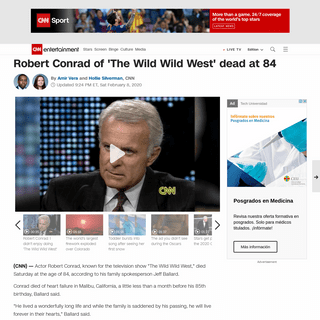
Are you over 18 and want to see adult content?
More Annotations

A complete backup of https://topten.ai
Are you over 18 and want to see adult content?

A complete backup of https://buyonepage.com
Are you over 18 and want to see adult content?

A complete backup of https://flexmastergeneral.com
Are you over 18 and want to see adult content?

A complete backup of https://estevanmercury.ca
Are you over 18 and want to see adult content?

A complete backup of https://chefman.com
Are you over 18 and want to see adult content?

A complete backup of https://graham-windham.org
Are you over 18 and want to see adult content?

A complete backup of https://superaviagra.com
Are you over 18 and want to see adult content?
A complete backup of https://db-palaispopulaire.de
Are you over 18 and want to see adult content?

A complete backup of https://portalkunstgeschichte.de
Are you over 18 and want to see adult content?

A complete backup of https://avval.ir
Are you over 18 and want to see adult content?

A complete backup of https://funnymonkey.com
Are you over 18 and want to see adult content?
A complete backup of https://newvision.fr
Are you over 18 and want to see adult content?
Favourite Annotations
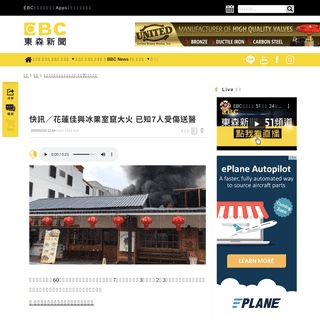
A complete backup of news.ebc.net.tw/news/society/196818
Are you over 18 and want to see adult content?
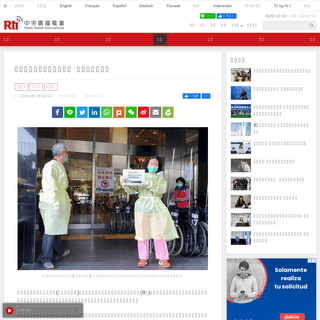
A complete backup of www.rti.org.tw/news/view/id/2050972
Are you over 18 and want to see adult content?

A complete backup of www.behindthesteelcurtain.com/2020/2/8/21128598/complete-xfl-schedule-for-week-1-this-weekend-landry-jones-
Are you over 18 and want to see adult content?
Text
other fields.
USP REFERENCE STANDARDS USP standards for drug reference standards are used to demonstrate identity, strength, purity and quality for medicines, dietary supplements and food ingredients. SEARCH & BUY REFERENCE STANDARDS USP currently offers more than 3,500 Reference Standards—highly characterized specimens of drug substances, excipients, food ingredients, impurities, degradation products, dietary supplements, compendial reagents and performance calibrators. LOGIN - USP ACCESS POINT Welcome to Access Point! Use your USP Access Point login credentials to register for events and courses, access your subscriptions to USP's free resources, applications, and more. COVID-19 VACCINE HANDLING TOOLKIT TRANSPORTING COVID-19 HQS162F_2021-01 The standard of trust Pfizer-BioNTech COVID-19 Vaccine 2 Moderna COVID-19 Vaccine3 Janssen Ad26 COVID-19 Vaccine 4 Janssen is a subsidiary of Johnson & Johnson Temperature* Maximum allowable storage time** Temperature* COVID-19 VACCINE HANDLING TOOLKIT MAXIMIZING DOSES OF V a ci n e o d i u m C h l o r i d S. 5 1. 5 2. 5 5 3 V a c c i n e. 5 0 5 1. 5 2. 5 5 3 V a ci n e Dilution Date/Time: V a c c i n e. 5 1. 5 2. 5 5 3 HQS162F_2021-01 The standard of trust Dilute the vaccine Continued on next page U.S. PHARMACOPEIAABOUT U.S. PHARMACOPEIAREFERENCE STANDARDSSTORECOMPOUNDINGUSP 795OUR IMPACT We are dedicated to helping improve global health through standards setting in compounding, biologics, pharmaceutical manufacturing andother fields.
USP REFERENCE STANDARDS USP standards for drug reference standards are used to demonstrate identity, strength, purity and quality for medicines, dietary supplements and food ingredients. SEARCH & BUY REFERENCE STANDARDS USP currently offers more than 3,500 Reference Standards—highly characterized specimens of drug substances, excipients, food ingredients, impurities, degradation products, dietary supplements, compendial reagents and performance calibrators. LOGIN - USP ACCESS POINT Welcome to Access Point! Use your USP Access Point login credentials to register for events and courses, access your subscriptions to USP's free resources, applications, and more. COVID-19 VACCINE HANDLING TOOLKIT TRANSPORTING COVID-19 HQS162F_2021-01 The standard of trust Pfizer-BioNTech COVID-19 Vaccine 2 Moderna COVID-19 Vaccine3 Janssen Ad26 COVID-19 Vaccine 4 Janssen is a subsidiary of Johnson & Johnson Temperature* Maximum allowable storage time** Temperature* COVID-19 VACCINE HANDLING TOOLKIT MAXIMIZING DOSES OF V a ci n e o d i u m C h l o r i d S. 5 1. 5 2. 5 5 3 V a c c i n e. 5 0 5 1. 5 2. 5 5 3 V a ci n e Dilution Date/Time: V a c c i n e. 5 1. 5 2. 5 5 3 HQS162F_2021-01 The standard of trust Dilute the vaccine Continued on next page USP COMPOUNDING STANDARDS AND BEYOND-USE DATES (BUDS) USP Compounding Standards and Beyond-Use Dates (BUDs) June 3, 2019 Comparing BUDs between the official and the revised The revised chapter addresses Compounded Nonsterile Preparations (CNSPs) requiringshorter BUDs and
DISSOLUTION PERFORMANCE VERIFICATION TESTING (PVT) The USP Performance Verification Test (PVT) is an integral part of the General Chapter Dissolution and assesses proper dissolution apparatusperformance.
561 ARTICLES OF BOTANICAL ORIGIN Crude Fiber Exhaust a weighed quantity of the Test Sample, representing about 2g of the drug, with ether.Add 200 mL of boiling dilute sulfuric acid (1 in 78) to the ether-exhausted marc, in a 500-mL flask, and connect the flask to a reflux condenser. 905 UNIFORMITY OF DOSAGE UNITS USP34 Stage 6 Harmonization 2 〈905〉 Uniformity of Dosage Units Official December 1, 2011 in conditions of normal use, and express the results as de-in which the terms are as defined in Table 2. livered dose. COVID-19 VACCINE HANDLING TOOLKIT The COVID-19 Vaccine Handling Toolkit is an informational resource and is intended to complement the following resources as well as those provided by the CDC, U.S. FDA, vaccine manufacturers, and state and local departments of health:OUR SCIENCE
Pharmaceutical science expertise, of our staff and world-wide volunteer experts, is the foundation of USP’s work to help advance public health and ensure the quality and safety of medicines, dietary supplements and foods. LOGIN - USP ACCESS POINT Welcome to Access Point! Use your USP Access Point login credentials to register for events and courses, access your subscriptions to USP's free resources, applications, and more. SUPPORTING THE GLOBAL HEALTH RESPONSE TO COVID-19 As a standards-setting public health organization that sits at the intersection of the pharmaceutical, regulatory, manufacturing, education and research sectors, USP is marshaling its technical expertise to help the global community respond to COVID-19. CHROMATOGRAPHIC COLUMNS The Chromatographic Database is an online compilation of the chromatographic columns used to validate the chromatographic procedures in USP–NF, Pharmacopeial Forum, Revision Bulletins, and Interim Revision Announcements.It provides a cumulative listing of columns referenced in gas and liquid chromatographic methods related to revisions made to USP–NF since January 1980. THE LAST-MILE MANUFACTURING OF VACCINES 3 Th ast-mil anufacturin accines: Recommendations for scaling-up production of COVID-19 vaccines in Africa I. Background Before the SARS-CoV-2 pandemic, Africa was facing USP GENERAL CHAPTER RADIOPHARMACEUTICALS Developing USP General Chapter USP is a not-for-profit, science-driven organization that has an established process for convening independent experts for the development and maintenance of quality standards. The process is public health focused, leveraging current science and technology, and draws on the expertise of scientists and healthcare practitioners while providing DISSOLUTION PERFORMANCE VERIFICATION TESTING (PVT) The USP Performance Verification Test (PVT) is an integral part of the General Chapter Dissolution and assesses proper dissolution apparatusperformance.
