Are you over 18 and want to see adult content?
More Annotations
Смотреть порно видео онлайн бесплатно в хорошем качестве
Are you over 18 and want to see adult content?
Home - Australian Securities Exchange - ASX
Are you over 18 and want to see adult content?
TV Forum - Discussing television presentation and channel branding
Are you over 18 and want to see adult content?

TravelNetto - Urlaub, Kur und Spa-Hotels in Polen - Polnische Ostsee
Are you over 18 and want to see adult content?
Gardenzzz | Helping you discover a WORLD of new outdoor products
Are you over 18 and want to see adult content?
Scam Review Scan -Trusted Consumer Reviews
Are you over 18 and want to see adult content?
Cukrářské potřeby, suroviny a pomůcky na pečení Brno
Are you over 18 and want to see adult content?

Info Pendaftaran STAN bisa dilihat DI WEB ini - ujiansma.com
Are you over 18 and want to see adult content?
Favourite Annotations
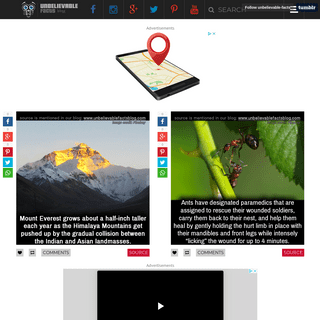
A complete backup of unbelievablefactsblog.com
Are you over 18 and want to see adult content?

A complete backup of solomonsseal.wordpress.com
Are you over 18 and want to see adult content?
A complete backup of babyshopstores.com
Are you over 18 and want to see adult content?
A complete backup of lastwordonprofootball.com
Are you over 18 and want to see adult content?

A complete backup of lightnowblog.com
Are you over 18 and want to see adult content?

A complete backup of recambioscoche.es
Are you over 18 and want to see adult content?
A complete backup of ceaufuse37.wordpress.com
Are you over 18 and want to see adult content?

A complete backup of wideawakegentile.wordpress.com
Are you over 18 and want to see adult content?
Text
infection.
CLINICAL LABORATORY EVALUATION PROGRAM How can the New York State Department of Health Clinical Laboratory Evaluation Program help you today? Laboratory Standards CLEP Program Guide (July 2020) Apply for a Permit Permit Modifications Health Commerce System (HCS) Limited Service Laboratories Certificate of Qualification (CQ) Applications Cytotech Registrations Lab Developed Test (LDT) Approval On-Site Survey LABORATORY STANDARDS Revisions to Laboratory Standards - May 2021. Summaries showing revisions to the General Systems Standards and Specialty Requirements by Category, for Microbiology and Cytopathology, are shown below. Changes are effective May 5, 2021. Also available is a side-by-side comparison of the former General Systems Standards, prior to August2020, and
MEDICAL MARIJUANA
REVISED May 2019 - Solicitation of Interest for Laboratories to Perform Testing of Medical Marijuana Products in New York State Guidance Procedure Microbiology - Medical Marijuana guidance procedure for a DOC Methods List of Approved Methods and Analytes - Certification Manual Item 180.7 NYS DOH LINC-250, revision 2 - Metals and Metalloids in Medical Marijuana Products bySENIOR STAFF
Director and Quality Assurance Officer, Environmental Inorganic Chemistry Laboratory; Derek Symula, Ph.D. Director, Division of Laboratory Quality Certification CERTIFICATE OF QUALIFICATION & LABORATORY DIRECTORSEE MORE ONWADSWORTH.ORG
INITIAL APPLICATION
These items are required for initial application: CLIA Application for Certification Form CMS-116 The facility name on the application must match the name of your business entity registered with the IRS for the facility's federal tax identification number (e.g., EIN, TIN). If your facility is doing business as (dba) under a name that is not the same as the facility name, you must provide this SPINAL CORD INJURY RESEARCH The Extramural Grants Administration Program works to further the efforts of the Spinal Cord Injury Research Board (SCIRB). Each year, approximately 1,100 New York residents suffer traumatic spinal cord injuries joining the nearly 288,000 people living in the United States with paralysis. SCIRB’s mission, since 1998, has been to support neurological spinal cord injury GUIDELINES FOR REMOTE BLOOD STORAGE NEW YORK STATE COUNCIL ON HUMAN BLOOD AND TRANSFUSION SERVICES Membership Roster – 2008 Donna Skerrett, M.D., M.S., Chairperson * Director, Transfusion Medicine and WADSWORTH CENTER, NEW YORK STATE DEPARTMENT OF HEALTH 4. Federal requirement. Section 493.43 of the CLIA regulations state that "except as specified in paragraph (b) of this section, all laboratories performing non-waived testing WADSWORTH CENTER, NEW YORK STATE DEPARTMENT OF HEALTHPUBLIC HEALTH PROGRAMSRESEARCHREGULATORY PROGRAMSEDUCATIONEXTRAMURAL FUNDINGABOUT US Wadsworth Center, at the forefront of the fight against tuberculosis in NYS and globally, celebrates World TB Day 2021. The World Health Organization estimates that almost 4,000 people lose their lives to TB and close to 28,000 people contract TB every day. Currently, 13 million people in the United States live with latent (hidden) TBinfection.
