 µg of lyophilized meningococcal serogroup A capsular polysaccharide conjugated to CRM197 and 5 μg each of capsular polysaccharide of serogroup C, W, and Y conjugated to CRM197 in 0.5 mL of phosphate-buffered saline, which is used to reconstitute the lyophilized MenA component. CORAVAX COVID-19 VACCINE CoraVax COVID-19 News. October 16, 2020 - Rabies virus-based COVID-19 vaccine CORAVAX™ induces high levels of neutralizing antibodies against SARS-CoV-2. The results presented in this preclinical study indicate that CORAVAX is an excellent candidate vaccine against COVID-19. August 28, 2020 - An award of $508,000 was granted to Thomas COVI-VAC COVID-19 VACCINE COVI-VAC COVID-19 is an intranasal, live-attenuated vaccine candidate generated using Codagenix’s proprietary deoptimization technology. COV-VAC-COVID -19 is designed to produce immunity against all SARS-CoV-2 proteins, not just the spike surface protein. Live-attenuated vaccines elicit a robust immune response and are associated with long CUBA SOBERANA 02 COVID-19 VACCINE Cuba Soberana 02 COVID-19 Vaccine Description. Soberana 02 (FINLAY-FR-2) COVID-19 vaccine candidate is a conjugate vaccine in which the virus antigen, the receptor-binding domain (RBD), is chemically bound to the tetanus toxoid.On August 13, 2020, Cuba's national regulatory agency, the Center for Quality Control of Medicines, Equipment, and Medical Devices authorized clinical trialsfor
VAXART COVID-19 ORAL VACCINE Vaxart COVID-19 Oral Vaccine (VXA-CoV2-1) Description. Vaxart announced the initiation of a program to develop an oral vaccine VXA-CoV2-1 targeting the novel coronavirus SARS-CoV-2 that causes COVID-19.. VXA-CoV2-1 triggers mucosal immune responses in humans. Mucosal immunity is believed to be the first defense against airborne viruses, such as coronavirus and flu.BRILIFE VACCINE
CAN MRNA VACCINES DEFEAT HERPES? UPenn HSV2 vaccine candidate embraces mRNA technology. A research team at the University of Pennsylvania (UPenn) has been working on preventing and treating genital herpes (HSV-2) for years and recently found success with an mRNA vaccine approach, even before the COVID-19 pandemic. The goals of a HSV-2 vaccine are to keep the herpes virusdormant.
WHICH VACCINE FIRST: HPV OR COVID-19 Recently, the US Centers for Disease Control and Prevention (CDC) estimated that HPV vaccinations are down by more than 20% compared to 2019. Adding to the challenge is the expected availability of the COVID-19 vaccine for adolescents. It is predicted that COVID-19 vaccines will be available for children as young as 12 years old earlythis summer.
COVID-19 VACCINE BREAKTHROUGH FATALITIES AND Furthermore, the CDC stated 'COVID-19 vaccines effectively prevent mild symptoms, severe disease, and hospitalizations due to COVID-19. But it is still possible for a fully vaccinated person to become infected if exposed to the SARS-CoV-2 beta coronavirus.'. As of May 17, 2021, hospitalized or fatal (353) vaccine Breakthrough Casesconfirmed by
COVID-19 ANTIBODIES REMAIN AT LEAST 10 MONTHS AFTER (Precision Vaccinations) Nearly 87% of naturally infected COVID-19 patients maintained antibodies to the SARS-CoV-2 coronavirus proteins for at least 10 months, according to an extensive Labcorp analysis of specimens announced on May 24, 2021.. The study was published in The Lancet - EClinicalMedicine offers real-world evidence of the body’s response to the virus and the possibility of HSV529 HERPES VACCINE HSV529 Herpes Vaccine Description. Sanofi Pasteur's HSV529 is a herpes vaccine candidate classified as a replication-defective virus. The virus possesses all the wild-type HSV virus components except two proteins UL5 and UL29, involved in viral DNA replication. VAXART COVID-19 ORAL VACCINE Vaxart COVID-19 Oral Vaccine (VXA-CoV2-1) Description. Vaxart announced the initiation of a program to develop an oral vaccine VXA-CoV2-1 targeting the novel coronavirus SARS-CoV-2 that causes COVID-19.. VXA-CoV2-1 triggers mucosal immune responses in humans. Mucosal immunity is believed to be the first defense against airborne viruses, such as coronavirus and flu.BRILIFE VACCINE
CAN MRNA VACCINES DEFEAT HERPES? UPenn HSV2 vaccine candidate embraces mRNA technology. A research team at the University of Pennsylvania (UPenn) has been working on preventing and treating genital herpes (HSV-2) for years and recently found success with an mRNA vaccine approach, even before the COVID-19 pandemic. The goals of a HSV-2 vaccine are to keep the herpes virusdormant.