711 DISSOLUTION
Stage 6 Harmonization 2 〈711〉 Dissolution Official December 1, 2011 Figure 1. Basket Stirring Element 2S (USP34) of 25±2 mm between the bottom of the blade and theinside bottom of the vessel is maintained during the test. 2-PROPANOL (1.2 ML/AMPULE; 3 AMPULES) Catalog # 1570428: Current Lot R048P0: Previous Lot I0L393 (Valid Use Date: 31-MAR-2019) SDS View: USP Certificate for Current Lot U.S. PHARMACOPEIAABOUT U.S. PHARMACOPEIAREFERENCE STANDARDSSTORECOMPOUNDINGUSP 795OUR IMPACT Ignore the politics. Trust the science. Recent politicization of the COVID-19 vaccine development and approval process has led to a concerning loss of confidence in vital governmental institutions designed to protect the public from harm. USP REFERENCE STANDARDS To confirm accuracy and reproducibility, USP Reference Standards are rigorously tested and evaluated by multiple independent laboratories including USP, commercial, regulatory, and academic labs. We also provide publicly available, official documentary standards for pharmaceutical ingredients in the USP–NF that link directly with ourprimary
OUR SCIENCE
Our Science. Pharmaceutical science expertise, of our staff and world-wide volunteer experts, is the foundation of USP’s work to help advance public health and ensure the quality and safety of medicines, dietary supplements and foods. The quality standards we develop help manufacturers deliver safe products to billions of peopleworldwide.
SEARCH & BUY REFERENCE STANDARDS Search & Buy Reference Standards. The following list of RSs have been assigned a Valid Use Date of October 31, 2020. Due to revisions to USP General Chapter and General Notices. 5.80, which will become official November 1, 2020, users will no longer be able to assume a value of 100.0% for these RS lots in quantitative USP compendial LOGIN - USP ACCESS POINT Welcome to Access Point! Use your USP Access Point login credentials to register for events and courses, access your subscriptions to USP's free resources, applications, and more. Note: Please do not bookmark this page. Refer to FAQs. Click here for Access Point Tutorial Create a new USP Access Point account, if you do not have one. EDUCATION AND RESOURCES The New USP–NF Online. Quickly find the resources you need: Drug substances, dosage forms, compounded preparations and excipients all in one location. General chapters describe procedures referred to in multiple monographs. Simplified navigation and comprehensive search tool. Annotations and bookmarks. Automatic notifications of upcomingchanges.
COVID-19 VACCINE HANDLING TOOLKIT TRANSPORTING COVID-19 HQS162F_2021-01 The standard of trust Pfizer-BioNTech COVID-19 Vaccine 2 Moderna COVID-19 Vaccine3 Janssen Ad26 COVID-19 Vaccine 4 Janssen is a subsidiary of Johnson & Johnson Temperature* Maximum allowable storage time** Temperature* DISSOLUTION PERFORMANCE VERIFICATION TESTING (PVT) The USP Performance Verification Test (PVT) is an integral part of the General Chapter Dissolution and assesses proper dissolution apparatus performance. PVT is a holistic test and by using the reference standard material and the standard procedure, laboratories can compare results from their instrument with other laboratories worldwide. USP COMPOUNDING STANDARDS AND BEYOND-USE DATES (BUDS) USP Compounding Standards and Beyond-Use Dates (BUDs) June 3, 2019 Comparing BUDs between the official and the revised The revised chapter addresses Compounded Nonsterile Preparations (CNSPs) requiringshorter BUDs and
IBUPROFEN (750 MG)
Catalog # 1335508: Current Lot R13060: Previous Lot R024X0 (Valid Use Date: 31-AUG-2021) SDS View: USP Certificate for Current Lot U.S. PHARMACOPEIAABOUT U.S. PHARMACOPEIAREFERENCE STANDARDSSTORECOMPOUNDINGUSP 795OUR IMPACT Ignore the politics. Trust the science. Recent politicization of the COVID-19 vaccine development and approval process has led to a concerning loss of confidence in vital governmental institutions designed to protect the public from harm. USP REFERENCE STANDARDS To confirm accuracy and reproducibility, USP Reference Standards are rigorously tested and evaluated by multiple independent laboratories including USP, commercial, regulatory, and academic labs. We also provide publicly available, official documentary standards for pharmaceutical ingredients in the USP–NF that link directly with ourprimary
OUR SCIENCE
Our Science. Pharmaceutical science expertise, of our staff and world-wide volunteer experts, is the foundation of USP’s work to help advance public health and ensure the quality and safety of medicines, dietary supplements and foods. The quality standards we develop help manufacturers deliver safe products to billions of peopleworldwide.
SEARCH & BUY REFERENCE STANDARDS Search & Buy Reference Standards. The following list of RSs have been assigned a Valid Use Date of October 31, 2020. Due to revisions to USP General Chapter and General Notices. 5.80, which will become official November 1, 2020, users will no longer be able to assume a value of 100.0% for these RS lots in quantitative USP compendial LOGIN - USP ACCESS POINT Welcome to Access Point! Use your USP Access Point login credentials to register for events and courses, access your subscriptions to USP's free resources, applications, and more. Note: Please do not bookmark this page. Refer to FAQs. Click here for Access Point Tutorial Create a new USP Access Point account, if you do not have one. EDUCATION AND RESOURCES The New USP–NF Online. Quickly find the resources you need: Drug substances, dosage forms, compounded preparations and excipients all in one location. General chapters describe procedures referred to in multiple monographs. Simplified navigation and comprehensive search tool. Annotations and bookmarks. Automatic notifications of upcomingchanges.