CLINICAL LABORATORY EVALUATION PROGRAM How can the New York State Department of Health Clinical Laboratory Evaluation Program help you today? Laboratory Standards CLEP Program Guide (July 2020) Apply for a Permit Permit Modifications Health Commerce System (HCS) Limited Service Laboratories Certificate of Qualification (CQ) Applications Cytotech Registrations Lab Developed Test (LDT) Approval On-Site Survey LABORATORY STANDARDS Revisions to Laboratory Standards - May 2021. Summaries showing revisions to the General Systems Standards and Specialty Requirements by Category, for Microbiology and Cytopathology, are shown below. Changes are effective May 5, 2021. Also available is a side-by-side comparison of the former General Systems Standards, prior to August2020, and
MEDICAL MARIJUANA
REVISED May 2019 - Solicitation of Interest for Laboratories to Perform Testing of Medical Marijuana Products in New York State Guidance Procedure Microbiology - Medical Marijuana guidance procedure for a DOC Methods List of Approved Methods and Analytes - Certification Manual Item 180.7 NYS DOH LINC-250, revision 2 - Metals and Metalloids in Medical Marijuana Products bySENIOR STAFF
Director and Quality Assurance Officer, Environmental Inorganic Chemistry Laboratory; Derek Symula, Ph.D. Director, Division of Laboratory Quality Certification CERTIFICATE OF QUALIFICATION & LABORATORY DIRECTORSEE MORE ONWADSWORTH.ORG
INITIAL APPLICATION
These items are required for initial application: CLIA Application for Certification Form CMS-116 The facility name on the application must match the name of your business entity registered with the IRS for the facility's federal tax identification number (e.g., EIN, TIN). If your facility is doing business as (dba) under a name that is not the same as the facility name, you must provide this SPINAL CORD INJURY RESEARCH The Extramural Grants Administration Program works to further the efforts of the Spinal Cord Injury Research Board (SCIRB). Each year, approximately 1,100 New York residents suffer traumatic spinal cord injuries joining the nearly 288,000 people living in the United States with paralysis. SCIRB’s mission, since 1998, has been to support neurological spinal cord injury GUIDELINES FOR REMOTE BLOOD STORAGE NEW YORK STATE COUNCIL ON HUMAN BLOOD AND TRANSFUSION SERVICES Membership Roster – 2008 Donna Skerrett, M.D., M.S., Chairperson * Director, Transfusion Medicine and WADSWORTH CENTER, NEW YORK STATE DEPARTMENT OF HEALTH 4. Federal requirement. Section 493.43 of the CLIA regulations state that "except as specified in paragraph (b) of this section, all laboratories performing non-waived testing ACCU REFERENCE MEDICAL LAB LLC Department of Health, Wadsworth Center. Public Health Programs . Environmental Health CYTOTECHNOLOGIST REGISTRATION New York State Public Health Law requires that all individuals employed as cytotechnologists at clinical laboratories applying for or holding a New York State permit be registered with the Department. New York State Education Law also requires that cytotechnologists be licensed. More information regarding licensure is available at State Education Department, Office of theKIMXS3CEIMMLIM
Department of Health, Wadsworth Center. Public Health Programs . Environmental Health NEW YORK STATE NEWBORN SCREENING PROGRAM Newborn screening refers to medical tests, the majority of which are genetic, performed to identify babies with certain disorders, which without intervention, may permanently impact newborns and their families. Early recognition and treatment of most of these disorders leads to a better outcome for the newborn. The Newborn Screening Program’s goal is to help affected babies LDT FAQ | NEW YORK STATE DEPARTMENT OF HEALTH, WADSWORTH I know a laboratory offers a test, but it does not show up in the search results. Try modifying your search terms or spelling. For example: searching for “genetics” in the ‘Analyte or Test Name’ field will return all tests that contain the word “genetics”, such as pharmacogenetics, but not pharmacogenetic, whereas searching for “genetic” will return tests with eitherword, i.e
ENCEPHALITIS