WHICH VACCINE FIRST: HPV OR COVID-19 Recently, the US Centers for Disease Control and Prevention (CDC) estimated that HPV vaccinations are down by more than 20% compared to 2019. Adding to the challenge is the expected availability of the COVID-19 vaccine for adolescents. It is predicted that COVID-19 vaccines will be available for children as young as 12 years old earlythis summer.
COVID-19 VACCINE BREAKTHROUGH FATALITIES AND Furthermore, the CDC stated 'COVID-19 vaccines effectively prevent mild symptoms, severe disease, and hospitalizations due to COVID-19. But it is still possible for a fully vaccinated person to become infected if exposed to the SARS-CoV-2 beta coronavirus.'. As of May 17, 2021, hospitalized or fatal (353) vaccine Breakthrough Casesconfirmed by
COVID-19 ANTIBODIES REMAIN AT LEAST 10 MONTHS AFTER (Precision Vaccinations) Nearly 87% of naturally infected COVID-19 patients maintained antibodies to the SARS-CoV-2 coronavirus proteins for at least 10 months, according to an extensive Labcorp analysis of specimens announced on May 24, 2021.. The study was published in The Lancet - EClinicalMedicine offers real-world evidence of the body’s response to the virus and the possibility of HSV529 HERPES VACCINE HSV529 Herpes Vaccine Description. Sanofi Pasteur's HSV529 is a herpes vaccine candidate classified as a replication-defective virus. The virus possesses all the wild-type HSV virus components except two proteins UL5 and UL29, involved in viral DNA replication. WELCOME TO PRECISION VACCINATIONS Precision Vaccinations publishes vaccine news patients can trust to make informed immunization decisions in partnership with doctors, nurses, and pharmacists.VACCINES LIST
VLA15 is a multivalent Lyme Disease vaccine candidate targeting the outer surface protein A (OspA) of Borrelia. It is designed for prophylactic, active immunization against Lyme disease aiming for protection against the majority of human pathogenic Borrelia species.VACCINES LIST
RH5.1/AS01 is a vaccine candidate that make many different types of antibodies against the same RH5 protein target. The malaria parasite's protein, RH5, must bind to a human protein onBRILIFE VACCINE
Brilife is a vector-based vaccine that takes VSV and genetically engineers to express the spike protein of the SARS-CoV-2 beta coronavirus on its envelope. The human body recognizes the spike protein expressed on the envelope and begins to develop an immunological response. COVID-19 VACCINE UPDATE: BREAKTHROUGH INFECTIONS AND VAERS (Precision Vaccinations) As of May 12, 2021, three experimental COVID-19 vaccines have been Authorized for emergency use in the USA. Since December 11, 2020, the U.S. CDC had reported 116,576,359 people had been fully vaccinated against COVID-19. This data represents about 35.1% of the US population. As of April 26, 2021, the total number of COVID-19 vaccine Breakthrough InfectionsVACCINES LIST
Actemra (Tocilizumab) is an Interleukin-6 (IL-6) rheumatoid arthritis that could help to save the lives of one in 12 intensive care patientswith severe Covid.