COVID-19 VACCINE HANDLING TOOLKIT TRANSPORTING COVID-19 HQS162F_2021-01 The standard of trust Pfizer-BioNTech COVID-19 Vaccine 2 Moderna COVID-19 Vaccine3 Janssen Ad26 COVID-19 Vaccine 4 Janssen is a subsidiary of Johnson & Johnson Temperature* Maximum allowable storage time** Temperature* DISSOLUTION PERFORMANCE VERIFICATION TESTING (PVT) The USP Performance Verification Test (PVT) is an integral part of the General Chapter Dissolution and assesses proper dissolution apparatus performance. PVT is a holistic test and by using the reference standard material and the standard procedure, laboratories can compare results from their instrument with other laboratories worldwide. USP COMPOUNDING STANDARDS AND BEYOND-USE DATES (BUDS) USP Compounding Standards and Beyond-Use Dates (BUDs) June 3, 2019 Comparing BUDs between the official and the revised The revised chapter addresses Compounded Nonsterile Preparations (CNSPs) requiringshorter BUDs and
IBUPROFEN (750 MG)
Catalog # 1335508: Current Lot R13060: Previous Lot R024X0 (Valid Use Date: 31-AUG-2021) SDS View: USP Certificate for Current Lot COVID-19 VACCINE HANDLING TOOLKIT The COVID-19 Vaccine Handling Toolkit is an informational resource and is intended to complement the following resources as well as those provided by the CDC, U.S. FDA, vaccine manufacturers, and state and local departments of health: CDC Vaccine Storage and Handling Official toolkit (February 5, 2021) CHROMATOGRAPHIC COLUMNS The Chromatographic Database is an online compilation of the chromatographic columns used to validate the chromatographic procedures in USP–NF, Pharmacopeial Forum, Revision Bulletins, and Interim Revision Announcements.It provides a cumulative listing of columns referenced in gas and liquid chromatographic methods related to revisions made to USP–NF since January 1980. DISSOLUTION PERFORMANCE VERIFICATION TESTING (PVT) The USP Performance Verification Test (PVT) is an integral part of the General Chapter Dissolution and assesses proper dissolution apparatus performance. PVT is a holistic test and by using the reference standard material and the standard procedure, laboratories can compare results from their instrument with other laboratories worldwide. THE LAST-MILE MANUFACTURING OF VACCINES 3 Th ast-mil anufacturin accines: Recommendations for scaling-up production of COVID-19 vaccines in Africa I. Background Before the SARS-CoV-2 pandemic, Africa was facing REFERENCE STANDARDS: ALPHABETICAL LISTINGS Please note that pre-registration is required to buy Reference Standards. If you're not able to "Select" an item from this list, it means the item is restricted and cannot be ordered online as we first need to ensure that DEA requirements are fulfilled. 1 2 BRIEFING 825 RADIOPHARMACEUTICALS—PREPARATION 1 2 BRIEFING 3 〈825〉 Radiopharmaceuticals—Preparation, Compounding, Dispensing, 4 and Repackaging. Radiopharmaceuticals (radioactive drugs) represent a 5 distinct class of drugs where processing activities include the use of 6 radionuclide generators,preparation and
561 ARTICLES OF BOTANICAL ORIGIN Crude Fiber Exhaust a weighed quantity of the Test Sample, representing about 2g of the drug, with ether.Add 200 mL of boiling dilute sulfuric acid (1 in 78) to the ether-exhausted marc, in a 500-mL flask, and connect the flask to a reflux condenser. 382 ELASTOMERIC CLOSURE FUNCTIONALITY IN INJECTABLE BRIEFING 382 Elastomeric Closure Functionality in Injectable Pharmaceutical Packaging/Delivery Systems. This proposed new general chapter addresses the 905 UNIFORMITY OF DOSAGE UNITS USP34 Stage 6 Harmonization 2 〈905〉 Uniformity of Dosage Units Official December 1, 2011 in conditions of normal use, and express the results as de-in which the terms are as defined in Table 2. livered dose. SALICYLIC ACID (125 MG) Catalog # 1609002: Current Lot K0F112: Previous Lot J3C400 (Valid Use Date: 31-DEC-2007) SDS View: USP Certificate for Current Lot U.S. PHARMACOPEIAABOUT U.S. PHARMACOPEIAREFERENCE STANDARDSSTORECOMPOUNDINGUSP 795OUR IMPACT Ignore the politics. Trust the science. Recent politicization of the COVID-19 vaccine development and approval process has led to a concerning loss of confidence in vital governmental institutions designed to protect the public from harm. USP REFERENCE STANDARDS To confirm accuracy and reproducibility, USP Reference Standards are rigorously tested and evaluated by multiple independent laboratories including USP, commercial, regulatory, and academic labs. We also provide publicly available, official documentary standards for pharmaceutical ingredients in the USP–NF that link directly with ourprimary
OUR SCIENCE
Our Science. Pharmaceutical science expertise, of our staff and world-wide volunteer experts, is the foundation of USP’s work to help advance public health and ensure the quality and safety of medicines, dietary supplements and foods. The quality standards we develop help manufacturers deliver safe products to billions of peopleworldwide.
SEARCH & BUY REFERENCE STANDARDS Search & Buy Reference Standards. The following list of RSs have been assigned a Valid Use Date of October 31, 2020. Due to revisions to USP General Chapter and General Notices. 5.80, which will become official November 1, 2020, users will no longer be able to assume a value of 100.0% for these RS lots in quantitative USP compendial LOGIN - USP ACCESS POINT Welcome to Access Point! Use your USP Access Point login credentials to register for events and courses, access your subscriptions to USP's free resources, applications, and more. Note: Please do not bookmark this page. Refer to FAQs. Click here for Access Point Tutorial Create a new USP Access Point account, if you do not have one. EDUCATION AND RESOURCES The New USP–NF Online. Quickly find the resources you need: Drug substances, dosage forms, compounded preparations and excipients all in one location. General chapters describe procedures referred to in multiple monographs. Simplified navigation and comprehensive search tool. Annotations and bookmarks. Automatic notifications of upcomingchanges.
COVID-19 VACCINE HANDLING TOOLKIT TRANSPORTING COVID-19 HQS162F_2021-01 The standard of trust Pfizer-BioNTech COVID-19 Vaccine 2 Moderna COVID-19 Vaccine3 Janssen Ad26 COVID-19 Vaccine 4 Janssen is a subsidiary of Johnson & Johnson Temperature* Maximum allowable storage time** Temperature* DISSOLUTION PERFORMANCE VERIFICATION TESTING (PVT) The USP Performance Verification Test (PVT) is an integral part of the General Chapter Dissolution and assesses proper dissolution apparatus performance. PVT is a holistic test and by using the reference standard material and the standard procedure, laboratories can compare results from their instrument with other laboratories worldwide. USP COMPOUNDING STANDARDS AND BEYOND-USE DATES (BUDS) USP Compounding Standards and Beyond-Use Dates (BUDs) June 3, 2019 Comparing BUDs between the official and the revised The revised chapter addresses Compounded Nonsterile Preparations (CNSPs) requiringshorter BUDs and
IBUPROFEN (750 MG)
Catalog # 1335508: Current Lot R13060: Previous Lot R024X0 (Valid Use Date: 31-AUG-2021) SDS View: USP Certificate for Current Lot U.S. PHARMACOPEIAABOUT U.S. PHARMACOPEIAREFERENCE STANDARDSSTORECOMPOUNDINGUSP 795OUR IMPACT Ignore the politics. Trust the science. Recent politicization of the COVID-19 vaccine development and approval process has led to a concerning loss of confidence in vital governmental institutions designed to protect the public from harm. USP REFERENCE STANDARDS To confirm accuracy and reproducibility, USP Reference Standards are rigorously tested and evaluated by multiple independent laboratories including USP, commercial, regulatory, and academic labs. We also provide publicly available, official documentary standards for pharmaceutical ingredients in the USP–NF that link directly with ourprimary
OUR SCIENCE
Our Science. Pharmaceutical science expertise, of our staff and world-wide volunteer experts, is the foundation of USP’s work to help advance public health and ensure the quality and safety of medicines, dietary supplements and foods. The quality standards we develop help manufacturers deliver safe products to billions of peopleworldwide.
SEARCH & BUY REFERENCE STANDARDS Search & Buy Reference Standards. The following list of RSs have been assigned a Valid Use Date of October 31, 2020. Due to revisions to USP General Chapter and General Notices. 5.80, which will become official November 1, 2020, users will no longer be able to assume a value of 100.0% for these RS lots in quantitative USP compendial LOGIN - USP ACCESS POINT Welcome to Access Point! Use your USP Access Point login credentials to register for events and courses, access your subscriptions to USP's free resources, applications, and more. Note: Please do not bookmark this page. Refer to FAQs. Click here for Access Point Tutorial Create a new USP Access Point account, if you do not have one. EDUCATION AND RESOURCES The New USP–NF Online. Quickly find the resources you need: Drug substances, dosage forms, compounded preparations and excipients all in one location. General chapters describe procedures referred to in multiple monographs. Simplified navigation and comprehensive search tool. Annotations and bookmarks. Automatic notifications of upcomingchanges.