Viral encephalitis encompasses a group of inflammatory diseases of the brain caused by a variety of viral agents and exhibiting common clinical symptoms. Symptoms may include headache, confusion, or other alterations in sensorium and / or behavior, nausea, or vomiting. Signs may include evidence of elevated intracranial pressure or meningeal irritation, cranial nerve palsies, paresis or FORMS | NEW YORK STATE DEPARTMENT OF HEALTH, WADSWORTH CENTER General Request Newborn Screening Materials Cystic Fibrosis Referral Appointment Confirmation Form Follow-Up Summary Form Hemoglobin Referral Appointment Confirmation Form Newborn Screening Transport Form Refusal of Diagnostic Testing for Cystic Fibrosis Form Refusal of Newborn Screening for Religious Reasons Form Request for Newborn Screening Results Diagnostic MICHELE CAGGANA, SC.D., FACMG Dr. Michele Caggana joined the Wadsworth Center in 1996 after receiving her doctoral degree from the Harvard School of Public Health and completing post-doctoral work in molecular virology at Wadsworth Center and clinical molecular genetics at the Mt. Sinai School of Medicine. Dr. Caggana is Chief of the Laboratory of Human Genetics and was appointed as Director of the Newborn KIRSTEN ST. GEORGE, PH.D. Dr. St. George is Director of Virology and Chief of the Laboratory of Viral Diseases (LVD) at the Wadsworth Center and a Clinical Professor in the Biomedical Sciences Department at the School of Public Health, University at Albany. The LVD comprises several service laboratories and research teams including general virology and specialty referenceservices in viral
NEW YORK STATE DEPARTMENT OF HEALTH WADSWORTH … A. Has the director, any assistant director(s), or those having a direct or indirect ownership interest, controlling interest, or corporate membership in the applying clinical facility ever been charged with violations of local, state or federal WADSWORTH CENTER, NEW YORK STATE DEPARTMENT OF HEALTHPUBLIC HEALTH PROGRAMSRESEARCHREGULATORY PROGRAMSEDUCATIONEXTRAMURAL FUNDINGABOUT US Wadsworth Center, at the forefront of the fight against tuberculosis in NYS and globally, celebrates World TB Day 2021. The World Health Organization estimates that almost 4,000 people lose their lives to TB and close to 28,000 people contract TB every day. Currently, 13 million people in the United States live with latent (hidden) TBinfection.
CLINICAL LABORATORY EVALUATION PROGRAM How can the New York State Department of Health Clinical Laboratory Evaluation Program help you today? Laboratory Standards CLEP Program Guide (July 2020) Apply for a Permit Permit Modifications Health Commerce System (HCS) Limited Service Laboratories Certificate of Qualification (CQ) Applications Cytotech Registrations Lab Developed Test (LDT) Approval On-Site Survey LABORATORY STANDARDS Revisions to Laboratory Standards - May 2021. Summaries showing revisions to the General Systems Standards and Specialty Requirements by Category, for Microbiology and Cytopathology, are shown below. Changes are effective May 5, 2021. Also available is a side-by-side comparison of the former General Systems Standards, prior to August2020, and
MEDICAL MARIJUANA
REVISED May 2019 - Solicitation of Interest for Laboratories to Perform Testing of Medical Marijuana Products in New York State Guidance Procedure Microbiology - Medical Marijuana guidance procedure for a DOC Methods List of Approved Methods and Analytes - Certification Manual Item 180.7 NYS DOH LINC-250, revision 2 - Metals and Metalloids in Medical Marijuana Products bySENIOR STAFF
Director and Quality Assurance Officer, Environmental Inorganic Chemistry Laboratory; Derek Symula, Ph.D. Director, Division of Laboratory Quality Certification CERTIFICATE OF QUALIFICATION & LABORATORY DIRECTORSEE MORE ONWADSWORTH.ORG
INITIAL APPLICATION
These items are required for initial application: CLIA Application for Certification Form CMS-116 The facility name on the application must match the name of your business entity registered with the IRS for the facility's federal tax identification number (e.g., EIN, TIN). If your facility is doing business as (dba) under a name that is not the same as the facility name, you must provide this SPINAL CORD INJURY RESEARCH The Extramural Grants Administration Program works to further the efforts of the Spinal Cord Injury Research Board (SCIRB). Each year, approximately 1,100 New York residents suffer traumatic spinal cord injuries joining the nearly 288,000 people living in the United States with paralysis. SCIRB’s mission, since 1998, has been to support neurological spinal cord injury GUIDELINES FOR REMOTE BLOOD STORAGE NEW YORK STATE COUNCIL ON HUMAN BLOOD AND TRANSFUSION SERVICES Membership Roster – 2008 Donna Skerrett, M.D., M.S., Chairperson * Director, Transfusion Medicine and WADSWORTH CENTER, NEW YORK STATE DEPARTMENT OF HEALTH 4. Federal requirement. Section 493.43 of the CLIA regulations state that "except as specified in paragraph (b) of this section, all laboratories performing non-waived testing WADSWORTH CENTER, NEW YORK STATE DEPARTMENT OF HEALTHPUBLIC HEALTH PROGRAMSRESEARCHREGULATORY PROGRAMSEDUCATIONEXTRAMURAL FUNDINGABOUT US Wadsworth Center, at the forefront of the fight against tuberculosis in NYS and globally, celebrates World TB Day 2021. The World Health Organization estimates that almost 4,000 people lose their lives to TB and close to 28,000 people contract TB every day. Currently, 13 million people in the United States live with latent (hidden) TBinfection.