VACCINES LIST
Menveo consists of two portions: 10 µg of lyophilized meningococcal serogroup A capsular polysaccharide conjugated to CRM197 and 5 μg each of capsular polysaccharide of serogroup C, W, and Y conjugated to CRM197 in 0.5 mL of phosphate-buffered saline, which is used to reconstitute the lyophilized MenA component. CORAVAX COVID-19 VACCINE CoraVax COVID-19 News. October 16, 2020 - Rabies virus-based COVID-19 vaccine CORAVAX™ induces high levels of neutralizing antibodies against SARS-CoV-2. The results presented in this preclinical study indicate that CORAVAX is an excellent candidate vaccine against COVID-19. August 28, 2020 - An award of $508,000 was granted to Thomas COVI-VAC COVID-19 VACCINE COVI-VAC COVID-19 is an intranasal, live-attenuated vaccine candidate generated using Codagenix’s proprietary deoptimization technology. COV-VAC-COVID -19 is designed to produce immunity against all SARS-CoV-2 proteins, not just the spike surface protein. Live-attenuated vaccines elicit a robust immune response and are associated with long CUBA SOBERANA 02 COVID-19 VACCINE Cuba Soberana 02 COVID-19 Vaccine Description. Soberana 02 (FINLAY-FR-2) COVID-19 vaccine candidate is a conjugate vaccine in which the virus antigen, the receptor-binding domain (RBD), is chemically bound to the tetanus toxoid.On August 13, 2020, Cuba's national regulatory agency, the Center for Quality Control of Medicines, Equipment, and Medical Devices authorized clinical trialsfor
VAXART COVID-19 ORAL VACCINE Vaxart COVID-19 Oral Vaccine (VXA-CoV2-1) Description. Vaxart announced the initiation of a program to develop an oral vaccine VXA-CoV2-1 targeting the novel coronavirus SARS-CoV-2 that causes COVID-19.. VXA-CoV2-1 triggers mucosal immune responses in humans. Mucosal immunity is believed to be the first defense against airborne viruses, such as coronavirus and flu.BRILIFE VACCINE
CAN MRNA VACCINES DEFEAT HERPES? UPenn HSV2 vaccine candidate embraces mRNA technology. A research team at the University of Pennsylvania (UPenn) has been working on preventing and treating genital herpes (HSV-2) for years and recently found success with an mRNA vaccine approach, even before the COVID-19 pandemic. The goals of a HSV-2 vaccine are to keep the herpes virusdormant.
WHICH VACCINE FIRST: HPV OR COVID-19 Recently, the US Centers for Disease Control and Prevention (CDC) estimated that HPV vaccinations are down by more than 20% compared to 2019. Adding to the challenge is the expected availability of the COVID-19 vaccine for adolescents. It is predicted that COVID-19 vaccines will be available for children as young as 12 years old earlythis summer.
COVID-19 VACCINE BREAKTHROUGH FATALITIES AND Furthermore, the CDC stated 'COVID-19 vaccines effectively prevent mild symptoms, severe disease, and hospitalizations due to COVID-19. But it is still possible for a fully vaccinated person to become infected if exposed to the SARS-CoV-2 beta coronavirus.'. As of May 17, 2021, hospitalized or fatal (353) vaccine Breakthrough Casesconfirmed by
COVID-19 ANTIBODIES REMAIN AT LEAST 10 MONTHS AFTER (Precision Vaccinations) Nearly 87% of naturally infected COVID-19 patients maintained antibodies to the SARS-CoV-2 coronavirus proteins for at least 10 months, according to an extensive Labcorp analysis of specimens announced on May 24, 2021.. The study was published in The Lancet - EClinicalMedicine offers real-world evidence of the body’s response to the virus and the possibility of HSV529 HERPES VACCINE HSV529 Herpes Vaccine Description. Sanofi Pasteur's HSV529 is a herpes vaccine candidate classified as a replication-defective virus. The virus possesses all the wild-type HSV virus components except two proteins UL5 and UL29, involved in viral DNA replication. VAXART COVID-19 ORAL VACCINE Vaxart COVID-19 Oral Vaccine (VXA-CoV2-1) Description. Vaxart announced the initiation of a program to develop an oral vaccine VXA-CoV2-1 targeting the novel coronavirus SARS-CoV-2 that causes COVID-19.. VXA-CoV2-1 triggers mucosal immune responses in humans. Mucosal immunity is believed to be the first defense against airborne viruses, such as coronavirus and flu.BRILIFE VACCINE
CAN MRNA VACCINES DEFEAT HERPES? UPenn HSV2 vaccine candidate embraces mRNA technology. A research team at the University of Pennsylvania (UPenn) has been working on preventing and treating genital herpes (HSV-2) for years and recently found success with an mRNA vaccine approach, even before the COVID-19 pandemic. The goals of a HSV-2 vaccine are to keep the herpes virusdormant.