COVID-19 VACCINE HANDLING TOOLKIT TRANSPORTING COVID-19 HQS162F_2021-01 The standard of trust Pfizer-BioNTech COVID-19 Vaccine 2 Moderna COVID-19 Vaccine3 Janssen Ad26 COVID-19 Vaccine 4 Janssen is a subsidiary of Johnson & Johnson Temperature* Maximum allowable storage time** Temperature* DISSOLUTION PERFORMANCE VERIFICATION TESTING (PVT) The USP Performance Verification Test (PVT) is an integral part of the General Chapter Dissolution and assesses proper dissolution apparatus performance. PVT is a holistic test and by using the reference standard material and the standard procedure, laboratories can compare results from their instrument with other laboratories worldwide. USP COMPOUNDING STANDARDS AND BEYOND-USE DATES (BUDS) USP Compounding Standards and Beyond-Use Dates (BUDs) June 3, 2019 Comparing BUDs between the official and the revised The revised chapter addresses Compounded Nonsterile Preparations (CNSPs) requiringshorter BUDs and
IBUPROFEN (750 MG)
Catalog # 1335508: Current Lot R13060: Previous Lot R024X0 (Valid Use Date: 31-AUG-2021) SDS View: USP Certificate for Current Lot COVID-19 VACCINE HANDLING TOOLKIT The COVID-19 Vaccine Handling Toolkit is an informational resource and is intended to complement the following resources as well as those provided by the CDC, U.S. FDA, vaccine manufacturers, and state and local departments of health: CDC Vaccine Storage and Handling Official toolkit (February 5, 2021) CHROMATOGRAPHIC COLUMNS The Chromatographic Database is an online compilation of the chromatographic columns used to validate the chromatographic procedures in USP–NF, Pharmacopeial Forum, Revision Bulletins, and Interim Revision Announcements.It provides a cumulative listing of columns referenced in gas and liquid chromatographic methods related to revisions made to USP–NF since January 1980. DISSOLUTION PERFORMANCE VERIFICATION TESTING (PVT) The USP Performance Verification Test (PVT) is an integral part of the General Chapter Dissolution and assesses proper dissolution apparatus performance. PVT is a holistic test and by using the reference standard material and the standard procedure, laboratories can compare results from their instrument with other laboratories worldwide. THE LAST-MILE MANUFACTURING OF VACCINES 3 Th ast-mil anufacturin accines: Recommendations for scaling-up production of COVID-19 vaccines in Africa I. Background Before the SARS-CoV-2 pandemic, Africa was facing REFERENCE STANDARDS: ALPHABETICAL LISTINGS Please note that pre-registration is required to buy Reference Standards. If you're not able to "Select" an item from this list, it means the item is restricted and cannot be ordered online as we first need to ensure that DEA requirements are fulfilled. 1 2 BRIEFING 825 RADIOPHARMACEUTICALS—PREPARATION 1 2 BRIEFING 3 〈825〉 Radiopharmaceuticals—Preparation, Compounding, Dispensing, 4 and Repackaging. Radiopharmaceuticals (radioactive drugs) represent a 5 distinct class of drugs where processing activities include the use of 6 radionuclide generators,preparation and
561 ARTICLES OF BOTANICAL ORIGIN Crude Fiber Exhaust a weighed quantity of the Test Sample, representing about 2g of the drug, with ether.Add 200 mL of boiling dilute sulfuric acid (1 in 78) to the ether-exhausted marc, in a 500-mL flask, and connect the flask to a reflux condenser. 382 ELASTOMERIC CLOSURE FUNCTIONALITY IN INJECTABLE BRIEFING 382 Elastomeric Closure Functionality in Injectable Pharmaceutical Packaging/Delivery Systems. This proposed new general chapter addresses the 905 UNIFORMITY OF DOSAGE UNITS USP34 Stage 6 Harmonization 2 〈905〉 Uniformity of Dosage Units Official December 1, 2011 in conditions of normal use, and express the results as de-in which the terms are as defined in Table 2. livered dose. SALICYLIC ACID (125 MG) Catalog # 1609002: Current Lot K0F112: Previous Lot J3C400 (Valid Use Date: 31-DEC-2007) SDS View: USP Certificate for Current Lot U.S. PHARMACOPEIAABOUT U.S. PHARMACOPEIAREFERENCE STANDARDSSTORECOMPOUNDINGUSP 795OUR IMPACT We are dedicated to helping improve global health through standards setting in compounding, biologics, pharmaceutical manufacturing andother fields.
USP REFERENCE STANDARDS To confirm accuracy and reproducibility, USP Reference Standards are rigorously tested and evaluated by multiple independent laboratories including USP, commercial, regulatory, and academic labs. We also provide publicly available, official documentary standards for pharmaceutical ingredients in the USP–NF that link directly with ourprimary
SEARCH & BUY REFERENCE STANDARDS Search & Buy Reference Standards. The following list of RSs have been assigned a Valid Use Date of October 31, 2020. Due to revisions to USP General Chapter and General Notices. 5.80, which will become official November 1, 2020, users will no longer be able to assume a value of 100.0% for these RS lots in quantitative USP compendialOUR SCIENCE
Our Science. Pharmaceutical science expertise, of our staff and world-wide volunteer experts, is the foundation of USP’s work to help advance public health and ensure the quality and safety of medicines, dietary supplements and foods. The quality standards we develop help manufacturers deliver safe products to billions of peopleworldwide.
LOGIN - USP ACCESS POINT Welcome to Access Point! Use your USP Access Point login credentials to register for events and courses, access your subscriptions to USP's free resources, applications, and more. Note: Please do not bookmark this page. Refer to FAQs. Click here for Access Point Tutorial Create a new USP Access Point account, if you do not have one. COVID-19 VACCINE HANDLING TOOLKIT MAXIMIZING DOSES OF V a ci n e o d i u m C h l o r i d S. 5 1. 5 2. 5 5 3 V a c c i n e. 5 0 5 1. 5 2. 5 5 3 V a ci n e Dilution Date/Time: V a c c i n e. 5 1. 5 2. 5 5 3 HQS162F_2021-01 The standard of trust Dilute the vaccine Continued on next page COVID-19 VACCINE HANDLING TOOLKIT TRANSPORTING COVID-19 HQS162F_2021-01 The standard of trust Pfizer-BioNTech COVID-19 Vaccine 2 Moderna COVID-19 Vaccine3 Janssen Ad26 COVID-19 Vaccine 4 Janssen is a subsidiary of Johnson & Johnson Temperature* Maximum allowable storage time** Temperature* DISSOLUTION PERFORMANCE VERIFICATION TESTING (PVT) The USP Performance Verification Test (PVT) is an integral part of the General Chapter Dissolution and assesses proper dissolution apparatus performance. PVT is a holistic test and by using the reference standard material and the standard procedure, laboratories can compare results from their instrument with other laboratories worldwide. USP COMPOUNDING STANDARDS AND BEYOND-USE DATES (BUDS) USP Compounding Standards and Beyond-Use Dates (BUDs) June 3, 2019 Comparing BUDs between the official and the revised The revised chapter addresses Compounded Nonsterile Preparations (CNSPs) requiringshorter BUDs and
IBUPROFEN (750 MG)
Catalog # 1335508: Current Lot R13060: Previous Lot R024X0 (Valid Use Date: 31-AUG-2021) SDS View: USP Certificate for Current Lot U.S. PHARMACOPEIAABOUT U.S. PHARMACOPEIAREFERENCE STANDARDSSTORECOMPOUNDINGUSP 795OUR IMPACT We are dedicated to helping improve global health through standards setting in compounding, biologics, pharmaceutical manufacturing andother fields.