CLINICAL LABORATORY EVALUATION PROGRAM How can the New York State Department of Health Clinical Laboratory Evaluation Program help you today? Laboratory Standards CLEP Program Guide (July 2020) Apply for a Permit Permit Modifications Health Commerce System (HCS) Limited Service Laboratories Certificate of Qualification (CQ) Applications Cytotech Registrations Lab Developed Test (LDT) Approval On-Site Survey LABORATORY STANDARDS Revisions to Laboratory Standards - May 2021. Summaries showing revisions to the General Systems Standards and Specialty Requirements by Category, for Microbiology and Cytopathology, are shown below. Changes are effective May 5, 2021. Also available is a side-by-side comparison of the former General Systems Standards, prior to August2020, and
MEDICAL MARIJUANA
REVISED May 2019 - Solicitation of Interest for Laboratories to Perform Testing of Medical Marijuana Products in New York State Guidance Procedure Microbiology - Medical Marijuana guidance procedure for a DOC Methods List of Approved Methods and Analytes - Certification Manual Item 180.7 NYS DOH LINC-250, revision 2 - Metals and Metalloids in Medical Marijuana Products bySENIOR STAFF
Director and Quality Assurance Officer, Environmental Inorganic Chemistry Laboratory; Derek Symula, Ph.D. Director, Division of Laboratory Quality Certification CERTIFICATE OF QUALIFICATION & LABORATORY DIRECTORSEE MORE ONWADSWORTH.ORG
INITIAL APPLICATION
These items are required for initial application: CLIA Application for Certification Form CMS-116 The facility name on the application must match the name of your business entity registered with the IRS for the facility's federal tax identification number (e.g., EIN, TIN). If your facility is doing business as (dba) under a name that is not the same as the facility name, you must provide this SPINAL CORD INJURY RESEARCH The Extramural Grants Administration Program works to further the efforts of the Spinal Cord Injury Research Board (SCIRB). Each year, approximately 1,100 New York residents suffer traumatic spinal cord injuries joining the nearly 288,000 people living in the United States with paralysis. SCIRB’s mission, since 1998, has been to support neurological spinal cord injury GUIDELINES FOR REMOTE BLOOD STORAGE NEW YORK STATE COUNCIL ON HUMAN BLOOD AND TRANSFUSION SERVICES Membership Roster – 2008 Donna Skerrett, M.D., M.S., Chairperson * Director, Transfusion Medicine and WADSWORTH CENTER, NEW YORK STATE DEPARTMENT OF HEALTH 4. Federal requirement. Section 493.43 of the CLIA regulations state that "except as specified in paragraph (b) of this section, all laboratories performing non-waived testing ACCU REFERENCE MEDICAL LAB LLC Department of Health, Wadsworth Center. Public Health Programs . Environmental Health CYTOTECHNOLOGIST REGISTRATION New York State Public Health Law requires that all individuals employed as cytotechnologists at clinical laboratories applying for or holding a New York State permit be registered with the Department. New York State Education Law also requires that cytotechnologists be licensed. More information regarding licensure is available at State Education Department, Office of theKIMXS3CEIMMLIM
Department of Health, Wadsworth Center. Public Health Programs . Environmental Health NEW YORK STATE NEWBORN SCREENING PROGRAM Newborn screening refers to medical tests, the majority of which are genetic, performed to identify babies with certain disorders, which without intervention, may permanently impact newborns and their families. Early recognition and treatment of most of these disorders leads to a better outcome for the newborn. The Newborn Screening Program’s goal is to help affected babies LDT FAQ | NEW YORK STATE DEPARTMENT OF HEALTH, WADSWORTH I know a laboratory offers a test, but it does not show up in the search results. Try modifying your search terms or spelling. For example: searching for “genetics” in the ‘Analyte or Test Name’ field will return all tests that contain the word “genetics”, such as pharmacogenetics, but not pharmacogenetic, whereas searching for “genetic” will return tests with eitherword, i.e
ENCEPHALITIS