WHICH VACCINE FIRST: HPV OR COVID-19 Recently, the US Centers for Disease Control and Prevention (CDC) estimated that HPV vaccinations are down by more than 20% compared to 2019. Adding to the challenge is the expected availability of the COVID-19 vaccine for adolescents. It is predicted that COVID-19 vaccines will be available for children as young as 12 years old earlythis summer.
COVID-19 VACCINE BREAKTHROUGH FATALITIES AND Furthermore, the CDC stated 'COVID-19 vaccines effectively prevent mild symptoms, severe disease, and hospitalizations due to COVID-19. But it is still possible for a fully vaccinated person to become infected if exposed to the SARS-CoV-2 beta coronavirus.'. As of May 17, 2021, hospitalized or fatal (353) vaccine Breakthrough Casesconfirmed by
COVID-19 ANTIBODIES REMAIN AT LEAST 10 MONTHS AFTER (Precision Vaccinations) Nearly 87% of naturally infected COVID-19 patients maintained antibodies to the SARS-CoV-2 coronavirus proteins for at least 10 months, according to an extensive Labcorp analysis of specimens announced on May 24, 2021.. The study was published in The Lancet - EClinicalMedicine offers real-world evidence of the body’s response to the virus and the possibility of HSV529 HERPES VACCINE HSV529 Herpes Vaccine Description. Sanofi Pasteur's HSV529 is a herpes vaccine candidate classified as a replication-defective virus. The virus possesses all the wild-type HSV virus components except two proteins UL5 and UL29, involved in viral DNA replication. WELCOME TO PRECISION VACCINATIONS Precision Vaccinations publishes vaccine news patients can trust to make informed immunization decisions in partnership with doctors, nurses, and pharmacists.VACCINES LIST
VLA15 is a multivalent Lyme Disease vaccine candidate targeting the outer surface protein A (OspA) of Borrelia. It is designed for prophylactic, active immunization against Lyme disease aiming for protection against the majority of human pathogenic Borrelia species.VACCINES LIST
RH5.1/AS01 is a vaccine candidate that make many different types of antibodies against the same RH5 protein target. The malaria parasite's protein, RH5, must bind to a human protein onBRILIFE VACCINE
Brilife is a vector-based vaccine that takes VSV and genetically engineers to express the spike protein of the SARS-CoV-2 beta coronavirus on its envelope. The human body recognizes the spike protein expressed on the envelope and begins to develop an immunological response. COVID-19 VACCINE UPDATE: BREAKTHROUGH INFECTIONS AND VAERS (Precision Vaccinations) As of May 12, 2021, three experimental COVID-19 vaccines have been Authorized for emergency use in the USA. Since December 11, 2020, the U.S. CDC had reported 116,576,359 people had been fully vaccinated against COVID-19. This data represents about 35.1% of the US population. As of April 26, 2021, the total number of COVID-19 vaccine Breakthrough InfectionsVACCINES LIST
Actemra (Tocilizumab) is an Interleukin-6 (IL-6) rheumatoid arthritis that could help to save the lives of one in 12 intensive care patientswith severe Covid.