USP REFERENCE STANDARDS To confirm accuracy and reproducibility, USP Reference Standards are rigorously tested and evaluated by multiple independent laboratories including USP, commercial, regulatory, and academic labs. We also provide publicly available, official documentary standards for pharmaceutical ingredients in the USP–NF that link directly with ourprimary
SEARCH & BUY REFERENCE STANDARDS Search & Buy Reference Standards. The following list of RSs have been assigned a Valid Use Date of October 31, 2020. Due to revisions to USP General Chapter and General Notices. 5.80, which will become official November 1, 2020, users will no longer be able to assume a value of 100.0% for these RS lots in quantitative USP compendialOUR SCIENCE
Our Science. Pharmaceutical science expertise, of our staff and world-wide volunteer experts, is the foundation of USP’s work to help advance public health and ensure the quality and safety of medicines, dietary supplements and foods. The quality standards we develop help manufacturers deliver safe products to billions of peopleworldwide.
LOGIN - USP ACCESS POINT Welcome to Access Point! Use your USP Access Point login credentials to register for events and courses, access your subscriptions to USP's free resources, applications, and more. Note: Please do not bookmark this page. Refer to FAQs. Click here for Access Point Tutorial Create a new USP Access Point account, if you do not have one. COVID-19 VACCINE HANDLING TOOLKIT MAXIMIZING DOSES OF V a ci n e o d i u m C h l o r i d S. 5 1. 5 2. 5 5 3 V a c c i n e. 5 0 5 1. 5 2. 5 5 3 V a ci n e Dilution Date/Time: V a c c i n e. 5 1. 5 2. 5 5 3 HQS162F_2021-01 The standard of trust Dilute the vaccine Continued on next page COVID-19 VACCINE HANDLING TOOLKIT TRANSPORTING COVID-19 HQS162F_2021-01 The standard of trust Pfizer-BioNTech COVID-19 Vaccine 2 Moderna COVID-19 Vaccine3 Janssen Ad26 COVID-19 Vaccine 4 Janssen is a subsidiary of Johnson & Johnson Temperature* Maximum allowable storage time** Temperature* DISSOLUTION PERFORMANCE VERIFICATION TESTING (PVT) The USP Performance Verification Test (PVT) is an integral part of the General Chapter Dissolution and assesses proper dissolution apparatus performance. PVT is a holistic test and by using the reference standard material and the standard procedure, laboratories can compare results from their instrument with other laboratories worldwide. USP COMPOUNDING STANDARDS AND BEYOND-USE DATES (BUDS) USP Compounding Standards and Beyond-Use Dates (BUDs) June 3, 2019 Comparing BUDs between the official and the revised The revised chapter addresses Compounded Nonsterile Preparations (CNSPs) requiringshorter BUDs and
IBUPROFEN (750 MG)
Catalog # 1335508: Current Lot R13060: Previous Lot R024X0 (Valid Use Date: 31-AUG-2021) SDS View: USP Certificate for Current Lot SEARCH & BUY REFERENCE STANDARDS Search & Buy Reference Standards. The following list of RSs have been assigned a Valid Use Date of October 31, 2020. Due to revisions to USP General Chapter and General Notices. 5.80, which will become official November 1, 2020, users will no longer be able to assume a value of 100.0% for these RS lots in quantitative USP compendial COVID-19 VACCINE HANDLING TOOLKIT The COVID-19 Vaccine Handling Toolkit is an informational resource and is intended to complement the following resources as well as those provided by the CDC, U.S. FDA, vaccine manufacturers, and state and local departments of health: CDC Vaccine Storage and Handling Official toolkit (February 5, 2021) CHROMATOGRAPHIC COLUMNS The Chromatographic Database is an online compilation of the chromatographic columns used to validate the chromatographic procedures in USP–NF, Pharmacopeial Forum, Revision Bulletins, and Interim Revision Announcements.It provides a cumulative listing of columns referenced in gas and liquid chromatographic methods related to revisions made to USP–NF since January 1980. DISSOLUTION PERFORMANCE VERIFICATION TESTING (PVT) The USP Performance Verification Test (PVT) is an integral part of the General Chapter Dissolution and assesses proper dissolution apparatus performance. PVT is a holistic test and by using the reference standard material and the standard procedure, laboratories can compare results from their instrument with other laboratories worldwide. THE LAST-MILE MANUFACTURING OF VACCINES 3 Th ast-mil anufacturin accines: Recommendations for scaling-up production of COVID-19 vaccines in Africa I. Background Before the SARS-CoV-2 pandemic, Africa was facing USP GENERAL CHAPTER RADIOPHARMACEUTICALS Developing USP General Chapter USP is a not-for-profit, science-driven organization that has an established process for convening independent experts for the development and maintenance of quality standards. The process is public health focused, leveraging current science and technology, and draws on the expertise of scientists and healthcare practitioners while providing 561 ARTICLES OF BOTANICAL ORIGIN Crude Fiber Exhaust a weighed quantity of the Test Sample, representing about 2g of the drug, with ether.Add 200 mL of boiling dilute sulfuric acid (1 in 78) to the ether-exhausted marc, in a 500-mL flask, and connect the flask to a reflux condenser. 382 ELASTOMERIC CLOSURE FUNCTIONALITY IN INJECTABLE BRIEFING 382 Elastomeric Closure Functionality in Injectable Pharmaceutical Packaging/Delivery Systems. This proposed new general chapter addresses the 905 UNIFORMITY OF DOSAGE UNITS USP34 Stage 6 Harmonization 2 〈905〉 Uniformity of Dosage Units Official December 1, 2011 in conditions of normal use, and express the results as de-in which the terms are as defined in Table 2. livered dose. SALICYLIC ACID (125 MG) Catalog # 1609002: Current Lot K0F112: Previous Lot J3C400 (Valid Use Date: 31-DEC-2007) SDS View: USP Certificate for Current Lot U.S. PHARMACOPEIAABOUT U.S. PHARMACOPEIAREFERENCE STANDARDSSTORECOMPOUNDINGUSP 795OUR IMPACT We are dedicated to helping improve global health through standards setting in compounding, biologics, pharmaceutical manufacturing andother fields.
USP REFERENCE STANDARDS To confirm accuracy and reproducibility, USP Reference Standards are rigorously tested and evaluated by multiple independent laboratories including USP, commercial, regulatory, and academic labs. We also provide publicly available, official documentary standards for pharmaceutical ingredients in the USP–NF that link directly with ourprimary
COVID-19 VACCINE HANDLING TOOLKIT The COVID-19 Vaccine Handling Toolkit is an informational resource and is intended to complement the following resources as well as those provided by the CDC, U.S. FDA, vaccine manufacturers, and state and local departments of health: CDC Vaccine Storage and Handling Official toolkit (February 5, 2021)OUR SCIENCE
Our Science. Pharmaceutical science expertise, of our staff and world-wide volunteer experts, is the foundation of USP’s work to help advance public health and ensure the quality and safety of medicines, dietary supplements and foods. The quality standards we develop help manufacturers deliver safe products to billions of peopleworldwide.