Viral encephalitis encompasses a group of inflammatory diseases of the brain caused by a variety of viral agents and exhibiting common clinical symptoms. Symptoms may include headache, confusion, or other alterations in sensorium and / or behavior, nausea, or vomiting. Signs may include evidence of elevated intracranial pressure or meningeal irritation, cranial nerve palsies, paresis or FORMS | NEW YORK STATE DEPARTMENT OF HEALTH, WADSWORTH CENTER General Request Newborn Screening Materials Cystic Fibrosis Referral Appointment Confirmation Form Follow-Up Summary Form Hemoglobin Referral Appointment Confirmation Form Newborn Screening Transport Form Refusal of Diagnostic Testing for Cystic Fibrosis Form Refusal of Newborn Screening for Religious Reasons Form Request for Newborn Screening Results Diagnostic MICHELE CAGGANA, SC.D., FACMG Dr. Michele Caggana joined the Wadsworth Center in 1996 after receiving her doctoral degree from the Harvard School of Public Health and completing post-doctoral work in molecular virology at Wadsworth Center and clinical molecular genetics at the Mt. Sinai School of Medicine. Dr. Caggana is Chief of the Laboratory of Human Genetics and was appointed as Director of the Newborn KIRSTEN ST. GEORGE, PH.D. Dr. St. George is Director of Virology and Chief of the Laboratory of Viral Diseases (LVD) at the Wadsworth Center and a Clinical Professor in the Biomedical Sciences Department at the School of Public Health, University at Albany. The LVD comprises several service laboratories and research teams including general virology and specialty referenceservices in viral
NEW YORK STATE DEPARTMENT OF HEALTH WADSWORTH … A. Has the director, any assistant director(s), or those having a direct or indirect ownership interest, controlling interest, or corporate membership in the applying clinical facility ever been charged with violations of local, state or federal WADSWORTH CENTER, NEW YORK STATE DEPARTMENT OF HEALTHPUBLIC HEALTH PROGRAMSRESEARCHREGULATORY PROGRAMSEDUCATIONEXTRAMURAL FUNDINGABOUT US Wadsworth Center, at the forefront of the fight against tuberculosis in NYS and globally, celebrates World TB Day 2021 The World Health Organization estimates that almost 4,000 people lose their lives to TB and close to 28,000 people contract TB every day. CLINICAL LABORATORY EVALUATION PROGRAM How can the New York State Department of Health Clinical Laboratory Evaluation Program help you today? Laboratory Standards CLEP Program Guide (July 2020) Apply for a Permit Permit Modifications Health Commerce System (HCS) Limited Service Laboratories Certificate of Qualification (CQ) Applications Cytotech Registrations Lab Developed Test (LDT) Approval On-Site Survey LABORATORY STANDARDS Current Standards May 2021 revisions to the General Systems Standards and Specialty Requirements by Category, for Microbiology and Cytopathology, may be viewed below under the heading of Revisions to Laboratory Standards - May 2021.SENIOR STAFF
Director and Quality Assurance Officer, Environmental Inorganic Chemistry Laboratory; Derek Symula, Ph.D. Director, Division of Laboratory Quality Certification BLOOD RESOURCES PROGRAM More than 350 facilities are approved by the department to provide blood services. One and a quarter million blood components are prepared annually. Approximately 90% of the blood is collected at blood centers, with the remainder collected by approximately about a dozen hospitals and a small number of independent facilities. Informational materials on blood services, including guidelines andINITIAL APPLICATION
These items are required for initial application: CLIA Application for Certification Form CMS-116 The facility name on the application must match the name of your business entity registered with the IRS for the facility's federal tax identification number (e.g., EIN, TIN). If your facility is doing business as (dba) under a name that is not the same as the facility name, you must provide this CERTIFICATE OF QUALIFICATION & LABORATORY DIRECTORSEE MORE ONWADSWORTH.ORG
MEDICAL MARIJUANA
REVISED May 2019 - Solicitation of Interest for Laboratories to Perform Testing of Medical Marijuana Products in New York State Guidance Procedure Microbiology - Medical Marijuana guidance procedure for a DOC Methods List of Approved Methods and Analytes - Certification Manual Item 180.7 NYS DOH LINC-250, revision 2 - Metals and Metalloids in Medical Marijuana Products by SPINAL CORD INJURY RESEARCH The Extramural Grants Administration Program works to further the efforts of the Spinal Cord Injury Research Board (SCIRB). Each year, approximately 1,100 New York residents suffer traumatic spinal cord injuries joining the nearly 288,000 people living in the United States with paralysis. SCIRB’s mission, since 1998, has been to support neurological spinal cord injury GUIDELINES FOR REMOTE BLOOD STORAGE NEW YORK STATE COUNCIL ON HUMAN BLOOD AND TRANSFUSION SERVICES Membership Roster – 2008 Donna Skerrett, M.D., M.S., Chairperson * Director, Transfusion Medicine and WADSWORTH CENTER, NEW YORK STATE DEPARTMENT OF HEALTHPUBLIC HEALTH PROGRAMSRESEARCHREGULATORY PROGRAMSEDUCATIONEXTRAMURAL FUNDINGABOUT US Wadsworth Center, at the forefront of the fight against tuberculosis in NYS and globally, celebrates World TB Day 2021 The World Health Organization estimates that almost 4,000 people lose their lives to TB and close to 28,000 people contract TB every day. CLINICAL LABORATORY EVALUATION PROGRAM How can the New York State Department of Health Clinical Laboratory Evaluation Program help you today? Laboratory Standards CLEP Program Guide (July 2020) Apply for a Permit Permit Modifications Health Commerce System (HCS) Limited Service Laboratories Certificate of Qualification (CQ) Applications Cytotech Registrations Lab Developed Test (LDT) Approval On-Site Survey LABORATORY STANDARDS Current Standards May 2021 revisions to the General Systems Standards and Specialty Requirements by Category, for Microbiology and Cytopathology, may be viewed below under the heading of Revisions to Laboratory Standards - May 2021.SENIOR STAFF