VACCINES LIST
Menveo consists of two portions: 10 µg of lyophilized meningococcal serogroup A capsular polysaccharide conjugated to CRM197 and 5 μg each of capsular polysaccharide of serogroup C, W, and Y conjugated to CRM197 in 0.5 mL of phosphate-buffered saline, which is used to reconstitute the lyophilized MenA component. CORAVAX COVID-19 VACCINE CoraVax COVID-19 News. October 16, 2020 - Rabies virus-based COVID-19 vaccine CORAVAX™ induces high levels of neutralizing antibodies against SARS-CoV-2. The results presented in this preclinical study indicate that CORAVAX is an excellent candidate vaccine against COVID-19. August 28, 2020 - An award of $508,000 was granted to Thomas COVI-VAC COVID-19 VACCINE COVI-VAC COVID-19 is an intranasal, live-attenuated vaccine candidate generated using Codagenix’s proprietary deoptimization technology. COV-VAC-COVID -19 is designed to produce immunity against all SARS-CoV-2 proteins, not just the spike surface protein. Live-attenuated vaccines elicit a robust immune response and are associated with long CUBA SOBERANA 02 COVID-19 VACCINE Cuba Soberana 02 COVID-19 Vaccine Description. Soberana 02 (FINLAY-FR-2) COVID-19 vaccine candidate is a conjugate vaccine in which the virus antigen, the receptor-binding domain (RBD), is chemically bound to the tetanus toxoid.On August 13, 2020, Cuba's national regulatory agency, the Center for Quality Control of Medicines, Equipment, and Medical Devices authorized clinical trialsfor
VAXART COVID-19 ORAL VACCINE Vaxart COVID-19 Oral Vaccine (VXA-CoV2-1) Description. Vaxart announced the initiation of a program to develop an oral vaccine VXA-CoV2-1 targeting the novel coronavirus SARS-CoV-2 that causes COVID-19.. VXA-CoV2-1 triggers mucosal immune responses in humans. Mucosal immunity is believed to be the first defense against airborne viruses, such as coronavirus and flu.BRILIFE VACCINE
CAN MRNA VACCINES DEFEAT HERPES? Philadelphia (Precision Vaccinations). A research team at the University of Pennsylvania (UPenn) has been working on preventing and treating genital herpes (HSV-2) for years and recently found success with an mRNA vaccine approach, even before the COVID-19 pandemic. WHICH VACCINE FIRST: HPV OR COVID-19 The American Cancer Society (ACS) announced today's efforts highlighting Mission: HPV Cancer Free, a campaign to eliminate human papillomavirus (HPV) cancers. The goal is to reach an HPV vaccination rate of 80% of 13-year-olds in the United States by 2026. COVID-19 ANTIBODIES REMAIN AT LEAST 10 MONTHS AFTER (Precision Vaccinations) Nearly 87% of naturally infected COVID-19 patients maintained antibodies to the SARS-CoV-2 coronavirus proteins for at least 10 months, according to an extensive Labcorp analysis of specimens announced on May 24, 2021.. The study was published in The Lancet - EClinicalMedicine offers real-world evidence of the body’s response to the virus and the possibility of COVID-19 VACCINE BREAKTHROUGH FATALITIES AND As of May 22, 2021, the US CDC reported 127,769,851 people over 12 years of age had been fully vaccinated against COVID-19. This data represents about 45.6% of the US population. The CDC also reported today the current 7-day moving average of daily new cases (27,789) decreased 19.5% compared with the previous 7-day moving average. HSV529 HERPES VACCINE HSV529 Herpes Vaccine Description. Sanofi Pasteur's HSV529 is a herpes vaccine candidate classified as a replication-defective virus. The virus possesses all the wild-type HSV virus components except two proteins UL5 and UL29, involved in viral DNA replication. VAXART COVID-19 ORAL VACCINE Vaxart COVID-19 Oral Vaccine (VXA-CoV2-1) Description. Vaxart announced the initiation of a program to develop an oral vaccine VXA-CoV2-1 targeting the novel coronavirus SARS-CoV-2 that causes COVID-19.. VXA-CoV2-1 triggers mucosal immune responses in humans. Mucosal immunity is believed to be the first defense against airborne viruses, such as coronavirus and flu.BRILIFE VACCINE