LOGIN - USP ACCESS POINT Welcome to Access Point! Use your USP Access Point login credentials to register for events and courses, access your subscriptions to USP's free resources, applications, and more. Note: Please do not bookmark this page. Refer to FAQs. Click here for Access Point Tutorial Create a new USP Access Point account, if you do not have one. THE LAST-MILE MANUFACTURING OF VACCINES 3 Th ast-mil anufacturin accines: Recommendations for scaling-up production of COVID-19 vaccines in Africa I. Background Before the SARS-CoV-2 pandemic, Africa was facing DISSOLUTION PERFORMANCE VERIFICATION TESTING (PVT) The USP Performance Verification Test (PVT) is an integral part of the General Chapter Dissolution and assesses proper dissolution apparatus performance. PVT is a holistic test and by using the reference standard material and the standard procedure, laboratories can compare results from their instrument with other laboratories worldwide. USP COMPOUNDING STANDARDS AND BEYOND-USE DATES (BUDS) USP Compounding Standards and Beyond-Use Dates (BUDs) June 3, 2019 Comparing BUDs between the official and the revised The revised chapter addresses Compounded Nonsterile Preparations (CNSPs) requiringshorter BUDs and
561 ARTICLES OF BOTANICAL ORIGIN Crude Fiber Exhaust a weighed quantity of the Test Sample, representing about 2g of the drug, with ether.Add 200 mL of boiling dilute sulfuric acid (1 in 78) to the ether-exhausted marc, in a 500-mL flask, and connect the flask to a reflux condenser. DEXAMETHASONE (125 MG) Catalog # 1176007: Current Lot R123C0: Previous Lot R07830 (Valid Use Date: 28-FEB-2021) SDS View: USP Certificate for Current Lot U.S. PHARMACOPEIAABOUT U.S. PHARMACOPEIAREFERENCE STANDARDSSTORECOMPOUNDINGUSP 795OUR IMPACT We are dedicated to helping improve global health through standards setting in compounding, biologics, pharmaceutical manufacturing andother fields.
USP REFERENCE STANDARDS To confirm accuracy and reproducibility, USP Reference Standards are rigorously tested and evaluated by multiple independent laboratories including USP, commercial, regulatory, and academic labs. We also provide publicly available, official documentary standards for pharmaceutical ingredients in the USP–NF that link directly with ourprimary
COVID-19 VACCINE HANDLING TOOLKIT The COVID-19 Vaccine Handling Toolkit is an informational resource and is intended to complement the following resources as well as those provided by the CDC, U.S. FDA, vaccine manufacturers, and state and local departments of health: CDC Vaccine Storage and Handling Official toolkit (February 5, 2021)OUR SCIENCE
Our Science. Pharmaceutical science expertise, of our staff and world-wide volunteer experts, is the foundation of USP’s work to help advance public health and ensure the quality and safety of medicines, dietary supplements and foods. The quality standards we develop help manufacturers deliver safe products to billions of peopleworldwide.
LOGIN - USP ACCESS POINT Welcome to Access Point! Use your USP Access Point login credentials to register for events and courses, access your subscriptions to USP's free resources, applications, and more. Note: Please do not bookmark this page. Refer to FAQs. Click here for Access Point Tutorial Create a new USP Access Point account, if you do not have one. THE LAST-MILE MANUFACTURING OF VACCINES 3 Th ast-mil anufacturin accines: Recommendations for scaling-up production of COVID-19 vaccines in Africa I. Background Before the SARS-CoV-2 pandemic, Africa was facing DISSOLUTION PERFORMANCE VERIFICATION TESTING (PVT) The USP Performance Verification Test (PVT) is an integral part of the General Chapter Dissolution and assesses proper dissolution apparatus performance. PVT is a holistic test and by using the reference standard material and the standard procedure, laboratories can compare results from their instrument with other laboratories worldwide. USP COMPOUNDING STANDARDS AND BEYOND-USE DATES (BUDS) USP Compounding Standards and Beyond-Use Dates (BUDs) June 3, 2019 Comparing BUDs between the official and the revised The revised chapter addresses Compounded Nonsterile Preparations (CNSPs) requiringshorter BUDs and
561 ARTICLES OF BOTANICAL ORIGIN Crude Fiber Exhaust a weighed quantity of the Test Sample, representing about 2g of the drug, with ether.Add 200 mL of boiling dilute sulfuric acid (1 in 78) to the ether-exhausted marc, in a 500-mL flask, and connect the flask to a reflux condenser. DEXAMETHASONE (125 MG) Catalog # 1176007: Current Lot R123C0: Previous Lot R07830 (Valid Use Date: 28-FEB-2021) SDS View: USP Certificate for Current Lot COVID-19 VACCINE HANDLING TOOLKIT The COVID-19 Vaccine Handling Toolkit is an informational resource and is intended to complement the following resources as well as those provided by the CDC, U.S. FDA, vaccine manufacturers, and state and local departments of health: CDC Vaccine Storage and Handling Official toolkit (February 5, 2021) LOGIN - USP ACCESS POINT Welcome to Access Point! Use your USP Access Point login credentials to register for events and courses, access your subscriptions to USP's free resources, applications, and more. Note: Please do not bookmark this page. Refer to FAQs. Click here for Access Point Tutorial Create a new USP Access Point account, if you do not have one. COVID-19 VACCINE HANDLING TOOLKIT MAXIMIZING DOSES OF V a ci n e o d i u m C h l o r i d S. 5 1. 5 2. 5 5 3 V a c c i n e. 5 0 5 1. 5 2. 5 5 3 V a ci n e Dilution Date/Time: V a c c i n e. 5 1. 5 2. 5 5 3 HQS162F_2021-01 The standard of trust Dilute the vaccine Continued on next page DISSOLUTION PERFORMANCE VERIFICATION TESTING (PVT) The USP Performance Verification Test (PVT) is an integral part of the General Chapter Dissolution and assesses proper dissolution apparatus performance. PVT is a holistic test and by using the reference standard material and the standard procedure, laboratories can compare results from their instrument with other laboratories worldwide. THE USP APPROACH FOR SELECTING COLUMNS OF EQUIVALENT The USP Database was developed by the USP Working Group on Column Equivalency using the NIST SRM 870. All data being displayed was generated by the column manufacturers after evaluation of the chromatographic phase. A review was published in Pharmacopeial Forum31
711 DISSOLUTION
Stage 6 Harmonization 2 〈711〉 Dissolution Official December 1, 2011 Figure 1. Basket Stirring Element 2S (USP34) of 25±2 mm between the bottom of the blade and theinside bottom of the vessel is maintained during the test. EPHEDRINE SULFATE (200 MG) (LIST CHEMICAL) restricted item please contact usp customer service at 8002278772 or 3018810666 to speak to a customer service representative for orderassistance.
CYANOCOBALAMIN (CRYSTALLINE) (2 X 150 MG) Purchase Cyanocobalamin RS (crystalline) (Vitamin B12) Reference Standard, (2 x 150 mg), USP-1152011, CAS No. -68-19-9. Has no added substances & is not a 1% mixture in mannitol. Use in USP-NF dietary supplement tests & assays., Order direct for USP service & support. VERAPAMIL HYDROCHLORIDE (200 MG) Catalog # 1711202: Current Lot H0H100: Previous Lot G1E095 (Valid Use Date: 31-JAN-2010) SDS View: USP Certificate for Current Lot SODIUM METABISULFITE (2 X 500 MG) Catalog # 1614396: Current Lot R043N0: Previous Lot F1L076 (Valid Use Date: 31-AUG-2017) SDS View: USP Certificate for Current Lot U.S. PHARMACOPEIAABOUT U.S. PHARMACOPEIAREFERENCE STANDARDSSTORECOMPOUNDINGUSP 795OUR IMPACT We are dedicated to helping improve global health through standards setting in compounding, biologics, pharmaceutical manufacturing andother fields.