Director and Quality Assurance Officer, Environmental Inorganic Chemistry Laboratory; Derek Symula, Ph.D. Director, Division of Laboratory Quality Certification BLOOD RESOURCES PROGRAM More than 350 facilities are approved by the department to provide blood services. One and a quarter million blood components are prepared annually. Approximately 90% of the blood is collected at blood centers, with the remainder collected by approximately about a dozen hospitals and a small number of independent facilities. Informational materials on blood services, including guidelines andINITIAL APPLICATION
These items are required for initial application: CLIA Application for Certification Form CMS-116 The facility name on the application must match the name of your business entity registered with the IRS for the facility's federal tax identification number (e.g., EIN, TIN). If your facility is doing business as (dba) under a name that is not the same as the facility name, you must provide this CERTIFICATE OF QUALIFICATION & LABORATORY DIRECTORSEE MORE ONWADSWORTH.ORG
MEDICAL MARIJUANA
REVISED May 2019 - Solicitation of Interest for Laboratories to Perform Testing of Medical Marijuana Products in New York State Guidance Procedure Microbiology - Medical Marijuana guidance procedure for a DOC Methods List of Approved Methods and Analytes - Certification Manual Item 180.7 NYS DOH LINC-250, revision 2 - Metals and Metalloids in Medical Marijuana Products by SPINAL CORD INJURY RESEARCH The Extramural Grants Administration Program works to further the efforts of the Spinal Cord Injury Research Board (SCIRB). Each year, approximately 1,100 New York residents suffer traumatic spinal cord injuries joining the nearly 288,000 people living in the United States with paralysis. SCIRB’s mission, since 1998, has been to support neurological spinal cord injury GUIDELINES FOR REMOTE BLOOD STORAGE NEW YORK STATE COUNCIL ON HUMAN BLOOD AND TRANSFUSION SERVICES Membership Roster – 2008 Donna Skerrett, M.D., M.S., Chairperson * Director, Transfusion Medicine and NEW YORK STATE NEWBORN SCREENING PROGRAM Newborn screening refers to medical tests, the majority of which are genetic, performed to identify babies with certain disorders, which without intervention, may permanently impact newborns and their families. Early recognition and treatment of most of these disorders leads to a better outcome for the newborn. The Newborn Screening Program’s goal is to help affected babies PHYSICIAN OFFICE LABORATORY EVALUATION PROGRAM The New York State Department of Health Physician Office Laboratory Evaluation Program (POLEP), under contract with the Center for Medicare and Medicaid Services (CMS), is responsible for managing activities related to administration of the federal program implementing the Clinical Laboratory Improvement Amendment of 1988 (CLIA) for physician office laboratories (POL) CYTOTECHNOLOGIST REGISTRATION New York State Public Health Law requires that all individuals employed as cytotechnologists at clinical laboratories applying for or holding a New York State permit be registered with the Department. New York State Education Law also requires that cytotechnologists be licensed. More information regarding licensure is available at State Education Department, Office of the LDT FAQ | NEW YORK STATE DEPARTMENT OF HEALTH, WADSWORTH This database only contains laboratory developed tests (LDTs) that were submitted to New York State's Department of Health (NYS DOH) for review and were subsequently approved. Tests that are either FDA approved, were not submitted to NYS DOH, or have not been approved forENCEPHALITIS