CAN MRNA VACCINES DEFEAT HERPES? Philadelphia (Precision Vaccinations). A research team at the University of Pennsylvania (UPenn) has been working on preventing and treating genital herpes (HSV-2) for years and recently found success with an mRNA vaccine approach, even before the COVID-19 pandemic. WHICH VACCINE FIRST: HPV OR COVID-19 The American Cancer Society (ACS) announced today's efforts highlighting Mission: HPV Cancer Free, a campaign to eliminate human papillomavirus (HPV) cancers. The goal is to reach an HPV vaccination rate of 80% of 13-year-olds in the United States by 2026. COVID-19 ANTIBODIES REMAIN AT LEAST 10 MONTHS AFTER (Precision Vaccinations) Nearly 87% of naturally infected COVID-19 patients maintained antibodies to the SARS-CoV-2 coronavirus proteins for at least 10 months, according to an extensive Labcorp analysis of specimens announced on May 24, 2021.. The study was published in The Lancet - EClinicalMedicine offers real-world evidence of the body’s response to the virus and the possibility of COVID-19 VACCINE BREAKTHROUGH FATALITIES AND As of May 22, 2021, the US CDC reported 127,769,851 people over 12 years of age had been fully vaccinated against COVID-19. This data represents about 45.6% of the US population. The CDC also reported today the current 7-day moving average of daily new cases (27,789) decreased 19.5% compared with the previous 7-day moving average. HSV529 HERPES VACCINE HSV529 Herpes Vaccine Description. Sanofi Pasteur's HSV529 is a herpes vaccine candidate classified as a replication-defective virus. The virus possesses all the wild-type HSV virus components except two proteins UL5 and UL29, involved in viral DNA replication. WELCOME TO PRECISION VACCINATIONS Precision Vaccinations publishes vaccine news patients can trust to make informed immunization decisions in partnership with doctors, nurses, and pharmacists.VACCINES LIST
RH5.1/AS01 is a vaccine candidate that make many different types of antibodies against the same RH5 protein target. The malaria parasite's protein, RH5, must bind to a human protein on COVID-19 VACCINE UPDATE: BREAKTHROUGH INFECTIONS AND VAERS (Precision Vaccinations) As of May 12, 2021, three experimental COVID-19 vaccines have been Authorized for emergency use in the USA. Since December 11, 2020, the U.S. CDC had reported 116,576,359 people had been fully vaccinated against COVID-19. This data represents about 35.1% of the US population. As of April 26, 2021, the total number of COVID-19 vaccine Breakthrough InfectionsVACCINES LIST
Menveo consists of two portions: 10 µg of lyophilized meningococcal serogroup A capsular polysaccharide conjugated to CRM197 and 5 μg each of capsular polysaccharide of serogroup C, W, and Y conjugated to CRM197 in 0.5 mL of phosphate-buffered saline, which is used to reconstitute the lyophilized MenA component.VACCINES LIST
Actemra (Tocilizumab) is an Interleukin-6 (IL-6) rheumatoid arthritis that could help to save the lives of one in 12 intensive care patientswith severe Covid.
VACCINES LIST
VLA15 is a multivalent Lyme Disease vaccine candidate targeting the outer surface protein A (OspA) of Borrelia. It is designed for prophylactic, active immunization against Lyme disease aiming for protection against the majority of human pathogenic Borrelia species.BRILIFE VACCINE
BriLife (IIBR-100) Vaccine Description. The Brilife (IIBR-100) vaccine candidate contains a recombinant vesicular stomatitis virus (VSV), an animal virus that does not cause disease in humans. The spike protein was replaced with that of SARS-CoV-2 that “results in rapid and potent induction of neutralizing antibodies against the SARS-CoV-2coronavirus.”