USP REFERENCE STANDARDS To confirm accuracy and reproducibility, USP Reference Standards are rigorously tested and evaluated by multiple independent laboratories including USP, commercial, regulatory, and academic labs. We also provide publicly available, official documentary standards for pharmaceutical ingredients in the USP–NF that link directly with ourprimary
COVID-19 VACCINE HANDLING TOOLKIT The COVID-19 Vaccine Handling Toolkit is an informational resource and is intended to complement the following resources as well as those provided by the CDC, U.S. FDA, vaccine manufacturers, and state and local departments of health: CDC Vaccine Storage and Handling Official toolkit (February 5, 2021)OUR SCIENCE
Our Science. Pharmaceutical science expertise, of our staff and world-wide volunteer experts, is the foundation of USP’s work to help advance public health and ensure the quality and safety of medicines, dietary supplements and foods. The quality standards we develop help manufacturers deliver safe products to billions of peopleworldwide.
LOGIN - USP ACCESS POINT Welcome to Access Point! Use your USP Access Point login credentials to register for events and courses, access your subscriptions to USP's free resources, applications, and more. Note: Please do not bookmark this page. Refer to FAQs. Click here for Access Point Tutorial Create a new USP Access Point account, if you do not have one. THE LAST-MILE MANUFACTURING OF VACCINES 3 Th ast-mil anufacturin accines: Recommendations for scaling-up production of COVID-19 vaccines in Africa I. Background Before the SARS-CoV-2 pandemic, Africa was facing DISSOLUTION PERFORMANCE VERIFICATION TESTING (PVT) The USP Performance Verification Test (PVT) is an integral part of the General Chapter Dissolution and assesses proper dissolution apparatus performance. PVT is a holistic test and by using the reference standard material and the standard procedure, laboratories can compare results from their instrument with other laboratories worldwide. USP COMPOUNDING STANDARDS AND BEYOND-USE DATES (BUDS) USP Compounding Standards and Beyond-Use Dates (BUDs) June 3, 2019 Comparing BUDs between the official and the revised The revised chapter addresses Compounded Nonsterile Preparations (CNSPs) requiringshorter BUDs and
561 ARTICLES OF BOTANICAL ORIGIN Crude Fiber Exhaust a weighed quantity of the Test Sample, representing about 2g of the drug, with ether.Add 200 mL of boiling dilute sulfuric acid (1 in 78) to the ether-exhausted marc, in a 500-mL flask, and connect the flask to a reflux condenser. DEXAMETHASONE (125 MG) Catalog # 1176007: Current Lot R123C0: Previous Lot R07830 (Valid Use Date: 28-FEB-2021) SDS View: USP Certificate for Current Lot U.S. PHARMACOPEIAABOUT U.S. PHARMACOPEIAREFERENCE STANDARDSSTORECOMPOUNDINGUSP 795OUR IMPACT We are dedicated to helping improve global health through standards setting in compounding, biologics, pharmaceutical manufacturing andother fields.
USP REFERENCE STANDARDS To confirm accuracy and reproducibility, USP Reference Standards are rigorously tested and evaluated by multiple independent laboratories including USP, commercial, regulatory, and academic labs. We also provide publicly available, official documentary standards for pharmaceutical ingredients in the USP–NF that link directly with ourprimary
COVID-19 VACCINE HANDLING TOOLKIT The COVID-19 Vaccine Handling Toolkit is an informational resource and is intended to complement the following resources as well as those provided by the CDC, U.S. FDA, vaccine manufacturers, and state and local departments of health: CDC Vaccine Storage and Handling Official toolkit (February 5, 2021)OUR SCIENCE
Our Science. Pharmaceutical science expertise, of our staff and world-wide volunteer experts, is the foundation of USP’s work to help advance public health and ensure the quality and safety of medicines, dietary supplements and foods. The quality standards we develop help manufacturers deliver safe products to billions of peopleworldwide.
LOGIN - USP ACCESS POINT Welcome to Access Point! Use your USP Access Point login credentials to register for events and courses, access your subscriptions to USP's free resources, applications, and more. Note: Please do not bookmark this page. Refer to FAQs. Click here for Access Point Tutorial Create a new USP Access Point account, if you do not have one. THE LAST-MILE MANUFACTURING OF VACCINES 3 Th ast-mil anufacturin accines: Recommendations for scaling-up production of COVID-19 vaccines in Africa I. Background Before the SARS-CoV-2 pandemic, Africa was facing DISSOLUTION PERFORMANCE VERIFICATION TESTING (PVT) The USP Performance Verification Test (PVT) is an integral part of the General Chapter Dissolution and assesses proper dissolution apparatus performance. PVT is a holistic test and by using the reference standard material and the standard procedure, laboratories can compare results from their instrument with other laboratories worldwide. USP COMPOUNDING STANDARDS AND BEYOND-USE DATES (BUDS) USP Compounding Standards and Beyond-Use Dates (BUDs) June 3, 2019 Comparing BUDs between the official and the revised The revised chapter addresses Compounded Nonsterile Preparations (CNSPs) requiringshorter BUDs and
561 ARTICLES OF BOTANICAL ORIGIN Crude Fiber Exhaust a weighed quantity of the Test Sample, representing about 2g of the drug, with ether.Add 200 mL of boiling dilute sulfuric acid (1 in 78) to the ether-exhausted marc, in a 500-mL flask, and connect the flask to a reflux condenser. DEXAMETHASONE (125 MG) Catalog # 1176007: Current Lot R123C0: Previous Lot R07830 (Valid Use Date: 28-FEB-2021) SDS View: USP Certificate for Current Lot COVID-19 VACCINE HANDLING TOOLKIT The COVID-19 Vaccine Handling Toolkit is an informational resource and is intended to complement the following resources as well as those provided by the CDC, U.S. FDA, vaccine manufacturers, and state and local departments of health: CDC Vaccine Storage and Handling Official toolkit (February 5, 2021) LOGIN - USP ACCESS POINT Welcome to Access Point! Use your USP Access Point login credentials to register for events and courses, access your subscriptions to USP's free resources, applications, and more. Note: Please do not bookmark this page. Refer to FAQs. Click here for Access Point Tutorial Create a new USP Access Point account, if you do not have one. COVID-19 VACCINE HANDLING TOOLKIT MAXIMIZING DOSES OF V a ci n e o d i u m C h l o r i d S. 5 1. 5 2. 5 5 3 V a c c i n e. 5 0 5 1. 5 2. 5 5 3 V a ci n e Dilution Date/Time: V a c c i n e. 5 1. 5 2. 5 5 3 HQS162F_2021-01 The standard of trust Dilute the vaccine Continued on next page DISSOLUTION PERFORMANCE VERIFICATION TESTING (PVT) The USP Performance Verification Test (PVT) is an integral part of the General Chapter Dissolution and assesses proper dissolution apparatus performance. PVT is a holistic test and by using the reference standard material and the standard procedure, laboratories can compare results from their instrument with other laboratories worldwide. THE USP APPROACH FOR SELECTING COLUMNS OF EQUIVALENT The USP Database was developed by the USP Working Group on Column Equivalency using the NIST SRM 870. All data being displayed was generated by the column manufacturers after evaluation of the chromatographic phase. A review was published in Pharmacopeial Forum31
711 DISSOLUTION
Stage 6 Harmonization 2 〈711〉 Dissolution Official December 1, 2011 Figure 1. Basket Stirring Element 2S (USP34) of 25±2 mm between the bottom of the blade and theinside bottom of the vessel is maintained during the test. EPHEDRINE SULFATE (200 MG) (LIST CHEMICAL) restricted item please contact usp customer service at 8002278772 or 3018810666 to speak to a customer service representative for orderassistance.