Viral encephalitis encompasses a group of inflammatory diseases of the brain caused by a variety of viral agents and exhibiting common clinical symptoms. Symptoms may include headache, confusion, or other alterations in sensorium and / or behavior, nausea, or vomiting. Signs may include evidence of elevated intracranial pressure or meningeal irritation, cranial nerve palsies, paresis or FORMS | NEW YORK STATE DEPARTMENT OF HEALTH, WADSWORTH CENTER General Request Newborn Screening Materials Cystic Fibrosis Referral Appointment Confirmation Form Follow-Up Summary Form Hemoglobin Referral Appointment Confirmation Form Newborn Screening Transport Form Refusal of Diagnostic Testing for Cystic Fibrosis Form Refusal of Newborn Screening for Religious Reasons Form Request for Newborn Screening Results Diagnostic MICHELE CAGGANA, SC.D., FACMG Dr. Michele Caggana joined the Wadsworth Center in 1996 after receiving her doctoral degree from the Harvard School of Public Health and completing post-doctoral work in molecular virology at Wadsworth Center and clinical molecular genetics at the Mt. Sinai School of Medicine. Dr. Caggana is Chief of the Laboratory of Human Genetics and was appointed as Director of the Newborn KIRSTEN ST. GEORGE, PH.D. Dr. St. George is Director of Virology and Chief of the Laboratory of Viral Diseases (LVD) at the Wadsworth Center and a Clinical Professor in the Biomedical Sciences Department at the School of Public Health, University at Albany. The LVD comprises several service laboratories and research teams including general virology and specialty referenceservices in viral
NEW YORK STATE DEPARTMENT OF HEALTH WADSWORTH … A. Has the director, any assistant director(s), or those having a direct or indirect ownership interest, controlling interest, or corporate membership in the applying clinical facility ever been charged with violations of local, state or federal ALEXEY KHODJAKOV, PH.D. We use laser microsurgery and sophisticated imaging techniques (light- and electron microscopy) to study molecular mechanisms responsible for faithful segregation of chromosomes during mitosis (cell division). The mitotic apparatus, a.k.a. the spindle, is a dynamic, self-assembling molecular machine and the goal of mitotic spindle assembly is to connect each chromosome to the Your browser does not support iFrames MAY 14, 2020 | 4:30 pm Information on Novel Coronavirus NY State remains on PAUSE through May 15. All non-essential workers are directed to work from home, and everyone is required to wear a face covering and maintain a 6-foot distance from others in public.Get the Facts
Submit
* Services
* News
* Government
* Local
* Location
* Translate
Select Language▼SERVICES
MY SERVICES SEE ALL
NEWS
GOVERNMENT
RECOMMENDED SEARCH RESULTS RECOMMENDED SEARCH RESULTSENTER LOCATION
ENTER LOCATION
Your location has been updated Skip to main content DEPARTMENT OF HEALTH, WADSWORTH CENTER__
* Public Health Programs * Environmental Health * Emergency Response * Environmental Biology * Environmental Organic Chemical Analysis * Inorganic Analytical Chemistry* Nuclear Chemistry
* Radon
* Radon Indoors
* Radon in Water
* Proficiency Testing * Infectious Diseases * Laboratory Response Network * Electronic Test Request/Reporting* Enrollment
* Test Catalog
* Request Tests
* Access Reports
* Packaging and Shipping * Help and Frequently Asked Questions* Arbovirology
* Antimicrobial Resistance Laboratory Network* Bacteriology
* Biodefense
* Bloodborne Viruses * Cellular Immunology * Diagnostic Immunology* Mycobacteriology
* Mycology
* Parasitology
* Rabies
* Virology
* Laboratory Services * Specimen Collection & Shipping * Anti-viral Resistance* Arbovirus Testing
* Encephalitis
* Enteric Viruses
* Exanthemous Viruses * Respiratory Viruses * Sexually Transmitted Diseases * Vaccine Preventable Disease Reference Center * Training & Education * Research & Development * Infectious Diseases Requisition Form * Newborn Screening and Genetics * Newborn Screening Program * Screened Disorders * Information for Families * Frequently Asked Questions for Families * Specimen Usage, Retention and Security Policies * Genetic Counseling Information * Guidelines to Opt-out * Information for Healthcare Providers * Overview of Laboratory Testing * Specimen Collection* Obtaining Results
* Follow-up Process
* Forms
* Request Newborn Screening Materials * Residual Blood Spot Usage * Frequently Asked Questions * Hospital Contacts and Designee * Notes for Hospitals * Specialty Care Centers * Becoming A Specialty Care Center * Search Specialty Care Centers * Laws & Regulations* Funding
* Selected Publications* History
* New York Mid-Atlantic Consortium* Research
* Research Areas
* Laboratories
* Cores
* Office of Research & Technology * Scientific Resources * Regulatory Programs * Clinical Laboratory Evaluation Program * Environmental Laboratory Approval Program * Physician Office Laboratory Evaluation Program * Breath & Blood Alcohol Testing * Blood Resources Program * Tissue Resources Program * Regulated Medical Waste Program * Laboratory Investigations Unit* Education
* Wadsworth Center Fellowship Program* Postgraduate
* APHL/CDC Fellowships * NIH Biodefense & Emerging Infectious Diseases Training Program * Postdoctoral & Visiting Scientists* Graduate
* School of Public Health Environmental Health Sciences * School of Public Health Biomedical Sciences* Undergraduate