COVI-VAC COVID-19 VACCINE COVI-VAC COVID-19 is a single-dose intranasal, live-attenuated vaccine candidate that is generated using Codagenix’s proprietary deoptimization technology. CORAVAX COVID-19 VACCINE CoraVax COVID-19 Description. CoraVax vaccine candidate is made from the spike protein component of the SARS-CoV-2 virus. CoraVax, similar to other vaccines, uses a 'carrier' vaccine. CUBA SOBERANA 02 COVID-19 VACCINE Cuba Soberana 02 COVID-19 Vaccine Description. Soberana 02 (FINLAY-FR-2) COVID-19 vaccine candidate is a conjugate vaccine in which the virus antigen, the receptor-binding domain (RBD), is chemically bound to the tetanus toxoid.On August 13, 2020, Cuba's national regulatory agency, the Center for Quality Control of Medicines, Equipment, and Medical Devices authorized clinical trialsfor
Precision Vaccinations* Clinical Trials
* Lab Tests
* Subscribe
Labs Trials
Subscribe
* Home
* Vaccines
* Cancer
* Coronavirus
* Flu
* Herpes
* HPV
* Measles
* Travel
* Zika
PRECISION VACCINATIONS→ May 7th, 2020
Eliminating _fake vaccine news_ with pharmacist, nurse, and physicianreview.
SARS-COV-2 VACCINE MOVES INTO PHASE 2 STUDY _Moderna mRNA1273 vaccine candidate approved for phase 2 clinical trial_* Coronavirus
* COVID-19
* SARS-CoV-2
PFIZER JOINTLY LAUNCHES CORONAVIRUS VACCINE STUDY _BNT162 vaccine candidate aims to preventCOVID-19 disease_
* Coronavirus
* COVID-19
* SARS-CoV-2
FOR CERTAIN CHILDREN, 2 FLU SHOTS CONFER BETTER PROTECTION _Influenza related pediatric fatalities reached 170 during 2020 flu season_* Flu
* Influenza
FDA REVISES COVID-19 TEST POLICY _Coronavirus antibody tests can produce false positive results_ HERPES IMPACTS WOMEN’S REPRODUCTIVE HEALTH _Genital or Oral Herpes cases exceed 1 billion_ JAPAN OFFERING AVIGAN FOR FREE TO 43 COUNTRIES _Avigan Favipiravir shows anti-viral activities against RNA viruses_ CHINA’S HPV VACCINE COMING SOON _Cecolin HPV vaccine was approved by the China National Medical ProductsAdministration_
ANTHRAX TREATMENT COCKTAIL FOUND IMMUNOGENETIC _Raxibacumab combines with Anthrax Vaccine Adsorbed does not negatively affect the vaccine immunogenicity_ LYME DISEASE VACCINE GAINS GLOBAL PARTNER _Valneva Lyme disease vaccine VLA15 targets the outer surface protein A (OspA) of Borrelia_ PHASE 3 STUDY FINDS SIMILAR EFFICACY BETWEEN REMDESIVIR DOSING DURATIONS _Gilead Remdesivir therapy is not yet licensed or approved anywhere_ RUBELLA VACCINE PRODUCTION TO INCREASE _Wistar RA 27/3 rubella vaccine is a live attenuatedvirus_
PHASE 4 STUDY LAUNCHES IN TEXAS TO PREVENT CORONAVIRUS DISEASE _Texas A&M, Baylor, MD Anderson COVID-19 prevention study uses approved tuberculosis vaccine_MEASLES VACCINES