CYANOCOBALAMIN (CRYSTALLINE) (2 X 150 MG) Purchase Cyanocobalamin RS (crystalline) (Vitamin B12) Reference Standard, (2 x 150 mg), USP-1152011, CAS No. -68-19-9. Has no added substances & is not a 1% mixture in mannitol. Use in USP-NF dietary supplement tests & assays., Order direct for USP service & support. VERAPAMIL HYDROCHLORIDE (200 MG) Catalog # 1711202: Current Lot H0H100: Previous Lot G1E095 (Valid Use Date: 31-JAN-2010) SDS View: USP Certificate for Current Lot SODIUM METABISULFITE (2 X 500 MG) Catalog # 1614396: Current Lot R043N0: Previous Lot F1L076 (Valid Use Date: 31-AUG-2017) SDS View: USP Certificate for Current Lot CONTROVERSIAL LEGISLATIVE PROVISION THREATENS PATIENT SAFETYLearn more
PROTECTING PEOPLE FROM POOR-QUALITY MEDICINES Learn about the Promoting the Quality of Medicines (PQM) program, funded by USAID and implemented by USP. Get more information HELPING TO ADDRESS THE GROWING GLOBAL THREAT OF ANTIMICROBIALRESISTANCE
Learn more about USP's effortsPrevious Next
Fulltext search
GLOBAL PRESENCE
* USP–Brazil
* USP–China
* USP–Ethiopia
* USP–Ghana
* USP–India
* USP–Indonesia
* USP–Nigeria
* USP–Pakistan
* USP–Philippines
* USP–Switzerland
Select
CountryUSP–BrazilUSP–ChinaUSP–EthiopiaUSP–GhanaUSP–IndiaUSP–IndonesiaUSP–NigeriaUSP–PakistanUSP–PhilippinesUSP–SwitzerlandLogin
* USP-NF Online
* FCC Online
* PF Online
* PF Online (Legacy)* FCC Forum
* CoE Portal
* USP Dictionary
* SDS Online
* USP Compounding
* USP Donor Portal
* USP Education
* Chromatographic Columns* DSC Online
SOCIAL MEDIA
* Linked In
* You Tube
* QualityMatters BlogTOP MENU
* Careers
* Store
* Help
* FAQs
* USP en Español
* Contact Us
* About
ABOUT USP
* About USP
* Convention Membership* Board of Trustees
* Leadership
* USP Volunteers
* Policies & Rules
* Careers
* Newsroom
* Public Policy
* Legal Recognition
* Annual Report
* Our Impact
OUR IMPACT
* Our Impact
* Antimicrobial Resistance * Generics Access Plan * Medicines We Can Trust * Promoting the Quality of Medicines Program (PQM)* Quality Institute
* True Impact Stories* Our Work
OUR WORK OVERVIEW
* Biologics
* Chemical Medicines * Compounding Standards * Dietary Supplements & Herbal Medicines* Excipients
* Food Safety & Integrity * Global Public Health * Harmonized Standards * Healthcare Quality & Safety * Reference Standards * Research & Innovation * Products & Services PRODUCTS & SERVICES OVERVIEW* Products
* Reference Standards* USP-NF
* Food Chemicals Codex * Dietary Supplements Compendium * USP Compounding Compendium* All Products
* Services
* Education Courses
* Verification Services * USP Academic Connection* Resources
* Authorized Distributors* Compendial Tools
* Dissolution
* Chromatographic Columns * Herbal Medicines Compendium * Medicines Quality Database * Safety Data Sheets * Newsletters & Updates* Events & Training
EVENTS & TRAINING
* Events & Training
* Search Events & Training* Courses
* Workshops
* User Forums
* USP at Events
* Stakeholder Forums * Expert Committee Meetings* Get Involved
GET INVOLVED OVERVIEW* Partner
* New or Revised Standards * Reference Standards * Compounding Monographs * Submission Guidelines* Donor Recognition
* Volunteer
* How to Become a Volunteer * View Volunteer Opportunities* Provide Input
* Comment on Proposed Standards* Review Work Plans
* Observe a Committee Meeting * Stakeholder Forums Empowering a healthy tomorrow A HEALTHIER WORLD NEEDS A STRONG FOUNDATION – ONE THAT ESTABLISHES QUALITY, SETS THE BAR FOR SCIENTIFIC RIGOR AND TECHNOLOGICAL PROGRESS, AND EPITOMIZES COLLABORATION BETWEEN INDUSTRY, NONPROFITS, GOVERNMENT AND ACADEMIA. FROM THE STANDARDS WE CREATE TO THE PARTNERSHIPS AND CONVERSATIONS WE FOSTER, OUR SCIENTISTS, ADVOCATES AND NETWORK OF EXPERTS ARE CRITICAL TO CONSTRUCTING AND REINFORCING THIS FOUNDATION TO ENSURE PEOPLE STAYHEALTHY.
/usp/video/USP_Anthem.mp4Learn more
LATEST STANDARDS UPDATESUSP-NFACCESS USP-NF
Role and applicability of USP General Chapters related to Compounding and Safe Handling of Hazardous Drugs(posted
30–Jul–2019)
------------------------- One new Revision Bulletins(posted
28–Jun–2019; Official 01–Jul–2019) ------------------------- Cumulative list updated (posted 28–Jun–2019) ------------------------- <797> Commentary updated(posted
28–Jun–2019)
REFERENCE STANDARDSPURCHASE Download full list as: EXCELRead the blog
USP volunteers help shape the future of drug manufacturing Consistent quality. Greater access. They’re what patients deserve and what USP strives to ensure across the globe through the development of public quality standards.Read more
Containing drug costs in the United States Building the marketplace for generic medicines is a key strategy to containing drug costs in the US, one that USP actively supports through our standards-setting work.Read more
USP volunteers help shape the future of drug manufacturing Consistent quality. Greater access. They’re what patients deserve and what USP strives to ensure across the globe through the development of public quality standards.Read more
Containing drug costs in the United States Building the marketplace for generic medicines is a key strategy to containing drug costs in the US, one that USP actively supports through our standards-setting work.Read more
Our legacy of championing access to medicines Ron Piervincenzi, Ph.D., shares his thoughts on USP receiving the Association of Accessible Medicines (AAM) Champion of Access award.Read the blog
Containing drug costs in the United States Building the marketplace for generic medicines is a key strategy to containing drug costs in the US, one that USP actively supports through our standards-setting work.Read more
Our legacy of championing access to medicines Ron Piervincenzi, Ph.D., shares his thoughts on USP receiving the Association of Accessible Medicines (AAM) Champion of Access award.Read the blog
USP volunteers help shape the future of drug manufacturing Consistent quality. Greater access. They’re what patients deserve and what USP strives to ensure across the globe through the development of public quality standards.Read more
Previous Next
TWEETS BY @USPHARMACOPEIAFalse
UPCOMING EVENTS & TRAINING Dietary Supplement - Verification Program Overview (On-Demand)Learn more...
View All Events & Training FOOTER SITE MAP (MAIN NAVIGATION)* About
* About USP
* Convention Membership* Board of Trustees
* Leadership
* USP Volunteers
* Policies & Rules
* Careers
* Newsroom
* Public Policy
* Legal Recognition
* Annual Report
* Our Impact
* Our Impact
* Antimicrobial Resistance * Generics Access Plan * Medicines We Can Trust * Promoting the Quality of Medicines Program (PQM)* Quality Institute
* True Impact Stories* Our Work
* Biologics
* Chemical Medicines * Compounding Standards * Dietary Supplements & Herbal Medicines* Excipients
* Food Safety & Integrity * Global Public Health * Harmonized Standards * Healthcare Quality & Safety * Reference Standards * Research & Innovation * Products & Services* Products
* Services
* Resources
* Events & Training
* Events & Training
* Search Events & Training* Courses
* Workshops
* User Forums
* USP at Events
* Stakeholder Forums * Expert Committee Meetings* Get Involved
* Partner
* Volunteer
* Provide Input
SOCIAL MEDIA
* Linked In
* You Tube
* QualityMatters BlogBOTTOM MENU
* Contact Us
* Code of Ethics
* Legal Notices
* Privacy Policy
* Terms of Use
* Sitemap
* ">© The United States Pharmacopeial Convention×
×
Back to top
Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0