* Research Experience for Undergraduates * Student Volunteers * Extramural Funding * Breast Cancer Research & Education * New York State Stem Cell Science - NYSTEM * Multiple Sclerosis * Spinal Cord Injury Research* About Us
* Affiliations
* Director's Office
* Employment Opportunities* History
* Landmark Achievements* News and Events
* Senior Staff
* Contact Us
* Privacy Policy
SEARCH FORM
Search
VIROLOGY
LABORATORIES
INORGANIC ANALYTICAL CHEMISTRYMYCOBACTERIOLOGY
CELLULAR IMMUNOLOGY
MYCOLOGY
ABOUT US
LABORATORY GUIDANCE DOCUMENTS FOR CORONAVIRUS DIAGNOSTIC IMMUNOLOGY NEWBORN SCREENING AND GENETICS BIOTHREAT ENVIRONMENTAL TESTINGEMERGENCY RESPONSE
ENVIRONMENTAL LABORATORY APPROVAL PROGRAMPARASITOLOGY
PUBLIC HEALTH PROGRAMS BLOOD RESOURCES PROGRAMBLOODBORNE VIRUSES
RABIES
SPINAL CORD INJURY RESEARCH SCIENTIFIC RESOURCES CLINICAL LABORATORY EVALUATION PROGRAM RESEARCH AND DEVELOPMENTBIODEFENSE
ARBOVIROLOGY
CORES
MULTIPLE SCLEROSIS
LABORATORY RESPONSE NETWORK PHYSICIAN OFFICE LABORATORY EVALUATION PROGRAM BREAST CANCER RESEARCH & EDUCATIONBACTERIOLOGY
RESEARCH AREAS
ENVIRONMENTAL BIOLOGYJOIN OUR TEAM
NUCLEAR CHEMISTRY
CLINICAL TESTING
REGULATORY PROGRAMS
NEW YORK MID-ATLANTIC CONSORTIUM ENVIRONMENTAL ORGANIC CHEMICAL ANALYSIS OFFICE OF RESEARCH & TECHNOLOGY BREATH & BLOOD ALCOHOL TESTING LABORATORY INVESTIGATIONS UNITPROFICIENCY TESTING
YOU ARE HERE
SCIENCE IN THE PURSUIT OF HEALTH®NEWS
FDA CORONAVIRUS FACT SHEETS General Cross-Validation Plan SARS-CoV-2 Assay: EZ1 to eMAG and easyMag NY SARS-CoV-2 Fact Sheet for Healthcare Providers - New York SARS-CoV-2 Real-time RT-PCR Diagnostic Panel NY SARS-CoV-2 Fact Sheet for Patients - New York SARS-C0V-2 Real-time RT-PCR... read more SEVEN FELLOWS, ONE GOAL Wadsworth Center has long been committed to providing the next generation of public health professionals with the necessary knowledge, skills and abilities. More than 300 students have made Wadsworth Center their summer home over the course of nearly 30...read more
DR. KIMBERLEE MUSSER APPOINTED DIRECTOR OF CLINICAL TESTING FOR WADSWORTH CENTER’S DAVID AXELROD INSTITUTE Wadsworth Center’s David Axelrod Institute, and all of Wadsworth Center for that matter, may be known to many as a place for research and education. But there’s a lot of clinical testing that goes on here too. You could even say the work performed here... read more* MORE NEWS
LABORATORY GUIDANCE DOCUMENTS FOR CORONAVIRUS Information for laboratories including interim guidelines for collection, handling, and testing clinical specimens from patients under investigation and biosafety guidelines for handling and processing specimens associated with the novel coronavirus can be found at https://www.cdc.gov/coronavirus/2019-ncov/lab/index.html.
_
ABOUT US
JOIN OUR TEAM
Innovative, multidisciplinary research. Complex diagnostics and state-of-the-art laboratory services. We merge clinical and environmental testing with fundamental, applied and translationalresearch. [
DEPARTMENT OF HEALTH, WADSWORTH CENTER PUBLIC HEALTH PROGRAMS * Emergency Response * Environmental Biology * Environmental Organic Chemical Analysis * Inorganic Analytical Chemistry* Nuclear Chemistry
* Proficiency Testing * Laboratory Response Network * Electronic Test Request/Reporting* Arbovirology
* Bacteriology
* Antimicrobial Resistance Laboratory Network* Biodefense
* Bloodborne Viruses * Cellular Immunology * Diagnostic Immunology* Mycobacteriology
* Mycology
* Parasitology
* Rabies
* Virology
* Newborn Screening Program * New York Mid-Atlantic ConsortiumRESEARCH
* Research Areas
* Laboratories
* Core Facilities
* Office of Research & Technology * Scientific ResourcesREGULATORY PROGRAMS
* Clinical Laboratory Evaluation Program * Environmental Laboratory Approval Program * Physician Office Laboratory Evaluation Program * Breath & Blood Alcohol Testing * Blood Resources Program * Tissue Resources Program * Regulated Medical Waste Program * Laboratory Investigations UnitEDUCATION
* Wadsworth Center Fellowship Program * APHL/CDC Fellowships * NIH Biodefense & Emerging Infectious Diseases Training Program * Postdoctoral & Visiting Scientists* Graduate
* Research Experience for Undergraduates * Student VolunteersEXTRAMURAL FUNDING
* Breast Cancer Research & Education * Multiple Sclerosis * New York State Stem Cell Science * Spinal Cord Injury ResearchABOUT US
* Affiliations
* Director's Office
* Employment Opportunities* History
* Landmark Achievements* News and Events
* Contact
* Senior Staff
* Privacy Policy
* Site Map
* NYS Department of Health Your browser does not support iFrames* Agencies
* App Directory
* Counties
* Events
* Programs
* Services
ORIGINAL TEXT
Contribute a better translation -------------------------Details
Copyright © 2024 ArchiveBay.com. All rights reserved. Terms of Use | Privacy Policy | DMCA | 2021 | Feedback | Advertising | RSS 2.0