REPORTED SAFE ONCE AGAIN _Evidence on the safety and effectiveness of MMR/MMRV/MMR+V vaccines supports their use for mass immunization_ $120 MILLION FINANCING TO ADVANCE MAPS VACCINES _Affinivax ASP3772 vaccine is currently being studied in pneumococcal infection in seniors_ AI-MODEL ENHANCE CLINICAL TRIAL SEARCHES BY 50% _Coronavirus clinical studies seek participants for vaccines and therapeutics_ INFLUENZA VACCINATION REDUCES UNNECESSARY ANTIBIOTIC PRESCRIPTIONSARTHRITIS DRUG
DISAPPOINTS AS A COVID-19 TREATMENT CATS CAN BECOME INFECTED WITH CORONAVIRUS INVASIVE MENINGOCOCCAL DISEASEVACCINE APPROVED
SEARCH FOR SARS-COV-2 ORIGIN CONTINUES CORONAVIRUS ANTIBODY TEST LAUNCHESMERS VACCINE
CANDIDATE HOLDS PROMISE FOR OTHER CORONAVIRUSES SELF-COLLECTION COVID-19 TESTKIT APPROVED
WHO
CONFIRMS NSAIDS DO NOT INCREASE HEALTH RISKS WEARING CONTACTS DOES NOT INCREASE CORONAVIRUS RISKHYDROXYCHLOROQUINE
PHASE 3 STUDY LAUNCHING IN THE USA LOWER CORONAVIRUS FATALITY RATE REPORTED ORAL RELATED CANCER CASESINCREASE
WHICH PANDEMIC FORECASTING MODEL IS BEST? EBOLA ‘COCKTAIL’ GAIN’S FDAREVIEW
POLYESTER SWABS APPROVED FORCOVID-19 TESTS
CAN REMDESIVIR TREAT BOTHCOVID-19 AND EBOLA?
$483 MILLION
FUNDING FOR CORONAVIRUS VACCINE DEVELOPMENT SANOFI EMBRACES SMARTPHONES FOR SARS-COV-2TESTS
EVEN FLORIDA'S 1% GOT TESTED For more articles, try our search .BIG STORIES
For Certain Children, 2 Flu Shots Confer Better Protection Lyme Disease Vaccine Gains Global Partner Phase 4 Study Launches in Texas To Prevent Coronavirus Disease Influenza Vaccination Reduces Unnecessary Antibiotic Prescriptions Visiting A Pharmacy? Stay CoveredNEW VAX DATA
Leronlimab (PRO 140) Coronavirus Vaccines mRNA-1273 SARS-CoV-2 Vaccine INO-4700 MERS-CoV Vaccine GLS-5300 MERS-CoV Vaccine Herpes Vaccine Candidates EXD-12 Herpes Simplex Vaccine VLA1553 Chikungunya Vaccine VLA15 Lyme Disease Vaccine BNT162 SARS-CoV-2 Vaccine ChAdOx1 nCoV-19 Vaccine INO-4800 DNA Coronavirus Vaccine HSV529 Herpes Vaccine VPM1002 Tuberculosis Vaccine Ad5-nCoV COVID-19 Vaccine Janssen Adenovirus-Based COVID-19 VaccineHydroxychloroquine
Avigan Antiviral Medication Bacille Calmette-Guerin Tuberculosis VaccineRemdesivir
SITE LINKS
* Contact
* Terms
* Privacy
* About
* Advisory
DISCLAIMER
Precision Vax LLC websites do not provide medical advice, diagnosis, treatment, or prescriptions. Read our terms of use for more info. Copyright ©2020 Precision Vax LLC WE USE COOKIES ON THIS SITEPrivacy policy
Accept
SEARCH
TRANSPARENCY NOTICE
You are leaving this site and connecting to , part of the PrecisionVax network.
Continue
TRANSPARENCY NOTICE
You are leaving this site and connecting to , one of our carefully-selected partner sites. Precision Vax may receive compensation for services rendered.Continue
IMPORTANT NOTICE
You are leaving this site and connecting to . Precision Vax is not liable or responsible for the content, information, security, or failure of any products or services promoted or advertised on third party sites.Continue
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0